Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Public Health
versão impressa ISSN 2504-3137versão On-line ISSN 2504-3145
Port J Public Health vol.37 no.1 Lisboa 2019
https://doi.org/10.1159/000497249
REVIEW ARTICLE
Born Preterm: A Public Health Issue
Nascer pré-termo: Um problema de Saúde Pública
Filomena Pinto a c Eduardo Fernandes c Daniel Virella d Alexandre Abrantes a b Maria Teresa Netod,e
a National School of Public Health, NOVA University of Lisbon, Lisbon, Portugal
b Department of Health Policy and Management, CISP – Public Health Research Centre, National School of Public Health, NOVA University of Lisbon, Lisbon, Portugal;
c Neonatal Intensive Care Unit, Dr. Alfredo da Costa Maternity, Central Hospitalar Lisbon Centre, EPE, Lisbon, Portugal
d Neonatal Intensive Care Unit, Hospital Dona Estefânia, Central Hospitalar Lisbon Centre, EPE, Lisbon, Portugal
e NOVA Medical School I Faculdade de Ciências Médicas, NOVA University of Lisbon, Lisbon, Portugal
ABSTRACT
Background: Prematurity accounts for about 10.6% of neonates worldwide and tends to increase as does survival from lower gestational ages. - Summary: The importance of preterm birth in public health stems from its link to infant and under-5 mortality, morbidity, and its economic impact. In both the short and long term, preterm birth consequences are inversely related with gestational age and carry a higher risk of mortality and morbidity with neurodevelopmental, sensorial, cognitive and physical health disturbances. Individuals needing lifelong support pose challenges to the responsiveness of health services and community systems. Public health can be decisive in prematurity prevention, providing data to policy-makers and reducing modifiable risk factors. This paper focuses on the long-term consequences of preterm birth and possible public health measures to tackle them. - Key Messages: Addressing social determinants of health can have the highest impact on prematurity outcomes.
Keywords: Morbidity Premature birth Prematurity Public health Very low birth weight
RESUMO
Introdução: A nível mundial a prevalência da prematuridade (10.6%) é crescente bem como a sobrevivência em idades gestacionais menores. Sumário: A importância da prematuridade prende-se com o contributo para a mortalidade infantil, para a inferior aos 5 anos e para a morbilidade, com impacto económico. A curto e longo prazo, as consequências do nascimento pré-termo relacionamse inversamente com a idade gestacional, comportam maior risco de alterações neurológicas, sensoriais, cognitivas, comportamentais e na saúde geral. A necessidade de apoio pode prolongar-se ao longo da vida, põe à prova os serviços de saúde e comunitários. A Saúde Publica pode ser decisiva na prevenção da prematuridade, fornecendo aos decisores dados que permitam a redução de riscos modificáveis. Os autores focam-se nas consequências a longo prazo e na abordagem da prematuridade no âmbito da Saúde Pública. Mensagens chave: A intervenção nos Determinantes Sociais de Saúde poderá ter impacto decisivo no prognóstico da prematuridade.
Palavras-chave: Morbilidade · Muito baixo peso · Nascimento pré-termo · Prematuridade · Saúde pública
Preterm birth (PB) is defined as birth occurring before 37 weeks or 259 days of gestational age (GA) 1. Prematurity is the main cause of an estimated one million neonatal deaths globally every year and a significant contributing factor to morbidities extending to adulthood [2 ,3 ). PB is sub-classified according to GA at birth into extremely preterm (< 28 weeks), very preterm (28 to < 32 weeks), and moderate to late preterm (32 to < 37 weeks) 4.
The risk of PB is considerable for both the higher and lower-income countries. However, there is a dramatic difference in survival of premature babies depending on where they are born. In high-income countries, half of the babies born at 24 weeks survive, while in low-income countries, half of those born at 32 weeks still die due to a lack of essential newborn (NB) care 5. In developing countries, the higher PB rate is commonly associated with infection (malaria, HIV) and malnutrition 6 . In most affluent countries, the improvement of socioeconomic conditions, technological development, reproductive medicine’s success, and high quality of perinatal care contributed to a heterogeneous prematurity rate but also to an increased survival of extreme premature infants and a decreasing viability threshold [7 - 10 ].
Survival to PB led to the emergence of a population of complex patients with different comorbidities, whose severity and frequency are inversely associated with GA [11 - 17 ]. Some of the classic pathologies related to prematurity have diminished significantly in the last decades: hyaline membrane disease, bronchopulmonary dysplasia, necrotizing enterocolitis (NEC), and retinopathy of prematurity (ROP). Yet, neurological morbidity, more specifically intraperiventricular hemorrhage (IPVH), presented no parallel decrease [4 ,12 ,14 ,18 ,19 ].
Low birth weight (LBW) is defined as the weight less than 2,500 g, very low birth weight (VLBW) less than 1,500 g, and extreme low birth weight (ELBW) less than 1,000 g 1. LBW can occur as a result of restricted fetal growth or PB. LBW infants have a greater risk of poor outcome 20; however, GA is a better predictor of mortality and morbidity than birthweight [21 - 23 ].
Children born prematurely, especially those of extreme PB, have an increased risk of lifelong effects on neurodevelopment with motor, cognitive, sensorial, physical, behavioral and psychopathological disturbances, compared with their term peers [24 - 26 ]. They require differentiated clinical care, early intervention, special educational programs, and social interventions, which burden families and health and community support systems [8 ,27 - 31 ].
PB is a public health (PH) issue due to its worldwide prevalence, increasing survival rate, short and long-term morbidity, high economic burden, and because it contributes to infant and under-5 mortality rates. This wide-ranging impact has demanded attention in many high-income countries [15 ,19 ,29 ,32 - 35 ].
The enlarged concept of “people born preterm” can help healthcare providers to understand their long-lasting needs, provide access to health resources available to support postnatal infant development, and improve awareness of atypical clinical profiles in the long-run 36 . This review addresses PB as a PH matter focusing on its life-long consequences and strategies to tackle them.
Prematurity: Prevalence
According to Chawanpaiboon et al.37 , the global estimated preterm rate is increasing, ranging from 9.8% in 2000 to 10.6% in 2014. The estimated regional PB rates ranged from 13.4% in North Africa to 8.7% in Europe. Even though 81% of these PB occurred in Asia and Sub-Saharan Africa, data from some high-income and high-middle-income countries suggest that their PB rates are also increasing [37 ,38 ]. According to data available from 67 countries, prevalence was higher in moderate or late preterm (84.7%), followed by very preterm (11.3%) and extremely preterm infants (4.1%) [4 ,37 ]. However, there are some challenges to the global interpretation of preterm epidemiology that can interfere with PB rate estimation: diverse assessment methods of GA, prematurity definitions, data availability, and varying quality of vital statistics between countries [4 ,37 ,39 ].
The 2015 European Perinatal Health Report [38] pointed out that prematurity rates are heterogeneous but may be increasing in most European countries when comparing 2010 with 2015, where they range between 6 and 12% of live births [20 ,38 ,40 ,41 ] (Fig. 1 ). The reasons for these trends are not always understood. A range of factors have been put forward: GA assessment criteria, multiple pregnancy rates, late maternal age, extreme body mass indexes, and socioeconomic issues, with lower-income families being at higher risk [4 ,32 ,34 ,38 ,41 ]. Some high- and middle-income countries have increased numbers of provider-initiated PBs [20 ,42 ]. Probably, higher survival has changed the perception of risks associated with prematurity, which led to an increase in non-spontaneous births 41 .
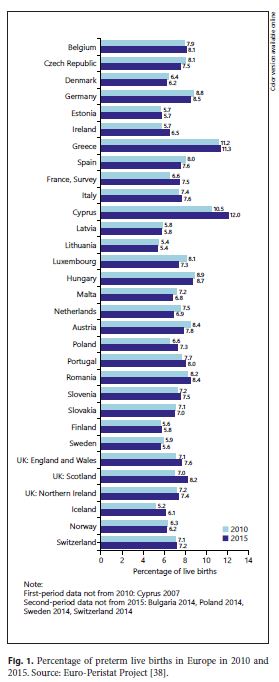
It is estimated that 15–20% of all births worldwide are LBW. In almost all OECD countries, the proportion of LBW infants has increased over the past two decades, being 6.5% in 2015, representing a rise of 15% between 1990 and 2015, mainly due to increase in PBs. [20] (Fig. 1)
In Portugal, prematurity has remained relatively stable in the second decade of the 21st century. According to Statistics Portugal (INE), the prematurity rate was of 7.8% in 2012 and 2013, 7.7% in 2014, 8% in 2015, and 7.8% in 2016 [20 ,43 ]. According to 2017 OECD indicators of health outcomes, Portugal registered an increase of 59% of LBW infants since 1990 20.
Prematurity: Risk Factors
Prematurity can be the result of a complex combination of medical, biological, genetic, psychosocial, and environmental conditions 44 . However, in about two-thirds of the cases the cause remains unknown, while the other third occurs upon medical recommendation for a wide-range of maternal and fetal pathologies [42 ,45 ,46 ].
Amongst multiple maternal risk factors, the following are emphasized: previous preterm deliveries, pregnancy at youngest or latest ages, short intervals between pregnancies, nutrition, lifestyle (physical activity, stress, workload), individual behavior (alcohol, tobacco, illicit drugs), social issues (unemployment, social support, relationship status, personal resources), medical maternal and pregnancy conditions (chronic hypertension, hypertensive disorders of pregnancy, diabetes mellitus, cardiac, respiratory, renal, autoimmune diseases, hyperthyroidism, infection), and infertility treatments [5 ,66 ,47 ].
Better understanding of the causes of PB will advance the development of solutions to prevent it 46 .
Prematurity: Impact on PH
Mortality
Globally, circa 44% of deaths in children under under-five occurred in the neonatal period, PB being the commonest cause of neonatal death [20 ,48 ,49 ]. Approximately 1 million children die each year due to complications of PB [2 ,20 ].
Each additional week of gestation results in an increased survival rate [29 ,49 ]. The GA survival threshold, defined as long-term survival above 50% 50 , has been decreasing. In some high-income countries, it is as low as 23 weeks 49 . Survival without major morbidity in VLBW infants is increasing, ranging from 53 to 71% in different studies [49 ,51 ,52 ].
The overall in-hospital survival differs among neonatal networks from 78 to 93% – range at 24 weeks 35–84% and at 29 weeks 92–98% 53 . Stoll et al. [12] described survival at discharge of 6% at 22 weeks and 92% at 28 weeks. In a study from Canada, Johnston et al. 138 estimated similar survival rates amongst live births at age 2 and 10, circa 56.0% survival at < 28 weeks, 92% at 28–32 weeks, and 98% at 33–36 weeks. Cheong et al. 54 describe a 73% survival at 8 years of a corrected age in a cohort of extreme preterm [11 ,12 ].
In a Swedish population-based study, Crump et al. [25] described that, among individuals born in the 1973–1979 period, low GA at birth was independently associated with increased mortality in young adulthood .
The survival threshold in Portugal is 25 weeks. According to the Portuguese Very Low Birth Weight Infant Registry, in 2013, the overall survival rate was 89%. Per GA intervals, survival rates were 95.3% for 28 and 31 weeks, 77.6% for 25 to 27 weeks, and 42.1 and 16.7% for 24 and 23 weeks, respectively 55 .
Reducing mortality associated with complications of prematurity will be crucial to reduce worldwide under-five mortality.
Morbidity
Improved survival of preterm infants leads to short- and long-term higher risk of neurological, cognitive, sensorial, respiratory, digestive, renal, cardiovascular, metabolic, immune, and psychosocial disturbances [4 ,22 ,25 ,26 ,36 ,56 ,57 ]. We will focus preferentially on those morbidities that are life-long in extreme and very preterm infants.
Neurodevelopmental impairment (NDI) is displayed in 5% of all PBs 58 . Survival without NDI at 2 years of age has become a common benchmark for success. While most neurodevelopmental disabilities are not major deficits, they should be clinically considered 59 . Some of these morbidities are not evident at discharge or even at 2 years of age 59 . Therefore, long-term follow-up is needed to properly evaluate the consequences of PB.
In the early years of life, preterm infants often continue to experience physical health problems that may require frequent medical visits and re-hospitalization. This may limit their participation in regular childhood activities, which may in turn affect their social skills.
Neurological morbidity is responsible for a high risk of cognitive, motor, and sensory impairment. Periventricular leukomalacia (PVL), IPVH, and post-hemorrhagic hydrocephaly are the main determinants of neurodevelopmental outcomes [17 ,22 ,60 - 65 ]. Motor deficits in children born preterm are generally identified earlier, but some of them are transitory (poor head control, hypotonia, or hypertonia) disappearing near 12 months 66 . However, some psychomotor disturbances will initially be clinically silent 67 . Different studies among VLBW/ELBW children at school-age 68 and ELBW children at ages 11–13 years 61 found a higher likelihood for developmental coordination disorder and an increased risk for long-term motor impairment. Preterm infants surviving into adulthood after neonatal IPVH or PVL continue to be at very high risk of poor neurological outcomes 69 .
Cerebral palsy (CP) is one of the major neurological complications of premature birth. Survivors of extreme prematurity have rates of CP in childhood 70–80 times higher than those in term infants [17 ,70 - 73 ]. Studies of time trends in the prevalence of CP have shown different results [37 ,70 ,73 - 75 ]. For children born in Portugal in 2001–2007, the prevalence of CP at 5 years of age was estimated to be 4.6-fold higher in children born at 32–36 weeks’ gestation, 45.1-fold higher in those born at 28–31 weeks, and 70.1-fold higher in children born at < 28 weeks’ gestation, compared with children born at term (Table 1 ) 76 .

Diagnosis of high CP risk can be established by 6 months of corrected age with a combination of medical history, standardized motor assessment and neuroimaging [15 ,17 ]. Most studies of VLBW infants show cognitive deficits (lower intelligence quotient, intellectual, learning disabilities, and executive dysfunction), academic underachievement and grade failures 77 . Early referral to CP-specific early intervention is critical to improve functional outcomes.
ROP is the main cause of visual deficit in preterm infants 78 . They have an increased risk of long-term visual disorders due to the combination of development interruption of the visual system due to preterm delivery and/or neurological complications, oxygen toxicity, infection, glycemia disorders, undernutrition, and genetic factors [15 ,77 - 82 ]. ROP is a progressive disease characterized by fibrovascular proliferation at the periphery of the retina with risk of retinal detachment. It has a multifactorial etiology, prematurity being its main determinant [83 - 85 ]. It is recorded in about 2–11% of VLBW infants [77 ,83 ]. The rate of blindness and/or severe visual impairment is inversely related to GA (1–2% at 26–27 weeks and 4–8% at ≤25 weeks) 77 . Myopia and hypermetropia arise in at least a quarter of children born < 28 weeks 86 . The need to prescribe glasses is also related to GA, with 24% at the age of 6 < 26 weeks’ GA wearing glasses versus 4% of term controls. This difference continues into adolescence and adulthood [87 ,88 ]. A high rate (4–5%) of late retinal detachment in ELBW infants during their late teens has been observed [79 ,81 ,88 ].
Hearing impairment has a detrimental effect on the development of language, learning, communication skills, quality of life, and on economic independence in adult life [89 ,90 ]. VLBW infants have central auditory processing difficulties, including in discerning simple speech sounds and worse auditory recognition than their full-term counterparts 91 ; a prevalence of 0.1–0.2% has been reported 92 , and may be 10–50% higher in VLBW infants [77 ,87 ,92 ]. Hypoxia, hyperbilirubinemia, infections, ototoxic therapeutics, and exposure to noise can irreversibly damage the cochlear, vestibular organs, the auditory nerve, and cortex 92 . Hearing loss may be progressive and diagnosed late in life (2–4 years of age), requiring long-term surveillance to timely introduce hearing aid wear [92 ,93 ].
Cognitive deficit is the most prevalent disability in the population of preterm children. Very preterm survivors have high rates of cognitive dysfunction and emotional troubles at school age that affect academic functioning and progress 94 . Case-control studies have shown that very preterm children have significantly lower intelligence quotient scores than term peers. The most significant neonatal risk factors are severe IVH, PVL, neonatal seizures, NEC, and long-term ventilation 95 . Cognitive dysfunctions seem influenced by environmental factors such as parental socioeconomic status and education [95 ,96 ]. Follow-up revealed that many of the difficulties persisted into adolescence and early adulthood 94 . Long-term monitoring will be important to identify later cognitive impairment and educational needs.
Language disorders have been reported in children born preterm in its different domains (receptive, expressive, articulation). Several biologic and environmental factors influence language outcomes. Regular exposure to speech is essential to auditory cortex and speech development, social interaction and later school achievement [97 ,98 ].
Behavioral and psychopathological disturbances are more prevalent in individuals born preterm and are also inversely related to the GA. These disturbances often coexist with motor, language, and cognitive problems [51 ,99 - 102 ].
The risk of behavioral problems, such as attention deficit hyperactivity disorder, is increased by 2.6–4 times in very preterm infants in early childhood. They also often present more school problems and anxious and depressive symptoms, risk of hospitalization for mental disorders and borderline behavioral problems while transitioning to adolescence. However, they were less prone to delinquency and risk-seeking behaviors than control young adults [16 ,99 ,103 ]. Lower self-esteem, lower employability, and lower income have also been reported as frequently found in adulthood [104 ,105 ]. Nevertheless, the self-perception of the quality of life seems to be positive 16. The relevance of the screening for behavioral and psychiatric changes and timely intervention in this area is emphasized.
Bronchopulmonary dysplasia is defined by oxygen dependence after 28 days of life or 36 weeks of postmenstrual age 106 . Arrested vascularization and augmented vasoreactivity may lead to the development of pulmonary hypertension in the weeks and months after PB [107 - 109 ]. It has a prevalence of about 40% in infants born at GA < 28 weeks 110 . It is associated with invasive ventilation, oxygen toxicity, pre- and postnatal infection, nutritional deficit, and possible genetic susceptibility [106 ,111 ]. In the long term, respiratory impairment, particularly if exposed to noxious substances, has been observed resulting in changes in the respiratory function, bronchial hyperreactivity, sometimes with an atypical clinical profile (asthma-like), reduced tolerance to exercise, increased risk of respiratory infections needing hospital admission, pulmonary hypertension, some needing ventilation support at home [19 ,30 ,106 ,112 ].
Intestinal failure is a reduction in the functional gut mass below a critical threshold necessary to maintain growth, hydration, and electrolyte balance 113 . The leading cause of intestinal failure in neonates is anatomic short bowel syndrome, and NEC inflammation and necrosis of the intestinal wall are its predominant cause in VLBW infants [102 ,113 - 115 ]. The long-term need for parenteral nutrition is associated with cholestasis, hepatic insufficiency, long-term central catheters, and higher risk of infections [15 ,116 ]. Besides a high lethality (15–60%), advances in parenteral nutrition, infection prevention, surgery techniques, and transplantation have improved prognosis 117 .
High-risk renal long-term outcomes in preterm NB appear to be associated with early acute renal injury [115 ,118 ]. PB occurs in the active phase of nephrogenesis, resulting in fewer nephrons. Nephrotoxic medications can potentiate kidney function impairment, increasing the risk of chronic kidney disease [119 ,120 ]. Acute neonatal kidney injury risk is around 12–39%, even though its connection with chronic kidney disease is not yet clear 118 . These patients should be monitored regularly for long-term kidney damage [121 ,122 ].
Cardiometabolic syndrome (CMS) is a combination of metabolic dysfunctions characterized by resistance to insulin, diminished tolerance to glucose, dyslipidemia, hypertension, and intra-abdominal adiposity 123 . Barker et al. [124 ,125 ] suggested, in the late 1990s, the inverse correlation between LBW, risk of cardiovascular disease, and type 2 diabetes in adult life. Variations in fetal and postnatal nutrition could be linked to the modification of genetic expression, resulting in the programming of long-term chronic disease [126 ,127 ]. In the long run, CMS is also reinforced by lifestyle: less physical exercise and inadequate diet. Adequate interventions may reduce the impact of this pathology [126 ,128 ].
Venous thromboembolism (VTE) is considered to be a complex disorder influenced by several genetic and environmental factors and affects circa 1 in 1,000 individuals per year 129 . Zöller et al. 130 , found that low GA at birth was associated with an increased risk of VTE in infancy, early childhood, and young adulthood, suggesting that PB could be an important risk factor for later VTE [129 ,131 ].
Disorders of the immune system can account for the high susceptibility of preterm neonates to infection [132 - 135 ]. PB interrupts the fetal acquisition of passive immunity and favors a different, precocious dermal and gastrointestinal microbiome acquisition, different from that of term neonates [132 - 137 ]. Better understanding of the characteristics of the immune system in preterm infants is challenging 135 .
Economic Impact
The economic impact of prematurity is assumed to be high, including not only the immediate neonatal intensive care costs but also ongoing long-term complex healthcare needs. The complexity of the clinical situation of some survivors, particularly those of earlier GA, determines the need for concerted multidisciplinary action and a consequent response by health, social, community, rehabilitation and special education departments.
The US Institute of Medicine (IOM) published in 2007 the report “Preterm Birth, Causes, Consequences and Prevention”, in which the estimated economic impact (medical, educational and lost productivity combined) of PB in the USA in 2005 accounted for USD 26.2 billion or USD 51,600 for each child born prematurely 44 . The average first-year medical costs, including both inpatient and outpatient care, were about 10 times greater for preterm (USD 32,325) than for term infants (USD 3,325). The average length of stay was nine times longer for a preterm NB (13 days), compared with a baby born at term (1.5 days) 44 .
Johnston et al. 138 concluded in 2014, that PB resulted in significant morbidity, mortality, and high costs for health services in Canada. Although the highest expenditure was concentrated in the neonatal period, the costs and resources use continued throughout childhood. Whilst the largest group was composed of moderately premature infants, it was the group of extreme prematurity that cost the most 138 .
A systematic review by Petrou et al. 139 on the long-term costs of prematurity after hospital discharge concluded that PB and LBW result in substantial costs for health systems, special education, social services, family and caregivers, and society in general.
Blencowe et al. 56 estimated that in 2010 all PBs worldwide would have been responsible for 77 million disability-adjusted life years, accounting for 3.1% of the global total, of which 3 million would be years lived with disability.
The evaluation of direct healthcare costs associated with prematurity encompasses both hospital admissions (initial and subsequent) and those related to outpatients (therapies, clinical, educational, and social support). Out-of-pocket expenses were substantial and significantly higher for very preterm and VLBW infants 5. The indirect costs (loss of productivity of the caregivers, emotional stress, and repercussion on the families and quality of life) although more difficult to assess, should be considered to allow for a more comprehensive quantification of the burden of individuals surviving prematurity. Several authors have drawn attention to the high social costs from a family perspective [28 ,140 ,141 ].
MPT Infants
Babies born MPT account for more than 80% of all PB, of which the majority remained without disabilities in the long-term; however, when compared to full-term infants, they have increased mortality and experience higher morbidity, which had been underestimated in the past 138 .
Johnston et al. 138 describe a survival rate of 93% at age 2 and 10 in moderate, and 98% in late preterm infants; the majority of children remained disability-free at age 10.
MPT however, have higher rates of mortality and short-term morbidity. Worse neurodevelopmental and school performance outcomes, increased risk of CP and costs per infant than their term peers, have been pointed out 138 . Other common neonatal morbidities in these infants include hypothermia (known to be associated with worse outcomes), hypoglycemia (increasing likelihood of long-term neurological sequelae), difficulties in establishing oral feeding (slow feeding, choking episodes, desaturation events, bradycardia, and apnea), jaundice, and short and long-term respiratory compromise (respiratory distress syndrome, transient tachypnoea and pneumonia, increased risk of bronchiolitis and wheezing) [142 - 144].
Long-term health and neurodevelopmental problems, as well as educational difficulties are known to occur 25 . Although very and extreme preterm infants were associated with the highest expenditures, MPT healthcare needs impact significantly upon pediatric healthcare, due to their larger population size [138 ,143 ].
Prematurity: Prevention
PH can be decisive in reducing modifiable risk factors of prematurity, improving perinatal care, and supporting postnatal infant development. Whilst the risk of adverse outcome is highest among very preterm [18 ,96 ], MPT represent a large population for whom PH policy can be most effective [103 ,143 - 145 ].
The prevention of prematurity requires a multifaceted approach aiming to globally improve women’s health, starting before pregnancy but also focusing on preconception and gestational periods [44 ,146 ]. Intervention on social determinants of health can reduce some risk factors, influencing PB rates and outcomes 147 . PH intervention can be crucial in controlling adverse social conditions, namely poverty, discrimination, violence, low education, and intervention for behavioral factors (alcohol, tobacco, or illicit drug use); regulations to protect women from workload or exposure to potential harmful pollutants, improving adequate spacing (> 24 months) and preventing teenage or unintended pregnancies [44 ,147].
In the preconception period, equality in access to primary, preventive, and obstetric risk care is highlighted. Regimentation of assisted reproductive therapies (ART), aimed to reduce the risk of higher-order multiples, focused on reducing the number of embryos for transfer (elective single embryo transfer policy) and eventually limiting candidate’s age to ART, are relevant issues [148 ,149 ]. During gestation, the relevance of early identification of women at risk for preterm delivery has been stressed as it leads to effective preventive treatments, control of chronic diseases, optimization of body mass index, eradication of non-medically indicated deliveries before 39 weeks, and effective management of preterm labor [5 ,44 ].
In assessing adequate preterm delivery, occurring in a high-level quality care, neonatal resuscitation, comprehensive neonatal intensive care, special infant feeding support, and management of neonatal complications are important issues.
It is essential to put in place adequate policies to integrate premature babies within interventional programs to track long-term disabling outcomes and to support postnatal infant development [150 ].
The need to research prematurity causes and their interrelationship, to search for newer therapeutics and preventive approaches of PB is key. PH can provide population-based surveillance systems to monitor trends in prematurity, prevention efforts, risk factor evolution, and outcomes in PB [151 - 153].
Conclusion
The increasing survival at progressively lower GA with a higher risk of morbidity, draws attention to the high number of people surviving from prematurity in terms of PH [28 ,41 ,154]. Dissemination of knowledge about the causes and prevention of PB, the extent of morbidity and mortality associated to prematurity, and their impact on families and support structures must be relevant to policy makers. Addressing social determinants of health can reduce PB rates and improve outcomes. This knowledge and consequent multifaceted action may contribute to gains in terms of health economics and optimization of the human potential of individuals born preterm and their families. The burden of PB highlights the crucial importance of prematurity prevention – it must be a priority issue in health policy makers’ agenda.
REFERENCES
1 Federation of Gynecology and Obstetrics (FIGO). WHO: recommended definitions, terminology and format for statistical tables related to the perinatal period and use of a new certificate for cause of perinatal deaths: modifications recommended by FIGO as amended October 14, 1976. Acta Obstet Gynecol Scand. 1977; 56(3): 247–53.
2 Liu L, Johnson HL, Cousens S, Perin J, Scott S, Lawn JE, et al.; Child Health Epidemiology Reference Group of WHO and UNICEF. Global, regional, and national causes of child mortality: an updated systematic analysis for 2010 with time trends since 2000. Lancet. 2012 Jun; 379(9832): 2151–61.
3 Laws P, Grayson N, Sullivan E. Australia’s mothers and babies 2004. Volume 18. Sydney: Perinatal Statistics Series; 2006.
4 Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller AB, Narwal R, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012 Jun; 379(9832): 2162–72.
5 March of Dimes. The Partnership for Maternal Newborn & Child Health, Save the Children, World Health Organization. Born too soon: the global action report on preterm birth. Geneva: World Health Organization; 2012. [ Links ]
6 Steer P. The epidemiology of preterm labour. BJOG. 2005 Mar; 112 Suppl 1: 1–3.
7 Ministério da Saúde, Portugal. Direção-Geral da Saude. Natalidade, mortalidade infantil, fetal e perinatal, 2008/2012. Lisboa: Direção- Geral da Saúde; 2012.
8 OECD Publishing. Health at a glance 2013: OECD indicators. Paris: OECD; 2013. [ Links ]
9 Wilson-Costello D, Friedman H, Minich N, Fanaroff AA, Hack M. Improved survival rates with increased neurodevelopmental disability for extremely low birth weight infants in the 1990s. Pediatrics. 2005 Apr; 115(4): 997–1003.
10 Project EN. EuroNeoNet annual report for VLGAI & individual report for each unit participating in the EuroNeoNet project. Bizkaia, Spain: ENN Project; 2015.
11 Allen MC, Cristofalo EA, Kim C. Outcomes of preterm infants: morbidity replaces mortality. Clin Perinatol. 2011 Sep; 38(3): 441–54.
12 Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010 Sep; 126(3): 443–56.
13 Costeloe K, Hennessy E, Gibson AT, Marlow N, Wilkinson AR; for the EPICure Study Group. The EPICure study: outcomes to discharge from hospital for infants born at the threshold of viability. Pediatrics. 2000 Oct; 106(4): 659–71.
14 Bonamy A, Zeitlin J, Piedvache A, Maier R, van Heijst A, Varendi H, et al. Wide variation in severe neonatal morbidity among very preterm infants in European regions. Arch Dis Child Fetal Neonatal Ed. 2019 Jan; 104(1):F36-F45. [ Links ]
15 Soleimani F, Zaheri F, Abdi F. Long-term neurodevelopmental outcomes after preterm birth. Iran Red Crescent Med J. 2014 Jun; 16(6):e17965. [ Links ]
16 Hack M, Cartar L, Schluchter M, Klein N, Forrest CB. Self-perceived health, functioning and well-being of very low birth weight infants at age 20 years. J Pediatr. 2007 Dec; 151(6): 635–41.
17 Pierrat V, Marchand-Martin L, Arnaud C, Kaminski M, Resche-Rigon M, Lebeaux C, et al.; EPIPAGE-2 writing group. Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ. 2017 Aug; 358:j3448.
18 Glass HC, Costarino AT, Stayer SA, Brett CM, Cladis F, Davis PJ. Outcomes for extremely premature infants. Anesth Analg. 2015 Jun; 120(6): 1337–51.
19 Raju TN, Pemberton VL, Saigal S, Blaisdell CJ, Moxey-Mims M, Buist S; Adults Born Preterm Conference Speakers and Discussants. Long-term healthcare outcomes of preterm birth: an executive summary of a conference sponsored by the National Institutes of Health. J Pediatr. 2017 Feb; 181: 309– 318.e1.
20 OECD. Health at a glance 2017: OECD indicators. Paris: OECD; 2017. [ Links ]
21 Veen S, Ens-Dokkum MH, Schreuder AM, Verloove-Vanhorick SP, Brand R, Ruys JH; The Collaborative Project on Preterm and Small for Gestational Age Infants (POPS) in The Netherlands. Impairments, disabilities, and handicaps of very preterm and very-lowbirthweight infants at five years of age. Lancet. 1991 Jul; 338(8758): 33–6.
22 Larroque B, Ancel PY, Marret S, Marchand L, André M, Arnaud C, et al.; EPIPAGE Study group. Neurodevelopmental disabilities and special care of 5-year-old children born before 33 weeks of gestation (the EPIPAGE study): a longitudinal cohort study. Lancet. 2008 Mar; 371(9615): 813–20.
23 MJ, Ruys JH. Neonatal mortality risk in relation to gestational age and birthweight. Results of a national survey of preterm and very-low-birthweight infants in the Netherlands. Lancet. 1986 Jan; 1(8472): 55–7.
24 Platt MJ. Outcomes in preterm infants. Public Health. 2014 May; 128(5): 399–403.
25 Crump C, Sundquist K, Sundquist J, Winkleby MA. Gestational age at birth and mortality in young adulthood. JAMA. 2011 Sep; 306(11): 1233–40.
26 Saigal S, Doyle LW. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet. 2008 Jan; 371(9608): 261–9.
27 Farooqi A, Hägglöf B, Sedin G, Serenius F. Impact at age 11 years of major neonatal morbidities in children born extremely preterm. Pediatrics. 2011 May; 127(5):e1247–57.
28 Hodek JM, von der Schulenburg JM, Mittendorf T. Measuring economic consequences of preterm birth - Methodological recommendations for the evaluation of personal burden on children and their caregivers. Health Econ Rev. 2011 Jul; 1(1): 6. [ Links ]
29 Liu L, Oza S, Hogan D, Perin J, Rudan I, Lawn JE, et al. Global, regional, and national causes of child mortality in 2000-13, with projections to inform post-2015 priorities: an updated systematic analysis. Lancet. 2015 Jan; 385(9966): 430–40.
30 Mwaniki MK, Atieno M, Lawn JE, Newton CR. Long-term neurodevelopmental outcomes after intrauterine and neonatal insults: a systematic review. Lancet. 2012 Feb; 379(9814): 445–52.
31 Ho LY. Follow-up care and outcome evaluation of high-risk preterm infants: a life-course commitment. Ann Acad Med Singapore. 2018 Feb; 47(2): 51–5.
32 Barfield WD. Public health implications of very preterm birth. Clin Perinatol. 2018 Sep; 45(3): 565–77.
33 Vogel JP, Chawanpaiboon S, Watananirun K, Lumbiganon P, Petzold M, Moller AB, et al. Global, regional and national levels and trends of preterm birth rates for 1990 to 2014: protocol for development of World Health Organization estimates. Reprod Health. 2016 Jun; 13(1): 76. [ Links ]
34 Blencowe H, Cousens S, Chou D, Oestergaard M, Say L, Moller AB, et al.; Born Too Soon Preterm Birth Action Group. Born too soon: the global epidemiology of 15 million preterm births. Reprod Health. 2013; 10(1 Suppl 1):S2. [ Links ]
35 Nogueira PJ, Costa AJ, Alves MI, Rosa MV. Estudo comparativo do número de óbitos e causas de morte da mortalidade infantil e suas componentes (2009-2011). Lisboa: Direção de Serviços de Informação e Análise. Direção Geral da Saúde; 2013. [ Links ]
36 Crump C. Medical history taking in adults should include questions about preterm birth. BMJ. 2014 Aug; 349 aug01 2:g4860. [ Links ]
37 Chawanpaiboon S, Vogel JP, Moller AB, Lumbiganon P, Petzold M, Hogan D, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob Health. 2019 Jan; 7(1):e37–46.
38 Euro-Peristat Project. European Perinatal Health Report. Core indicators of the health and care of pregnant women and babies in Europe in 2015. November 2018. Available at www.europeristat.com.
39 Beck S, Wojdyla D, Say L, Betran AP, Merialdi M, Requejo JH, et al. The worldwide incidence of preterm birth: a systematic review of maternal mortality and morbidity. Bull World Health Organ. 2010 Jan; 88(1): 31–8.
40 Zeitlin J, Szamotulska K, Drewniak N, Mohangoo AD, Chalmers J, Sakkeus L, et al.; Euro-Peristat Preterm Study Group. Preterm birth time trends in Europe: a study of 19 countries. BJOG. 2013 Oct; 120(11): 1356–65.
41 Zeitlin J, Mohangoo A, Delnord M. European Perinatal Health Report. Health and care of pregnant women and babies in Europe in 2010. Available at www.europeristat.com. [ Links ]
42 Goldenberg RL, Gravett MG, Iams J, Papageorghiou AT, Waller SA, Kramer M, et al. The preterm birth syndrome: issues to consider in creating a classification system. Am J Obstet Gynecol. 2012 Feb; 206(2): 113–8.
43 INE. Estatísticas demográficas 2016. Lisboa: Instituto Nacional de Estatística; 2017. [ Links ]
44 Behrman RE, Butler AS; Committee on Understanding Premature Birth and Assuring Healthy Outcomes. Preterm birth: causes, consequences, and prevention. Washington (DC): National Academies Press; 2007. [ Links ]
45 Main DM, Gabbe SG, Richardson D, Strong S. Can preterm deliveries be prevented? Am J Obstet Gynecol. 1985 Apr; 151(7): 892–8.
46 Martin JA, Osterman MJ. Describing the increase in preterm births in the United States, 2014-2016. NCHS Data Brief. 2018 Jun; 312(312): 1–8.
47 McDonald SD, Han Z, Mulla S, Beyene J; Knowledge Synthesis Group. Overweight and obesity in mothers and risk of preterm birth and low birth weight infants: systematic review and meta-analyses. BMJ. 2010 Jul; 341 jul20 1:c3428–3428.
48 Lawn J. 4 million neonatal deaths: an analysis of available cause-of-death data and systematic country estimates with a focus on “birth asphyxia” (PhD thesis). London: UCL; 2009. 202 p.
49 Manuck TA, Rice MM, Bailit JL, Grobman WA, Reddy UM, Wapner RJ, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal- Fetal Medicine Units Network. Preterm neonatal morbidity and mortality by gestational age: a contemporary cohort. Am J Obstet Gynecol. 2016 Jul; 215(1): 103.e1–14.
50 Seri I, Evans J. Limits of viability: definition of the gray zone. J Perinatol. 2008 May; 28(S1 Suppl 1):S4–8.
51 Fanaroff AA, Stoll BJ, Wright LL, Carlo WA, Ehrenkranz RA, Stark AR, et al.; NICHD Neonatal Research Network. Trends in neonatal morbidity and mortality for very low birthweight infants. Am J Obstet Gynecol. 2007 Feb; 196(2): 147.e1–8.
52 Rüegger C, Hegglin M, Adams M, Bucher HU; Swiss Neonatal Network. Population based trends in mortality, morbidity and treatment for very preterm- and very low birth weight infants over 12 years. BMC Pediatr. 2012 Feb; 12(1): 17. [ Links ]
53 Helenius K, Sjörs G, Shah PS, Modi N, Reichman B, Morisaki N, et al.; International Network for Evaluating Outcomes (iNeo) of Neonates. Survival in very preterm infants: an international comparison of 10 national neonatal networks. Pediatrics. 2017 Dec; 140(6): e20171264. [ Links ]
54 Cheong J, Lee K, Boland R, Spittle A, Opie G, Burnett A, et al. Changes in long-term prognosis with increasing postnatal survival and the occurrence of postnatal morbidities in extremely preterm infants offered intensive care: a prospective observational study. Lancet Child Adolesc Health. 2018 Dec; 2(12): 872-879. [ Links ]
55 Sociedade Portuguesa de Neonatologia. Registo nacional do MBP (Internet). Lisboa: Sociedade Portuguesa de Neonatologia; 2019 (cited 2017 Oct 22). Available from: http://www.spneonatologia.pt/documents/presentations/registo-nacional-do-mbp/
56 Blencowe H, Lee AC, Cousens S, Bahalim A, Narwal R, Zhong N, et al. Preterm birth-associated neurodevelopmental impairment estimates at regional and global levels for 2010. Pediatr Res. 2013 Dec; 74(S1 Suppl 1): 17–34.
57 Robaei D, Kifley A, Gole GA, Mitchell P. The impact of modest prematurity on visual function at age 6 years: findings from a population- based study. Arch Ophthalmol. 2006 Jun; 124(6): 871–7.
58 Younge N, Goldstein RF, Bann CM, Hintz SR, Patel RM, Smith PB, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Survival and neurodevelopmental outcomes among periviable infants. N Engl J Med. 2017 Feb; 376(7): 617–28.
59 Hadders-Algra M. Two distinct forms of minor neurological dysfunction: perspectives emerging from a review of data of the Groningen Perinatal Project. Dev Med Child Neurol. 2002 Aug; 44(8): 561–71.
60 Volpe JJ. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 2009 Jan; 8(1): 110–24.
61 Mukerji A, Shah V, Shah PS. Periventricular/ intraventricular hemorrhage and neurodevelopmental outcomes: a meta-analysis. Pediatrics. 2015 Dec; 136(6): 1132–43.
62 Walsh B, Inder T, Volpe J. Pathophysiology of intraventricular hemorrhage in the neonate. In: Polin R, editor. Fetal and Neonatal Physiology. 5th ed. New York: Elsevier; 2017. p. 1333–1349.e6.
63 Inder TE, Volpe JJ. Intraventricular hemorrhage in the neonate. In: Polin R, Fox W, Abman S, editors. Fetal and Neonatal Physiology. 3rd ed. Philadelphia: Saunders; 2011. pp. 1830–47.
64 Volpe JJ. Intracranial hemorrhage: germinal matrix – intraventricular hemorrhage of the premature infant. In: Volpe JJ, editor. Neurology of the newborn. 4th ed. Philadelphia: Saunders; 2001. pp. 517–88.
65 Murphy BP, Inder TE, Rooks V, Taylor GA, Anderson NJ, Mogridge N, et al. Posthaemorrhagic ventricular dilatation in the premature infant: natural history and predictors of outcome. Arch Dis Child Fetal Neonatal Ed. 2002 Jul; 87(1):F37–41.
66 Wilson-Costello D, Payne A. Early childhood neurodevelopmental outcomes of high-risk neonates. 10th ed. Philadelphia, Saunders; 2015. p. 1018-1031. [ Links ]
67 Joo JW, Choi JY, Rha DW, Kwak EH, Park ES. Neuropsychological outcomes of preterm birth in children with no major neurodevelopmental impairments in early life. Ann Rehabil Med. 2015 Oct; 39(5): 676–85.
68 Edwards J, Berube M, Erlandson K, Haug S, Johnstone H, Meagher M, et al. Developmental coordination disorder in school-aged children born very preterm and/or at very low birth weight: a systematic review. J Dev Behav Pediatr. 2011; 32: 678-687. [ Links ]
69 Danks M, Maideen MF, Burns YR, O’Callaghan MJ, Gray PH, Poulsen L, et al. The long-term predictive validity of early motor development in “apparently normal” ELBW survivors. Early Hum Dev. 2012 Aug; 88(8): 637–41.
70 Himmelmann K, Hagberg G, Beckung E, Hagberg B, Uvebrant P. The changing panorama of cerebral palsy in Sweden. IX. Prevalence and origin in the birth-year period 1995-1998. Acta Paediatr. 2005 Mar; 94(3): 287–94.
71 Platt M, Cans C, Johnson A, Surman G, et al. Trends in cerebral palsy among infants of very low birthweight (< 1500 g) or born prematurely (< 32 weeks) in 16 European centres: a database study. Lancet. 2007; 369: 43–50.
72 Hagberg B, Hagberg G, Beckung E, Uvebrant P. Changing panorama of cerebral palsy in Sweden. VIII. Prevalence and origin in the birth year period 1991-94. Acta Paediatr. 2001 Mar; 90(3): 271–7.
73 Himmelmann K, Uvebrant P. The panorama of cerebral palsy in Sweden part XII shows that patterns changed in the birth years 2007-2010. Acta Paediatr. 2018 Mar; 107(3): 462–8.
74 Winter S, Autry A, Boyle C, Yeargin-Allsopp M. Trends in the prevalence of cerebral palsy in a population-based study. Pediatrics. 2002 Dec; 110(6): 1220–5.
75 Wilson-Costello D, Friedman H, Minich N, Siner B, Taylor G, Schluchter M, et al. Improved neurodevelopmental outcomes for extremely low birth weight infants in 2000- 2002. Pediatrics. 2007 Jan; 119(1): 37–45.
76 Virella D, Folha T, Andrada M, Cadete A, Gouveia R, Gaia T, et al. Vigilância nacional da paralisia cerebral aos 5 anos de idade: crianças nascidas entre 2001 e 2007. 2a. Lisboa: Federação das Associações Portuguesas de Paralisia Cerebral; 2018. [ Links ]
77 Farooqi A, Hägglöf B, Sedin G, Gothefors L, Serenius F. Chronic conditions, functional limitations, and special health care needs in 10- to 12-year-old children born at 23 to 25 weeks gestation in the 1990s: a Swedish national prospective follow-up study. Pediatrics. 2006 Nov; 118(5):e1466-e1477. [ Links ]
78 Rudanko SL, Fellman V, Laatikainen L. Visual impairment in children born prematurely from 1972 through 1989. Ophthalmology. 2003 Aug; 110(8): 1639–45.
79 Chhablani PP, Kekunnaya R. Neuro-ophthalmic manifestations of prematurity. Indian J Ophthalmol. 2014 Oct; 62(10): 992–5.
80 Webber AL, Wood J. Amblyopia: prevalence, natural history, functional effects and treatment. Clin Exp Optom. 2005 Nov; 88(6): 365– 75.
81 Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013 Dec; 74(S1 Suppl 1): 35–49.
82 Yu B, Guo Q, Fan G, Liu N. Assessment of cortical visual impairment in infants with periventricular leukomalacia: a pilot eventrelated FMRI study. Korean J Radiol. 2011 Jul-Aug; 12(4): 463–72.
83 Hellström A, Smith LE, Dammann O. Retinopathy of prematurity. Lancet. 2013 Oct; 382(9902): 1445–57.
84 O’Connor AR, Wilson CM, Fielder AR. Ophthalmological problems associated with preterm birth. Eye (Lond). 2007 Oct; 21(10): 1254–60.
85 Saigal S, Stoskopf B, Boyle M, Paneth N, Pinelli J, Streiner D, et al. Comparison of current health, functional limitations, and health care use of young adults who were born with extremely low birth weight and normal birth weight. Pediatrics. 2007 Mar; 119(3):e562–73. 8
86 O’Connor AR, Stephenson T, Johnson A, Tobin MJ, Moseley MJ, Ratib S, et al. Long-term ophthalmic outcome of low birth weight children with and without retinopathy of prematurity. Pediatrics. 2002 Jan; 109(1): 12–8.
87 Marlow N, Wolke D, Bracewell MA, Samara M; EPICure Study Group. Neurologic and developmental disability at six years of age after extremely preterm birth. N Engl J Med. 2005 Jan; 352(1): 9–19.
88 Saigal SL, Stoskopf BL, Streiner D, Burrows E. Physical growth and current health status of infants who were of extremely low birth weight and controls at adolescence. Pediatrics. 2001 Aug; 108(2): 407-15. [ Links ]
89 Rushing S, Ment LR. Preterm birth: a cost benefit analysis. Semin Perinatol. 2004 Dec; 28(6): 444–50.
90 Olusanya BO, Neumann KJ, Saunders JE. The global burden of disabling hearing impairment: a call to action. Bull World Health Organ. 2014 May; 92(5): 367–73.
91 Therien JM, Worwa CT, Mattia FR, deRegnier RA. Altered pathways for auditory discrimination and recognition memory in preterm infants. Dev Med Child Neurol. 2004 Dec; 46(12): 816–24.
92 Cristobal R, Oghalai JS. Hearing loss in children with very low birth weight: current review of epidemiology and pathophysiology. Arch Dis Child Fetal Neonatal Ed. 2008 Nov; 93(6):F462–8.
93 Engdahl B, Eskild A. Birthweight and the risk of childhood sensorineural hearing loss. Paediatr Perinat Epidemiol. 2007 Nov; 21(6): 495–500.
94 Doyle LW, Anderson PJ; Victorian Infant Collaborative Study Group. Improved neurosensory outcome at 8 years of age of extremely low birthweight children born in Victoria over three distinct eras. Arch Dis Child Fetal Neonatal Ed. 2005 Nov; 90(6):F484–8.
95 Voss W, Jungmann T, Wachtendorf M, Neubauer AP. Long-term cognitive outcomes of extremely low-birth-weight infants: the influence of the maternal educational background. Acta Paediatr. 2012 Jun; 101(6): 569-73. [ Links ]
96 Aylward GP. Neurodevelopmental outcomes of infants born prematurely. J Dev Behav Pediatr. 2005 Dec; 26(6): 427–40.
97 Putnick DL, Bornstein MH, Eryigit-Madzwamuse S, Wolke D. Long-term stability of language performance in very preterm, moderate-late preterm, and term children. J Pediatr. 2017 Feb; 181: 74–79.e3.
98 Barre N, Morgan A, Doyle LW, Anderson PJ. Language abilities in children who were very preterm and/or very low birth weight: a meta-analysis. J Pediatr. 2011 May; 158(5): 766–774.e1.
99 Vanderbilt D, Gleason MM. Mental health concerns of the premature infant through the lifespan. Child Adolesc Psychiatr Clin N Am. 2010 Apr; 19(2): 211–28.
100 Vinall J, Grunau RE. Impact of repeated procedural pain-related stress in infants born very preterm. Pediatr Res. 2014; 75(5): 584– 7.
101 Haller S, Deindl P, Cassini A, Suetens C, Zingg W, Abu Sin M, et al. Neurological sequelae of healthcare-associated sepsis in very-low-birthweight infants: umbrella review and evidence-based outcome tree.Euro Surveill. 2016; 21(8): 30143. [ Links ]
102 Bell MJ, Ternberg JL, Feigin RD, Keating JP, Marshall R, Barton L, et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg. 1978 Jan; 187(1): 1–7.
103 Guy A, Seaton SE, Boyle EM, Draper ES, Field DJ, Manktelow BN, et al. Infants born late/moderately preterm are at increased risk for a positive autism screen at 2 years of age. J Pediatr. 2015 Feb; 166(2): 269–75.e3.
104 Saigal S, Day KL, Van Lieshout RJ, Schmidt LA, Morrison KM, Boyle MH. Health, wealth, social integration, and sexuality of extremely low-birth-weight prematurely born adults in the fourth decade of life. JAMA Pediatr. 2016 Jul; 170(7): 678–86.
105 Mathewson KJ, Chow CH, Dobson KG, Pope EI, Schmidt LA, Van Lieshout RJ. Mental health of extremely low birth weight survivors: A systematic review and meta-analysis. Psychol Bull. 2017 Apr; 143(4): 347–83.
106 Davidson LM, Berkelhamer SK. Bronchopulmonary dysplasia: chronic lung disease of infancy and long-term pulmonary outcomes. J Clin Med. 2017 Jan; 6(1): 4. [ Links ]
107 Baker CD, Abman SH, Mourani PM. Pulmonary hypertension in preterm infants with bronchopulmonary dysplasia. Pediatr Allergy Immunol Pulmonol. 2014 Mar; 27(1): 8–16.
108 Shannon J, Wikenheiser-Brokamp K, Greenberg J. Lung growth and development. In: Broaddus C, Mason R, Ernst J, King T, Lazarus S, Murray J, et al., editors. Textbook of Respiratory Medicine. 6th ed. Philadelphia: Saunders; 2016. p. 22–31.e4.
109 Baraldi E, Filippone M. Chronic lung disease after premature birth. N Engl J Med. 2007 Nov; 357(19): 1946–55.
110 Zysman-Colman Z, Tremblay GM, Bandeali S, Landry JS. Bronchopulmonary dysplasia - trends over three decades. Paediatr Child Health. 2013 Feb; 18(2): 86–90.
111 O’Connor M, Cornfield D, Austin E. Pulmonary hypertension in the premature infant: a challenging comorbidity in a vulnerable population. Curr Opin Pediatr. 2016; 28(3): 324–30.
112 Bolton CE, Bush A, Hurst JR, Kotecha S, Mc- Garvey L. Lung consequences in adults born prematurely. Thorax. 2015 Jun; 70(6): 574– 80.
113 Amin SC, Pappas C, Iyengar H, Maheshwari A. Short bowel syndrome in the NICU. Clin Perinatol. 2013 Mar; 40(1): 53–68.
114 Wales PW, Christison-Lagay ER. Short bowel syndrome: epidemiology and etiology.Semin Pediatr Surg. 2010 Feb; 19(1): 3–9.
115 Stey A, Barnert ES, Tseng CH, Keeler E, Needleman J, Leng M, et al. Outcomes and costs of surgical treatments of necrotizing enterocolitis. Pediatrics. 2015 May; 135(5): e1190–7.
116 Rees CM, Pierro A, Eaton S. Neurodevelopmental outcomes of neonates with medically and surgically treated necrotizing enterocolitis. Arch Dis Child Fetal Neonatal Ed. 2007 May; 92(3):F193–8.
117 Soltys KA, Bond G, Sindhi R, Rassmussen SK, Ganoza A, Khanna A, et al. Pediatric intestinal transplantation. Semin Pediatr Surg. 2017 Aug; 26(4): 241–9.
118 Charlton JR, Guillet R. Neonatal acute kidney injury: diagnosis, exposures, and longterm outcomes. Neoreviews. 2018; 19(6): e322–e 336.
119 Chevalier RL. Developmental renal physiology of the low birth weight pre-term newborn. J Urol. 1996 Aug; 156(2 Pt 2): 714–9.
120 Iyengar A, Nesargi S, George A, Sinha N, Selvam S, Luyckx VA. Are low birth weight neonates at risk for suboptimal renal growth and function during infancy? BMC Nephrol. 2016 Jul; 17(1): 100. [ Links ]
121 Greenberg JH, Coca S, Parikh CR. Longterm risk of chronic kidney disease and mortality in children after acute kidney injury: a systematic review. BMC Nephrol. 2014 Nov; 15(1): 184. [ Links ]
122 Mammen C, Al Abbas A, Skippen P, Nadel H, Levine D, Collet JP, et al. Long-term risk of CKD in children surviving episodes of acute kidney injury in the intensive care unit: a prospective cohort study. Am J Kidney Dis. 2012 Apr; 59(4): 523–30.
123 Kelli H, Kassas I, Lattouf O. Cardio metabolic syndrome: a global epidemic. J Diabetes Metab. 2015; 6(3): 2–14.
124 Barker DJ. Early growth and cardiovascular disease. Arch Dis Child. 1999 Apr; 80(4): 305–7.
125 Barker DJ, Bull AR, Osmond C, Simmonds SJ. Fetal and placental size and risk of hypertension in adult life. BMJ. 1990 Aug; 301(6746): 259–62.
126 Parkinson J, Hyde M, Gale C, Santhakumaran S, Modi N. Preterm birth and the metabolic syndrome in adult life: a systematic review and meta-analysis. Pediatrics. 2013; 131(4):e1240-e1263. [ Links ]
127 Gluckman PD, Hanson MA, Cooper C, Thornburg KL. Effect of in utero and earlylife conditions on adult health and disease. N Engl J Med. 2008 Jul; 359(1): 61–73.
128 Kajantie E, Hovi P. Is very preterm birth a risk factor for adult cardiometabolic disease? Semin Fetal Neonatal Med. 2014 Apr; 19(2): 112–7.
129 Rosendaal FR. Venous thrombosis: a multicausal disease. Lancet. 1999 Apr; 353(9159): 1167–73.
130 Zöller B, Li X, Sundquist J, Sundquist K, Crump C. Gestational age and risk of venous thromboembolism from birth through young adulthood. Pediatrics. 2014 Aug; 134(2):e473– 80.
131 Setty BA, O’Brien SH, Kerlin BA. Pediatric venous thromboembolism in the United States: a tertiary care complication of chronic diseases. Pediatr Blood Cancer. 2012 Aug; 59(2): 258–64.
132 Hornik CP, Fort P, Clark RH, Watt K, Benjamin DK Jr, Smith PB, et al. Early and late onset sepsis in very-low-birth-weight infants from a large group of neonatal intensive care units. Early Hum Dev. 2012 May; 88 Suppl 2:S69–74.
133 Sonnenschein-van der Voort AM, ArendsLR, de Jongste JC, Annesi-Maesano I, Arshad SH, Barros H, et al. Preterm birth, infant weight gain, and childhood asthma risk: a meta-analysis of 147,000 European children. J Allergy Clin Immunol. 2014 May; 133(5): 1317–29.
134 Barbarot S, Gras-Leguen C, Colas H, Garrot E, Darmaun D, Larroque B, et al. Lower risk of atopic dermatitis among infants born extremely preterm compared with higher gestational age. Br J Dermatol. 2013 Dec; 169(6): 1257–64.
135 Goedicke-Fritz S, Härtel C, Krasteva-Christ G, Kopp MV, Meyer S, Zemlin M. Preterm birth affects the risk of developing immunemediated diseases. Front Immunol. 2017 Oct; 8: 1266. [ Links ]
136 Groer MW, Gregory KE, Louis-Jacques A, Thibeau S, Walker WA. The very low birth weight infant microbiome and childhood health. Birth Defects Res C Embryo Today. 2015 Dec; 105(4): 252–64.
137 Groer MW, Luciano AA, Dishaw LJ, Ashmeade TL, Miller E, Gilbert JA. Development of the preterm infant gut microbiome: a research priority. Microbiome. 2014 Oct; 2(1): 38. [ Links ]
138 Johnston KM, Gooch K, Korol E, Vo P, Eyawo O, Bradt P, et al. The economic burden of prematurity in Canada. BMC Pediatr. 2014 Apr; 14(1): 93. [ Links ]
139 Petrou S, Sach T, Davidson L. The long-term costs of preterm birth and low birth weight: results of a systematic review. Child Care Health Dev. 2001 Mar; 27(2): 97–115.
140 van Exel J, Bobinac A, Koopmanschap M, Brouwer W. The invisible hands made visible: recognizing the value of informal care in healthcare decision-making. Expert Rev Pharmacoecon Outcomes Res. 2008 Dec; 8(6): 557–61.
141 Hoefman RJ, van Exel J, Brouwer W. How to include informal care in economic evaluations. Pharmacoeconomics. 2013 Dec; 31(12): 1105–19.
142 Morgan J, Boyle E. The late preterm infant. Paediatr Child Health (Oxford). 2018; 28(1): 13–7.
143 Vohr B. Long-term outcomes of moderately preterm, late preterm, and early term infants. Clin Perinatol. 2013 Dec; 40(4): 739– 51.
144 Laptook AR. Neurologic and metabolic issues in moderately preterm, late preterm, and early term infants. Clin Perinatol. 2013 Dec; 40(4): 723–38.
145 Kramer MS, Demissie K, Yang H, Platt RW, Sauvé R, Liston R; Fetal and Infant Health Study Group of the Canadian Perinatal Surveillance System. The contribution of mild and moderate preterm birth to infant mortality. JAMA. 2000 Aug; 284(7): 843–9.
146 WHO. WHO recommendations on antenatal care for a positive pregnancy experience. WHO: Geneva; 2016. [ Links ]
147 Lorch SA, Enlow E. The role of social determinants in explaining racial/ethnic disparities in perinatal outcomes. Pediatr Res. 2016 Jan; 79(1-2): 141–7.
148 Sharma R, Biedenharn KR, Fedor JM, Agarwal A. Lifestyle factors and reproductive health: taking control of your fertility. Reprod Biol Endocrinol. 2013 Jul; 11(1): 66. [ Links ]
149 Conselho Nacional de Procriação Medicamente Assistida. Parecer relativo ao projeto de decreto-lei que procede a regulamentação da Lei n.o 17/2016, de 20 de junho (Internet). Lisboa: Conselho Nacional de Procriação Medicamente Assistida; 2017 (cited 2018 Dec 10). Available from: http://www.cnpma.org.pt/
150 Carter FA, Msall ME. Long-term functioning and participation across the life course for preterm neonatal intensive care unit graduates. Clin Perinatol. 2018 Sep; 45(3): 501–27.
151 Shapiro-Mendoza CK, Barfield WD, Henderson Z, James A, Howse JL, Iskander J, et al. CDC Grand rounds: public health strategies to prevent preterm birth. MMWR Morb Mortal Wkly Rep. 2016 Aug; 65(32): 826–30.
152 Shulman H, Angelo D, Harrison L, Smith R, Warner L. The pregnancy risk assessment monitoring system (PRAMS): overview of design and methodology. Am J Public Health. 2018 Oct; 108(10): 1305-1313. [ Links ]
153 Gould JB. The role of regional collaboratives: the California Perinatal Quality Care Collaborative model. Clin Perinatol. 2010 Mar; 37(1): 71–86.
154 Correia S, Rodrigues T, Montenegro N, Barros H. Critical evaluation of national vital statistics: the case of preterm birth trends in Portugal. Acta Obstet Gynecol Scand. 2015 Nov; 94(11): 1215–22.
The authors have no conflicts of interest to declare.














