Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Portuguese Journal of Nephrology & Hypertension
Print version ISSN 0872-0169
Port J Nephrol Hypert vol.27 no.3 Lisboa Sept. 2013
Transplant glomerulopathy: clinico-pathologic features
Glomerulopatia do transplante: análise clínico-patológica
Cristina Silva1, Patricia Cotovio1, Maria Marques1, Nuno Afonso1, Maria Rosario Sancho3, Fernanda Carvalho2, Helder Trindade3, Armando Carreira1, Mario Campos1, Fernando Nolasco2
1 Department of Nephrology, Centro Hospitalar e Universitário Coimbra – Hospital Geral. Coimbra, Portugal.
2 Department of Nephrology, Hospital de Curry Cabral. Lisboa, Portugal.
3 Centro de Histocompatibilidade do Sul. Lisboa, Portugal.
ABSTRACT
Transplant glomerulopathy is a sign of chronic kidney allograft damage. It has a distinct morphology and is associated with poor allograft survival. We aimed to assess the prevalence and clinic-pathologic features of transplant glomerulopathy, as well as determine the functional and histological implications of its severity. We performed a single-centre retrospective observational study during an eight-year period. Kidney allograft biopsies were diagnosed and scored according to the Banff classification, coupled with immunofluorescence studies. The epidemiology, clinical presentation, outcomes (patient and graft survival) and anti-HLA alloantibodies were evaluated.
Transplant glomerulopathy was diagnosed in 60 kidney transplant biopsies performed for clinical reasons in 49 patients with ABO compatible renal transplant and a negative T-cell complement dependent cytotoxicity crossmatch at transplantation. The estimated prevalence of transplant glomerulopathy was 7.4% and its cumulative prevalence increased over time. C4d staining in peritubular capillaries (27.6%) was lower than the frequency of anti-HLA antibodies (72.5%), the majority against both classes I and II. Transplant glomerulopathy was associated with both acute (mainly glomerulitis and peritubular capillaritis) and chronic histologic abnormalities. At diagnosis, 30% had mild, 23.3% moderate and 46.7% severe transplant glomerulopathy. The severity of transplant glomerulopathy was associated with the severity of interstitial fibrosis. Other histological features, as well as clinical manifestations and graft survival, were unrelated to transplant glomerulopathy severity.
Key-Words:Antibody; glomerulopathy; histology; kidney; transplant.
RESUMO
A glomerulopatia do transplante e um sinal de lesão cronica do enxerto renal. Apresenta uma morfologia distinta e esta associada a uma pior sobrevida do enxerto. O objectivo do estudo foi avaliar a prevalência e características clinico-patológicas da glomerulopatia do transplante, bem como determinar as implicações funcionais e histológicas da sua gravidade. Estudo retrospectivo e observacional realizado num único centro durante um período de oito anos. As biopsias de enxerto renal foram diagnosticadas e avaliadas segundo a classificação Banff, complementadas com estudos de imunofluorescência. Analise da epidemiologia, apresentação clinica, outcomes (sobrevida do paciente e enxerto) e aloanticorpos anti-HLA. A glomerulopatia de transplante foi diagnosticada em 60 biopsias realizadas por motivos clinicos, em 49 receptores de transplante renal ABO compatível e com crossmatch antilinfocitario negativo por citotoxicidade dependente do complemento aquando transplante. A prevalência estimada da glomerulopatia do transplante foi 7.4%, a prevalência cumulativa aumentou ao longo do tempo. A deteção de C4d nos capilares peritubulares (27.6%) foi inferior a dos anticorpos anti-HLA (72.5%), a maioria contra as classes I e II. A glomerulopatia do transplante associou-se tanto as lesões histologicas agudas (principalmente glomerulite e capilarite peritubular), como cronicas. Aquando diagnostico, 30%, 23.3% e 46.7% apresentavam glomerulopatia do transplante leve, moderada e severa, respectivamente. Verificou-se uma associação entre a gravidade da glomerulopatia do transplante e a severidade da fibrose intersticial. Os outros indicadores histológicos, as manifestações Clinicas e a sobrevida do enxerto, foram independentes da gravidade da glomerulopatia do transplante.
Palavras-Chave:Anticorpo; glomerulopatia; histologia; rim; transplante.
INTRODUCTION
Transplant glomerulopathy (TG) is a unique pathologic entity distinct from other forms of chronic allograft injury and is associated with poor kidney transplant outcomes1.
It develops as a maladaptive/remodelling response to sustained or episodic endothelial injury, and is now widely accepted as the cardinal histologic phenotype of chronic antibody-mediated rejection2. It primarily is an endothelial pathology affecting kidney microcirculation and includes a constellation of histologic features on light and electron microscopies3, with immunofluorescence typically negative or with non-specific IgM and C34. Morphologic features of TG evolve over time5.
The clinical presentation of TG lags behind the initial histologic stages of the disease5. Its reported prevalence, based on biopsies performed for clinical reasons, varies from 1.6 to 7%4-9, but these numbers clearly underestimate its true prevalence.
The aim of this work was to assess the prevalence and clinico-pathologic features of TG, as well as determine the functional and histological implications of TG severity.
SUBJECTS AND METHODS
Renal transplant patients
From January 2004 to December 2011, a total of 1261 kidney allograft biopsies were evaluated at the Renal Morphology Laboratory of the Hospital de Curry Cabral. After the exclusion of biopsies with insufficient/inadequate material (n = 96), biopsies of patients with missing follow-up (n = 191) and donor kidney biopsies (n = 308), 666 cases were available for analysis. Of these, 60 were diagnosed with TG based on glomerular double contours, after excluding other conditions with similar histological changes (n = 2, one with membranoproliferative glomerulonephritis and another one with cortical necrosis). The biopsies were performed to investigate worsening renal function and/or proteinuria.
Clinical and laboratory data of patients were extracted from their medical records, including demographic and clinical variables, factors related to renal transplantation, post-transplantation day at biopsy, arterial hypertension, serum levels of creatinine and proteinuria at biopsy, history of acute rejection episodes and graft outcome. A semiquantitative analysis of the Panel-reactive antibodies (PRA) was performed: 0, 1 (1-25%), 2 (26-50%), 3 (51-75%) and 4 (> 75%).
Anti-HLA antibody analysis
Recipient sera taken in the peribiopsy period was screened for Human Leukocyte Antigens (HLA) antibodies and sera found to have anti-HLA class I and/or class II antibodies was further evaluated to determine HLA specificities using the Luminex technology.
Histopathology
The biopsies were diagnosed and scored according to the Banff classification10,11.
Transplant glomerulopathy was diagnosed by light microscopy based on double contours of the glomerular basement membrane10; the diagnosis was supported by immunofluorescence studies, which showed mesangial IgM and/or C3 or negative immunofluorescence findings. Electron microscopy studies were not performed.
Indirect Immunofluorescence Staining for C4d
C4d staining was done in all biopsies with available frozen tissue. Scoring of C4d staining was based on the percentage of stained tissue on immunofluorescence with an intensity > 1+ and a linear, circumferential staining pattern of peritubular capillaries.
It was graded as negative, focal (1-50% of sampled capillaries) or diffuse (> 50% of sampled capillaries) positive12.
Statistical analysis
Parametric variables were expressed as means ± standard deviation (SD), non-parametric variables were expressed as median and interquartile range (IQR) and, qualitative data were described as percentages.
Categorical variables were compared using the Chi-square test. Fishers exact test was used on a 2x2 table with ≥ 1 cells with an expected frequency ≤ 5. Differences between groups (i.e., TG severity/increasing Banff cg score from cg1– cg3) were analysed with the Kruskal-Wallis test. Graft survival was analysed by Kaplan-Meier and Cox proportional hazards modelling. All p-values were two-tailed and values < 0.05 were considered statistically significant.
Confidence intervals included 95% of predicted values.
Analyses were carried out using SPSS software (version 20; SPSS Inc. Chicago IL, USA).
RESULTS
Prevalence of TG and clinical findings
Of the 666 renal allograft biopsies included in the present study, TG was diagnosed in 60 biopsies, in 49 patients. The prevalence of TG in our renal transplant population, based on biopsies performed for clinical reasons, was 7.4%.
The cumulative prevalence of TG increased with time, from 16.3% at 3 years, to 26.5% at 5 years, 51% at 7 years, and 61.2% at 9 years posttransplant.
The demographic and clinical details of kidney transplantation recipients with TG are outlined in Table I. All patients were submitted to an ABO compatible renal transplant and had a negative T-cell crossmatch at transplantation using an anti-human globulin enhanced complement dependent cytotoxicity assay. The median HLA-A-B-DR mismatch was 4 and no kidney transplant was HLA identical. In the current study, 40.8% of patients with TG had experienced prior T-cell (85%) or antibody-mediated acute rejection (25%) episodes; 10.2% of patients had multiple previous acute rejection episodes.
Transplant glomerulopathy was diagnosed at a mean of 8.2 years post-transplant, with a mean serum creatinine of 2.8 mg/dL and with 78% of patients with urine protein levels > 1 g/day.

Anti-HLA antibodies
Of the 60 biopsies, 40 had available sera for anti-HLA analysis. Alloantibody was detected in 72.5% of cases, either against class I HLA (17%), or class II HLA (7%), or both (76%). Donor-specific antibodies (DSA) were identified in 58.6% anti-HLA antibody containing-sera, with 59.3% and 37.5% DSA for class I and class II, respectively (Table II). It is important to note that in a non-negligible percentage of cases, donor specificity could not be identified (reported as Indeterminate in Table II).
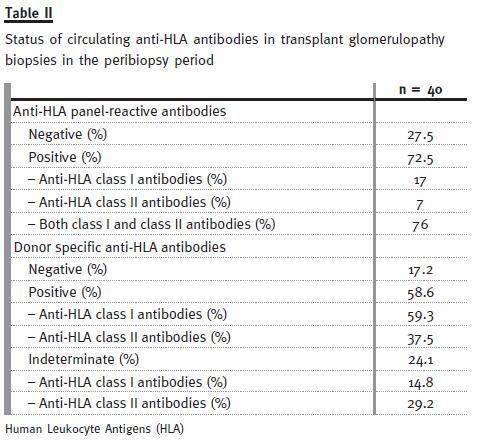
TG and other histologic lesions
At diagnosis, 18 of 60 biopsies (30%) had mild, 14 (23.3%) moderate and 28 (46.7%) severe TG (cg1, cg2, cg3, correspondingly).
The histopathology of allograft biopsies with TG is shown in Figure 1 and Figure 2.
We observed glomerulitis (Banff g score) and peritubular capillaritis (Banff cpt score) in 41.7% and 51.1% of TG biopsy specimens, respectively.
Other morphologic indicators of activity were uncommon: interstitial inflammation (Banff i score), tubulitis (Banff t score) and intimal arteritis (Banff v score).
Interstitial fibrosis (Banff ci score) and tubular atrophy (Banff ct score) were commonly seen in biopsies with TG, followed by other chronicity indexes: mesangial matrix expansion (Banff mm score), arteriolar hyaline thickening (Banff ah score) and vascular fibrous intimal thickening (Banff cv score).
C4d deposition in peritubular capillaries
C4d deposits in peritubular capillaries was observed in 16 (27.6%) biopsy specimens with TG at the time of diagnosis, with either diffuse staining in 11 (68.8%) or focal staining in five (31.2%).
Positive C4d staining was associated with anti-HLA antibodies (OR 9.3, 95% CI 1.1-82.6, p = 0.03). Nevertheless, about half (51.7%) of the C4d negative TG cases had circulating alloantibodies in the peribiopsy period.
Functional and histological implications of TG severity
Although arterial hypertension, allograft dysfunction and proteinuria were commonly seen in biopsies with TG, their severity did not relate significantly with higher Banff cg scores (p = NS).
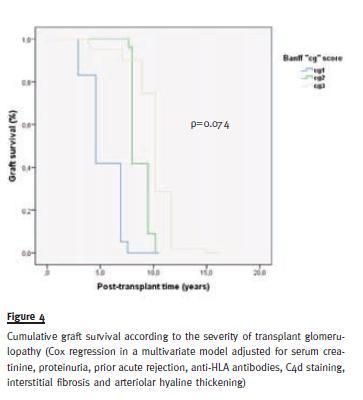
After a follow-up of 9.9 ± 5.2 years, one (2%) patient expired and 34 (69.4%) lost their allograft from causes other than patients death. No difference was observed in death-censored graft survival between TG groups (Figure 3 and Figure 4).

The severity of interstitial fibrosis (Banff ci score) increased with higher Banff cg scores (p = 0.042).
Overall, the frequency and severity of other histologic lesions (Banff g, cpt, i, t, v, ct, mm, ah and cv scores) did not increase significantly with growing severity of TG (p = NS).
DISCUSSION
Even though TG has been known since the early days of kidney transplantation, understanding of this injury pattern is still incomplete2. Transplant glomerulopathy is generally thought to occur as a late complication of kidney transplantation in association with decreased function, proteinuria and hypertension. Nevertheless, TG can occur within months of transplantation and in the absence of proteinuria6,8,13,14. Previous studies have consistently described a prevalence of 1.6–7% in conventional kidney transplant recipients based on biopsies performed for clinical causes4-9.
In our series, TG was diagnosed by light microscopy at a mean of 8.2 years after transplantation, with a minimum of 6 months. The estimated prevalence of TG was 7.4% and we found that TG was a time-dependent pathology. We are aware that these numbers under recognize TG, since many allografts develop TG sub-clinically, and many chronically failing kidney transplants are not biopsied.
Since our study was based on biopsies performed for clinical reasons, it was not surprising to have a great fraction of cases with graft dysfunction, proteinuria and hypertension at the time of the diagnosis of TG. The reported incidence of prior acute rejection was high (40.8%), with multiple episodes in 10.2% of recipients.
The percentage of T-cell and antibody-mediated rejection episodes was similar to that of previous reports5. Donor-specific antibodies, particularly HLA antigen class II antibodies, can spur insidious graft injury and, therefore, constitute a central causal factor for TG15-17. The reasons why particularly HLA antigen class II antibodies (with or without anti-class I) are associated with TG are unclear. Likely, there is an influence of antibody affinity avidity, as well as an ability to engage with Fc-γ receptors. The localization of target antigens also might be a determining factor2.
In our review, 72.5% of TG cases had circulating anti-HLA antibodies in the peribiopsy period, the majority against both classes I and II. Anti-HLA class II DSA were detected in 37.5% cases.
The reported incidence of C4d positivity in TG is quite variable17,18, probably related to criteria for biopsying patients, as well as differences in C4d detection methods and the defining criteria for C4d positivity. In our series, 27.6% had C4d deposition in peritubular capillaries.
Sis et al.5 suggested that the incidence of C4d deposition in TG was lower than the incidence of circulating alloantibodies, indicating that C4d deposition along capillaries might be negative or fluctuating and suggested that C4d negativity did not necessarily exclude alloantibody-mediated glomerular damage.
Our data showed that about half (51.7%) of the C4d negative TG cases had circulating alloantibodies in the peribiopsy period.
The morphologic indicators of activity in TG are C4d staining and microcirculation inflammation (glomerulitis and/or peritubular capillaritis)2. We observed glomerulitis and peritubular capillaritis in 41.7% and 51.1% of TG biopsy specimens, respectively. The incidence of other acute lesion scoring was residual.
Depending on the duration of the disease, chronic histologic abnormalities are seen in concurrence with TG, as a result of progression of capillary damage and subsequent ischaemic injury (segmental and/or focal glomerulosclerosis, interstitial fibrosis, tubular atrophy, arteriolar hyalinosis, arterial fibrous intimal thickening and, sometimes, loss of peritubular capillaries)2. Our data revealed a predominance of chronicity indexes, namely interstitial fibrosis (95%) and tubular atrophy (88.3%).
A major question addressed by this study was the functional and histological implications of TG severity. Our data revealed no differences in the frequency and/or severity of arterial hypertension, allograft dysfunction and proteinuria with TG severity; graft survival did not differ either. Transplant glomerulopathy was associated with acute and chronic histologic abnormalities. Nevertheless, the severity of these abnormalities was varied and, only interstitial fibrosis and TG were associated.
The results should be interpreted within the context of the studys limitations. As in all observational studies, the possibility of residual confounding or misclassification of outcomes cannot be excluded.
In conclusion, our revision assessed the prevalence and clinico-pathologic features of TG in a cohort of biopsies performed for clinical reasons. We demonstrated that the severity of TG was associated with the severity of interstitial fibrosis at the time of diagnosis of TG. Other histologic features, as well as clinical manifestations and graft survival, were unrelated to TG severity.
References
1. Cosio FG, Gloor JM, Sethi S, Stegall MD. Transplant glomerulopathy. Am J Transplant 2008;8(3):492-496. [ Links ]
2. Husain S, Sis B. Advances in the Understanding of transplant glomerulopathy. Am J Kidney Dis 2013;62(2):352-363. [ Links ]
3. Colvin RB. Antibody-mediated renal allograft rejection: Diagnosis and pathogenesis. J Am Soc Nephrol 2007;18(4):1046-1056. [ Links ]
4. Maryniak RK, First MR, Weiss MA. Transplant glomerulopathy: Evolution of morphologically distinct changes. Kidney Int 1985;27(5):799-806. [ Links ]
5. Sis B, Campbell PM, Mueller T, et al. Transplant glomerulopathy, late antibodymediated rejection and the ABCD tetrad in kidney allograft biopsies for cause. Am J Transplant 2007;7(7):1743-1752. [ Links ]
6. Habib R, Broyer M. Clinical significance of allograft glomerulopathy. Kidney Int Suppl 1993;43:S95-98. [ Links ]
7. Sijpkens YW, Joosten SA, Wong MC, et al. Immunologic risk factors and glomerular C4d deposits in chronic transplant glomerulopathy. Kidney Int 2004;65(6):2409-2418. [ Links ]
8. Suri DL, Tomlanovich SJ, Olson JL, Meyer TW. Transplant glomerulopathy as a cause of late graft loss. Am J Kidney Dis 2000;35(4):674-680. [ Links ]
9. Vongwiwatana A, Gourishankar S, Campbell PM, Solez K, Halloran PF. Peritubular capillary changes and C4d deposits are associated with transplant glomerulopathy but not IgA nephropathy. Am J Transplant 2004;4(1):124-129. [ Links ]
10. Racusen LC, Solez K, Colvin RB, et al. The Banff 97 working classification of renal allograft pathology. Kidney Int 1999;55(2):713-23. [ Links ]
11. Solez K, Colvin RB, Racusen LC, et al. Banff 07 classification of renal allograft pathology: Updates and future directions. Am J Transplant 2008;8(4):753-760 [ Links ]
12. Racusen LC, Colvin RB, Solez K, et al. Antibody-mediated rejection criteria – An addition to the Banff 97 classification of renal allograft rejection. Am J Transplant 2003;3(6):708-714. [ Links ]
13. Habib R, Zurowska A, Hinglais N, et al. A specific glomerular lesion of the graft: Allograft glomerulopathy. Kidney Int Suppl 1993;42:S104-111. [ Links ]
14. Banfi G, Villa M, Cresseri D, Ponticelli C. The clinical impact of chronic transplant glomerulopathy in cyclosporine era. Transplantation 2005;80(10):1392-1397. [ Links ]
15. Gaston RS, Cecka JM, Kasiske BL, et al. Evidence for antibody-mediated injury as a major determinant of late kidney allograft failure. Transplantation 2010;90(1):68-74. [ Links ]
16. Einecke G, Sis B, Reeve J, et al. Antibody-mediated microcirculation injury is the major cause of late kidney transplant failure. Am J Transplant 2009;9(11):2520-2531. [ Links ]
17. Sellares J, de Freitas DG, Mengel M, et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant 2012;12(2):388-399 [ Links ]
18. Al Aly Z, Yalamanchili P, Cortese C, Salinas-Madrigal L, Bastani B. C4d peritubular capillary staining in chronic allograft nephropathy and transplant glomerulopathy: an uncommon finding. Transpl Int 2005;18(7):800-805. [ Links ]
19. Batal I, Girnita A, Zeevi A, et al. Clinical significance of the distribution of C4d deposits in different anatomic compartments of the allograft kidney. Mod Pathol 2008; 21(121):1490-1498. [ Links ]
Dra Cristina Silva
Rua Luciano Freire, 6 r/c
1600-143 Lisboa
Portugal
E-mail: cristinamcsilva@gmail.com
Conflict of interest statement. None declared.
Received for publication: 14/06/2013
Accepted in revised form: 18/08/2013














