INTRODUCTION
Kidney supportive care (KSC) integrates palliative care principles within nephrological practice, encompassing the full spectrum of kidney disease, from conservative care (management without dialysis) to dialysis withdrawal and end-of-life care.1-3Successful dialysis withdrawal is a complex and emotionally challenging therapeutic option for nephrologists, patients, and their families.1 This requires a patient‑centered approach that respects individual beliefs and values, establishes realistic expectations, and minimizes suffering to facilitate a dignified and personalized end‑of‑life experience. 1-3Often, as patients experience increased physical dependence on treatment, healthcare professionals may delay withdrawal discussions, and families may struggle to accept mortality, thereby increasing the complexity of the situation.4 This case report describes a patient with end‑stage chronic kidney disease (CKD) undergoing hemodialysis, who was misdiagnosed with peritoneal carcinomatosis. This report concludes with a discussion of the case and a proposed framework concerning KSC, focusing on dialysis withdrawal.
CASE REPORT
We present a 56‑year‑old man with a relevant past medical history of diabetes with both microvascular‑ and macrovascular end‑organ damage (retinopathy, end‑stage CKD undergoing hemodialysis, and peripheral arterial dis- ease), hypertension, alcoholic liver disease with clinically significant portal hypertension, and dilated cardiomyopathy, which all translated an age‑adjusted Charlson Comorbidity Index of 11 points. Upon presentation to the emergency department, the patient reported a three‑week history of diffuse abdominal pain, accompanied by asthenia, anorexia, and an unintentional weight loss of 1.5 kg. There was no known history of immunosuppression or apparent epidemiological context. Physical examination revealed an emaciated and malnourished male (body mass index [BMI] 15.4 kg/m²) with notable ascites, peripheral edema, and abdominal guarding. Initial laboratory analysis demonstrated expectedly low nutritional parameters (serum albumin 18 g/L, potassium 4.0 mmol/L, pre‑dialysis urea 80 mg/dL) and elevated inflammatory markers (C‑reactive protein [CRP] 172.5 mg/L) without leukocytosis. A thoracoabdominal computed tomography (CT) scan reported bulky and loculated ascites, peritoneum thickening, and diffuse micronodularity of the great omentum and mesentery, favoring peritoneal carcinomatosis (Fig. 1). The patient was admitted for etiologic investigation and continuation of care.
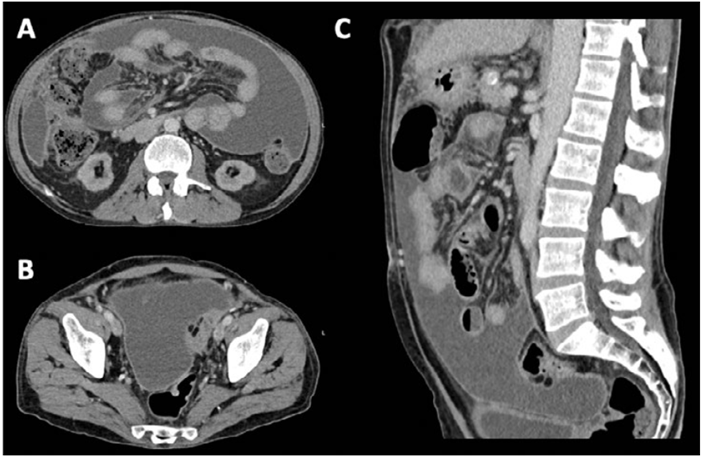
Figure1. CT‑scan transversal (A and B) and sagittal planes (C). Bulky ascites, peritoneum thickening, and diffuse micronodularity of the great omentum and across the mesentery, favoring peritoneal carcinomatosis.
his hospitalization, the patient underwent three diagnostic paracenteses. All samples demonstrated in- creased cellularity (up to 454 cells) with a predominantly mononuclear population (ranging from 73% to 95%). The total protein in the ascitic fluid was approximately 30.0 g/L, and the ascitic albumin was low at approximately 11.5 g/L, resulting in a serum‑ascites albumin gradient (SAAG) of 0.7 g/L. Adenosine deaminase levels were elevated (84 U/L). Gram stains, bacterial and fungal cultures were negative in all three ascitic fluid draws, as well as the Nucleic Acid Amplification Testing (NAAT) of Mycobacterium tuberculosis complex. No malignant cells were detected in the ascitic fluid. Blood cultures for viral, bacterial, fungal, and mycobacterial pathogens were negative, as were gastric lavage cultures for mycobacteria. Respiratory secretions were not collected as the patient did not exhibit sputum or other respiratory symptoms at the time, and no further attempts were made. Further investigation revealed an isolated elevation of IgA (1180 mg/dL), with a minor β2 spike observed on serum protein electrophoresis. The free light chain ratio and serum/urine immunofixation studies showed no abnormalities. Prostate‑specific antigen (PSA) levels were within the normal range. To enhance diagnostic sensitivity, a PET scan was performed. This revealed abnormal, heterogeneous, and markedly increased 18F‑FDG uptake throughout the peritoneum and mesenteric fat, as well as in the right laterocervical, supraclavicular, and mediastinal lymph nodes (adenopathies). Histopathological analysis of the cervical fine‑needle lymph node biopsy revealed non‑necrotizing epithelioid granulomas with multinucleated giant cells (Fig. 2). No malignant cells were identified, consistent with previous findings. Microbiological studies were not conducted on this specimen. To date, no evidence of malignant cells has been detected, and all microbiological studies have been negative. Nevertheless, the primary clinical diagnostic hypothesis remained a peritoneal carcinomatosis due to an occult primary neoplasm.
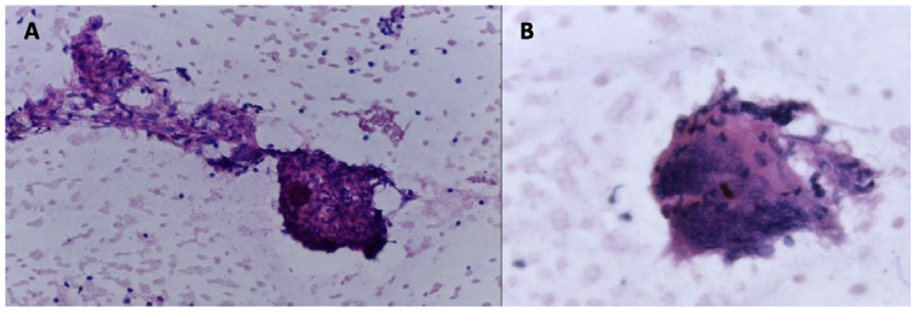
Figure 2. Fine needle cervical lymph nodule biopsy. A- Epithelioid granuloma (HE, 100x). B- Giant cell (HE, 400x). These findings corroborate the presence of a granulomatous process.
At this time, the patient’s functional status was severely compromised, with an Eastern Cooperative Oncology Group (ECOG) Performance Status Scale of 3. Given the clinical presentation, diagnostic uncertainties and the management of the suspected primary neoplasm, a multi- disciplinary consensus involving Nephrology, Palliative Care and Oncology, considered that a conservative treatment approach focusing on best supportive care was in the patient´s best interest and, accordingly, hemodialysis was withdrawn. After four days without hemodialysis, nearing three weeks of hospitalization, one (and only one) of the ongoing mycobacterial cultures from the ascitic fluid yielded a positive result. A subsequent thoracic CT scan revealed newer multiple millimetric centrilobular nodules distributed diffusely and bilaterally. Sputum NAAT for Mycobacterium tuberculosis complex was positive, while the urinary NAAT was negative. Magnetic resonance imaging and lumbar puncture ruled out central nervous system involvement. The diagnosis was revised to disseminated tuberculosis. Despite his frailty and poor prognosis, a decision was made to pursue curative intent with antibacillary therapy, and hemodialysis was subsequently resumed. The patient’s condition began to improve, particularly with the introduction of parenteral nutrition and protein‑enriched diets. He was discharged after eight weeks of hospitalization, being asymptomatic and demonstrating an overall improved clinical status and a significant improvement in nutritional parameters (BMI 17.0 kg/m² and serum albumin 28 g/L). Two years following the diagnosis the patient remains alive, still under maintenance hemodialysis.
DISCUSSION
This case highlights the diagnostic challenges encountered in a patient with end‑stage CKD on hemodialysis who was initially misdiagnosed with a progressive and irreversible condition, leading to the consideration of best supportive care and dialysis withdrawal. Our patient’s diagnostic march revealed no histopathological evidence of malignant cells, contradicting the hypothesis of peritoneal carcinomatosis. Despite clinical findings favoring mycobacterial infection such as increased ADA and granulomatous inflammation, initial diagnostic confirmation was delayed due to initial negative NAAT results and mycobacterial blood cultures. Furthermore, initial respiratory specimen collection was limited due to the absence of respiratory symptoms and sputum production. Microbiological studies were not performed on the lymphoid tissue, and further biopsies were deemed unfeasible given the patient’s deteriorating condition. The decision for a conservative treatment approach was reached through a multidisciplinary consensus involving Nephrology, Palliative Care and Oncology, heavily influenced by the patient’s frailty, poor functional status and presumed poor prognosis. In this context, the decision to withdraw hemodialysis was primarily medically driven, rather than arising from a psychosocial or shared decision‑making process.5 Nonetheless, the family was involved throughout, understanding the clinical deterioration and the futility of continued hemodialysis under the initial diagnostic impression. Upon the positive mycobacterial culture, the diagnosis was redefined to disseminated tuberculosis, prompting a shift to curative intent with antibacillary treatment and the resumption of hemodialysis. The potential for the patient to respond to active treatment was carefully considered. Despite the initial uncertainty, treatment was initiated, and the patient demonstrated a favorable response, evidenced by improved nutritional status and overall clinical condition at the time of discharge (Fig. 3).
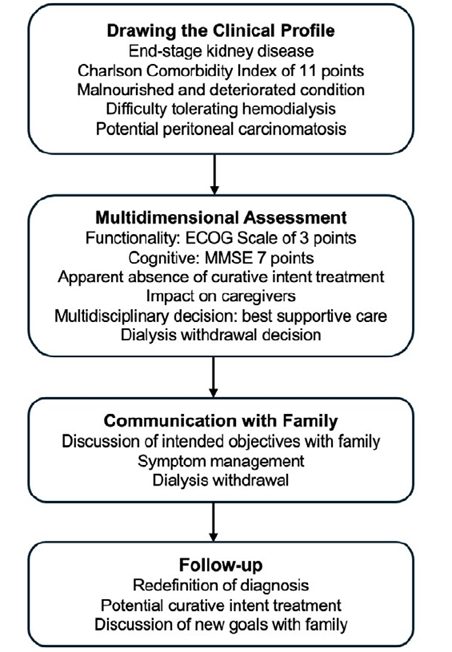
Figure 3 The flowchart of the decision‑making process taken in our patient. The diagram illustrates the initial clinical profiling, a comprehensive multidimensional assessment, the crucial communication with the family regarding symptom management and dialysis withdrawal, and a follow‑up stage where the diagnosis and treatment options were reconsidered.
Several nuances distinguished our case from the ideal scenario for dialysis withdrawal, which typically involves thoughtful, discussed, and shared decision‑making that respects the patient’s preferences, minimizes suffering, and addresses the psychological well‑being of the family.1,3,6The inpatient setting, coupled with the diagnostic challenges and the acute nature of the illness, further complicated the decision‑making process, underscoring the importance of well‑considered, discussed, and shared decisions in such circumstances. This highlights the importance of structured, proactive discussions regarding dialysis goals and end-of-life care, ideally conducted in advance of acute decompensation.
CONCLUSION
Dialysis withdrawal decisions are an inherent part of the nephrological practice and one of the most ethically and emotionally challenging decisions. This case underscores the critical importance of a thorough and accurate diagnostic process when considering withdrawal from hemodialysis, as diagnostic uncertainty could lead to premature and potentially harmful decisions. Making decisions in an acute context can often be precipitous, as time can provide clarity. A multidisciplinary, patient‑centered approach and robust diagnostic workup are essential to ensure that decisions about dialysis and end-of-life care are made with the patient’s best interests in mind.















