Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.32 no.1 Lisboa mar. 2018
CASE REPORT
Coexistence of pheochromocytoma and renal artery stenosis in a pediatric patient with hypertension
Inês Serras*1, Rute Baeta Baptista*2, Telma Francisco3, Anaxore Casimiro4, David Lito4, Rui Alves5, Margarida Abranches3
*equally contributing first authors
1 Department of Pediatrics – Centro Hospitalar Universitário do Algarve. Algarve, Portugal.
2 Department of Pediatrics – Hospital de Dona Estefânia; Centro Hospitalar de Lisboa Central. Lisboa, Portugal.
3 Pediatric Nephrology Unit – Hospital de Dona Estefânia; Centro Hospitalar de Lisboa Central. Lisboa, Portugal.
4 Pediatric Intensive Care Unit – Hospital de Dona Estefânia; Centro Hospitalar de Lisboa Central. Lisboa, Portugal.
5 Department of Pediatric Surgery – Hospital de Dona Estefânia; Centro Hospitalar de Lisboa Central. Lisboa, Portugal.
ABSTRACT
Pheochromocytoma and renal artery stenosis are surgically treatable causes of hypertension. Although rare, the coexistence of pheochromocytoma and renal artery stenosis has been described in case reports. Common pathophysiological mechanisms other than extrinsic compression may be involved in this association, such as catecholamine-induced vasospasm. The early recognition of the association of pheochromocytoma with renal artery stenosis is essential for appropriate treatment planning. We present the case of a previously healthy tenyear-old boy who presented with hypertensive encephalopathy, tachycardia and diaphoresis. Hypertension was found to be secondary to a catecholamine-producing tumor associated with coexisting renal artery stenosis.
Hypertension resolved a few months after successful pheochromocytoma excision, without renal artery revascularization.
Key-words: Child; hypertension; pheochromocytoma; posterior reversible encephalopathy syndrome; renal artery stenosis.
INTRODUCTION
Catecholamine-secreting neoplasms account for 0.1 - 0.6% of hypertension cases in adults1, and represent up to 1.7% of those cases in the pediatric population2. Pheochromocytomas are rare catecholamine-secreting neuroendocrine tumors derived from chromaffin cells located in the adrenal medulla3. Renal artery stenosis is present in about 3.3% of patients with pheochromocytoma4.
Pheochromocytoma and renal artery stenosis are both surgically treatable causes of secondary hypertension.
The failure to recognize a concomitant renal artery stenosis in a patient with pheochromocytoma may lead to persistent hypertension after successful tumoral resection. A missed diagnosis of pheochromocytoma in a hypertensive patient with renal artery stenosis may expose the unprepared patient to the risks of a catecholamine storm during the vascular intervention5.
Thus, the recognition of this rare association is essential for appropriate treatment planning6. There are several pathophysiologic mechanisms potentially involved in this combination, such as mechanical compression exerted by the mass on the renal hilum, fibrous bands emanating from the tumor, catecholamine-induced vasospam, fibromuscular dysplasia, and the myointimal proliferation in arteries long-term exposed to the effects of high blood pressure and vasoactive amines produced locally7. Some patients with a catecholamine-secreting tumor and renal artery stenosis, may actually present a pseudostenosis in which the reduction in the caliber of the renal artery is mostly dependent on the reversible vasospasm induced by the tumoral products. In this subgroup of patients treating only the pheochromocytoma may lead to spontaneous postoperative resolution of the renovascular component of hypertension.
On the contrary, patients with significant vascular remodelling may require renal artery revascularization to definitively treat hypertension. The distinction between these subgroups of patients is clinically relevant but challenging6.
CASE REPORT
A ten-year-old African boy with an unremarkable personal and family medical history presented with an eight-month history of occasional headaches and vomiting.
The patient was admitted to hospital in his hometown of Cape Verde after a first generalised tonic-clonic afebrile seizure.
During hospitalization, he was found to have an elevated blood pressure of 200/140 mmHg, difficult to control under captopril, nifedipine, and spironolactone.
Neurologically, the seizures evolved to generalized tonic–clonic status epilepticus, which resolved after a loading dose of phenobarbital. Following consciousness recovery, the child remained encephalopathic for 48 hours and reported amaurosis fugax. The laboratory findings were unremarkable, including the cerebrospinal fluid cytochemical examination. The patient was referred to our hospital after initiating empiric intravenous ceftriaxone and valproic acid at maintenance dose.
On admission to our hospital, the patient was asymptomatic and the general condition was good. On physical examination, blood pressure was 180/130 mmHg and pulse rate was 100 beats per minute, with no significant differences in either the four-limb measurements or in the measurements by body positioning.
The remaining physical exam was unremarkable, except for the fact that the patient was diaphoretic despite being apyretic.
Initial laboratorial investigation revealed hemoglobin 16.3g/dL; platelet count 476000/uL; white-cell count 6200/uL (neutrophils 3350/uL – 54.1%); C-reactive protein 2.3mg/L; creatinine 0.58mg/dL; blood urea nitrogen 19mg/dL; sodium 129mmol/L; potassium 4.3mmol/L; chloride 89mmol/L; and glucose 109mg/dL. Urine toxicological screen was negative.
Evidence of acute target organ damage was actively sought. Urinalysis, echocardiogram and fundoscopic examination after pupillary dilation were normal. The electroencephalogram showed no evidence of paroxysmal activity. Cerebral magnetic resonance imaging (MRI) revealed bilateral frontoparietotemporal cortical and subcortical white matter lesions, hyperintense in T2/FLAIR and hypointense in T1, coexisting with slight sulcal and ventricular enlargement.
Kidney ultrasound showed asymmetrical kidneys (longitudinal axis of the right kidney – 99.5 mm; longitudinal axis of the left kidney – 67.0 mm). A solid nodular mass (41.7mm x 28.0mm) in the left renal hilum was noted. The doppler study showed reduced peak systolic velocities on the left renal artery segment located distally to the mass (40cm/sec), compared with the right renal artery (120cm/sec). The origin of the left renal artery was inaccessible to ultrasonographic evaluation due to the interposition of the mass and aerocholia. The endocrinologic study documented a very significant elevation of the plasmatic and urinary normetanephrines. A state of hyperreninemic hyperaldosteronism was also identified (Table I). The following etiologic evaluation included an abdominopelvic MRI that showed a heterogeneous adrenal mass (43 x 42mm) reaching the left renal hilum, without invasion of the renal parenchyma. The nuclear scintigraphy with 123I-labeled metaiodobenzylguanidine (mIBG) confirmed the presence of an unilateral left adrenal pheochromocytoma, with no other glandular, skeletal or other ectopic 123I- mIBG captation.
The patient was admitted to the pediatric intensive care unit (PICU). Hyponatremia resolved upon discontinuation of spironolactone. Phenoxybenzamine was started at 0.2mg/Kg/day once daily, and increased gradually over three weeks (up to 2.6 mg/Kg/day) to achieve blood pressure levels inferior to the 95th percentile for age, sex and height. Labetalol perfusion was added to phenoxybenzamine in the third day of PICU admission, with doses ranging from 0.125 mg/kg/hr to 0.5mg/kg/hr, according to target blood pressure levels and normal heart rate for age.
The tumor was approached through an anterior laparotomy. It was adherent to the left renal vessels.
After careful dissection of the adhesions, a complete left adrenalectomy was performed, sparing the renal vessels and the renal parenchyma. The impression of the renal vessels in the resected tumoral mass could be seen macroscopically (Fig. 1). The histologic examination confirmed a benign pheochromocytoma with no capsular invasion.
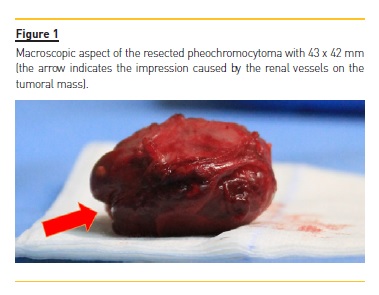
In the postoperative period, oral nifedipine was started due to persistent stage II hypertension with normal heart rate for age. Doppler sonography examination at one-month post-operative continued to document a reduced peak systolic velocity on the left renal artery (28.3 cm/sec) compared to that on the right renal artey (55.3 cm/sec). The patient was discharged home normotensive eighteen days after surgery. At two-month follow-up, biochemical and doppler parameters were normal (table 1). At five-months after surgery, the patient remained asymptomatic with blood pressure values in the 55th – 90th percentile and nifedipine was discontinued.
DISCUSSION
In our patient, hypertensive emergency was the initial presentation of systemic arterial hypertension. A better compliance with routine primary health care consultations might have allowed the diagnosis of hypertension in an asymptomatic phase, obviating the morbidity of prolonged severe hypertension8,9.
An acute and severe elevation in blood pressure (stage II hypertension) may be classified as hypertensive emergency or hypertensive urgency – according to the presence or absence of acute target organ damage, respectively. Although these definitions represent a continuum, the distinction is clinically relevant and evidence of acute organ damage should be actively screened. While a child with hypertensive urgency may be treated with oral medication, a child with hypertensive emergency should be admitted to an intensive care unit to receive intravenous medication, preferably continuous infusion rather than bolus. The goal is to gradually lower blood pressure (by no more than 25% of the overall planned reduction in the first eight hours) to avoid ischemic iatrogenic lesions10,11.
In the case presented, occasional headaches and vomiting were the early manifestations of hypertensive encephalopathy. This initial symptoms latter evolved to seizures and altered consciousness. Hypertensive encephalopathy is the most common manifestation of hypertensive emergency in children. In the early stages, it can manifest as isolated visual changes or headaches12.
Unfortunately, our patient remained symptomatic for several months, which may explain the sequelar lesions of posterior reversible encephalopathy syndrome (PRES) found in the cerebral magnetic resonance.
PRES is a clinico-radiological syndrome characterized by the abrupt onset of neurological symptoms (nausea/vomiting, headaches, visual abnormalities, seizures, consciousness impairment, and focal signs) associated with imaging findings of white matter vasogenic edema. Although PRES is also known as acute hypertensive encephalopathy or reversible posterior leukoencephalopathy, the term has become popular as it reflects the association with elevated blood pressure and the most frequent localization of neuroimaging findings along with the typical outcome. However, this nomenclature may be misleading. Although PRES is often benign, it may not be completely reversible in patients chronically exposed to the inciting event. Neuroimaging findings in PRES have been described as predominantly localized to the occipital and parietal regions, but patterns consistent with vasogenic edema can be found in a non-posterior distribution, as that exhibited by our patient13.
Hypertension in a child should be considered secondary until proven otherwise14,15. In our patient, the classic triad of tachycardia, diaphoresis and headaches was suggestive of a catecholamine excess. When a catecholamine-producing tumor is suspected, current guidelines recommend the measurements of fractionated metanephrines as the initial testing (either in urine or plasma, or both, as available). The parent catecholamines are metabolised intratumorally to metanephrines (i.e. norepinephrine to normetanephrine and epinephrine to metanephrine). Consequently, the measurements of fractionated metanephrines provide superior diagnostic sensitivity to measurements of the parent compounds16. In our patient, plasmatic levels of normetanephrines were tenfold above the upper limit of normal. Studies have shown the presence of a catecholamine-producing tumor is almost certain when an elevation of more than fourfold of the fractionated metanephrines is found. In this setting, an anatomic imaging study directed to the abdomen or pelvis is recommended, either computed tomography (CT) or magnetic resonance imaging (MRI). The functional studies, such as 123I-mIBG scintigraphy, are less sensitive but more specific. The 123I-mIBG is a radioiodinelabeled analog of norepinephrine taken up by tumor cells but not by normal adrenal medullary cells. 123I-mIBG scintigraphy is useful to detect multiple tumors when the CT or MRI are positive17. In this case, an abdominopelvic MRI showed a left adrenal mass that the 123I-mIBG scintigraphy confirmed to be a left pheochromocytoma without metastatic disease.
Total adrenalectomy is the preferred treatment option for unilateral adrenal gland tumors but the pertinence of simultaneous renal artery revascularization is less clear18. A benign pheochromocytoma is potentially cured surgically and no biopsy should be attempted19.
When the diagnosis of pheochromocytoma is confirmed biochemically, preoperative pharmacological alpha-1 blockade is initiated to prevent the risks associated with the surges of catecholamines during induction of anesthesia and manipulation of the tumor. Phenoxybenzamine is the therapy of choice in children. It is a noncompetitive and long-acting alpha-blocker reported to be superior to the short-acting alpha-1 blockers (doxazosin and prazosin) in preventing the effects of the intraoperative catecholamine storm. Phenoxybenzamine should be started at least seven to ten days before surgery at 0.2 mg/Kg/day once daily (maximum of 10 mg/dose) and titrated upward by 0.2 mg/Kg/day each four days, until blood pressure is inferior to the 95th percentile for age, sex and height. At least three days before surgery, a beta-blocker can be added to improve blood pressure control and, especially, to decrease reflex tachycardia to normal heart rate range for age. Note that beta-blockage should never be used prior to alpha-blockage, since unopposed alpha-receptors stimulation may lead to a severe hypertensive crisis. A few days before surgery oral salt intake is liberalized and patients receive intravenous fluid calculated to 1.5 times the daily maintenance fluid requirements to prevent postoperative hypotension2,20.
Complete blood pressure normalization usually occurs in the first week after pheochromocytoma excision in patients with no other causes of hypertension20.
In our patient, the initial persistence of hypertension postoperatively is likely to be due to residual renal artery stenosis (documented by the asymmetric peak systolic velocity and resistive index in renal arteries Doppler evaluation). However, the patient evolved favorably without performing any renal artery revascularization strategy. Complete resolution of biochemical and doppler signs of renal artery stenosis was achieved gradually after two months and the patient remains normotensive without any medication. The spontaneous resolution of the renal artery stenosis in this case suggests that it was at least partially attributable to reversible mechanisms and may be interpreted as a pseudostenosis. A second preoperative imaging evaluation under complete pharmacologic alpha-blockage may have been helpful, since a normalization of the imagiologic findings is expected in the context of a pseudostenosis6,7. The preoperative distinction between stenosis and pseudostenosis in a patient with pheochromocytoma is relevant. The first subgroup of patients require a vascular intervention to avoid persistent hypertension after tumor ressection, whereas those with a pseudostenosis may be adequately treated with pheochromocytoma excision only18.
References
1. Lenders JWM, Eisenhofer G, Mannelli M, Pacak K. Phaeochromocytoma. Lancet. 2005;366(9486):665–75. [ Links ]
2. Waguespack SG, Rich T, Grubbs E, Ying AK, Perrier ND, Ayala-Ramirez M, et al. A Current review of the etiology, diagnosis, and treatment of pediatric pheochromocytoma and paraganglioma. J Clin Endocrinol Metab. 2010;95(5):2023–37. [ Links ]
3. Dahia PLM. Pheochromocytoma and paraganglioma pathogenesis: learning from genetic heterogeneity. Nat Rev Cancer. 2014;14(2):108–19. [ Links ]
4. Sarathi V, Bandgar T, Lila AR, Deshpande AA, Dalvi AN, Patwardhan S, et al. Coexistence of pheochromocytoma/praganglioma and renal artery stenosis. Indian J Endocrinol Metab. 2012;16(6):1009–11. [ Links ]
5. Kota SK, Kota SK, Meher LK, Tripathy PR, Sruti J. Clinical Case Report Based Study Pheochromocytoma with renal artery stenosis : a case-based review of literature. J Cardiovasc Dis Res. 2012;3(1):36–9. [ Links ]
6. Vaze D, Trehan A, Saxena A, Joshi K, Narasimhan KL. Extraadrenal pheochromocytoma with renal artery pseudostenosis – an important pitfall. Urology. 2012;80(4):925–7. [ Links ]
7. Camberos A, Bautista N, Rubenzik M, Applebaum H. Renal artery stenosis and pheochromocytoma: coexistence and treatment. J Pediatr Surg. 2000;35(5):714–6. [ Links ]
8. Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140(3):e20171904. [ Links ]
9. Rao G. Diagnosis, epidemiology, and management of hypertension in children. Pediatrics. 2016;138(2). [ Links ]
10. Olgun G, John E. Hypertension in the pediatric intensive care unit. J Pediatr Intensive Care. 2016;5(2):50–8. [ Links ]
11. Ehrmann BJ, Selewski DT, Troost JP, Hieber SM, Gipson DS. Hypertension and health outcomes in the pediatric intensive care unit. Pediatr Crit Care Med. 2014;15(5):417–27. [ Links ]
12. Chandar J, Zilleruelo G. Hypertensive crisis in children. Pediatr Nephrol. 2012;27(5):741–51. [ Links ]
13. Ettinger N, Pearson M, Lamb FS, Wellons JC. Pediatric posterior reversible encephalopathy syndrome presenting with isolated cerebellar edema and obstructive hydrocephalus. J Neurosurg Pediatr. 2014;14(4):344–7. [ Links ]
14. Gupta-Malhotra M, Banker A, Shete S, Hashmi SS, Tyson JE, Barratt MS, et al. Essential hypertension vs. secondary hypertension among children. Am J Hypertens. 2015;28(1):73–80. [ Links ]
15. Rimoldi SF, Scherrer U, Messerli FH. Secondary arterial hypertension: When, who, and how to screen? Eur Heart J. 2014;35(19):1–12. [ Links ]
16. Pacak K, Eisenhofer G, Ahlman H, Bornstein SR, Gimenez-Roqueplo A-P, Grossman AB, et al. Pheochromocytoma: recommendations for clinical practice from the First International Symposium. Nat Clin Pract Endocrinol Metab. 2007;3(2):92–102. [ Links ]
17. Pappachan JM, Raskauskiene D, Sriraman R, Edavalath M, Hanna FW. Diagnosis and management of pheochromocytoma: a practical guide to clinicians. Curr Hypertens Rep. 2014; 16(7):442. [ Links ]
18. Kuzmanovska D, Sahpazova E, Kocova M, Damjanovski G. The interesting case phaeochromocytoma associated with reversible renal artery stenosis. Nephrol Dial Transplant. 2001;16(10):2092–4. [ Links ]
19. Bausch B, Wellner U, Bausch D, Schiavi F, Barontini M, Sanso G, et al. Long-term prognosis of patients with pediatric pheochromocytoma. Endocr Relat Cancer. 2014;21(1):17–25. [ Links ]
20. Romero M, Kapur G, Baracco R, Valentini RP, Mattoo TK, Jain A. Treatment of hypertension in children with catecholamine-secreting tumors: a systematic approach. J Clin Hypertens. 2015;17(9):720–5. [ Links ]
Rute Baeta Baptista, MD
Department of Pediatrics – Hospital de Dona Estefânia
Centro Hospitalar de Lisboa Central. Lisboa, Portugal
E‑mail: rute.baeta.baptista@gmail.com
Disclosure of potential conflicts of interest: none declared
Received for publication: 5 Dec 2017
Accepted in revised form: 1 Mar, 2018














