Introduction
Lumbar puncture (LP) is one of the most frequently performed invasive diagnostic procedures in children and adolescents.1
Common LP complications include post-dural puncture headache (PDPH) and mild pain at the puncture site (5-10%).2,3 Infrequent complications, such as cranial nerve palsy, meningitis, spinal epidermoid tumors, spinal hematoma, and cerebral herniation, have rarely been described in Pediatrics.4
Epidural cerebrospinal fluid (CSF) hygroma can occur after dura puncture due to CSF extravasation from the thecal sac. Although infrequent, the epidural collection can be large enough to cause neurological dysfunction.5
The authors present the clinical report of an adolescent with symptomatic epidural CSF collection following a non-traumatic LP.
Case report
An 11-year-old girl was admitted to a local hospital with a 24-hour history of fever, headache, and vomiting, without diarrhea. Family history and epidemiological data were unremarkable. On admission, she presented symptoms of confusion, but was clinically stable (temperature: 38.1°C, heart rate: 91 beats per minute, blood pressure: 120/60 mmHg, respiratory rate: 21 breaths per minute). On physical examination, the girl presented signs of meningeal irritation (positive Kernig and Brudzinski signs and nuchal rigidity), pharyngeal erythema, and tonsillar exudate. No skin rash was observed. Cardiovascular, respiratory, and abdominal examinations were unremarkable.
Laboratory results revealed increased white blood cell count (21000/mm3) and positive C-reactive protein (14.6 mg/dL). A rapid strep throat test was collected, with negative result. Other standard laboratory findings were within the normal range. Blood culture was negative. Brain computed tomography (CT) scan showed signs of cerebral edema with global loss of grey-white matter differentiation, narrow ventricles and descent of the cerebellar tonsils, and left-sided frontal and maxillary sinusitis, with no signs of cerebral venous thrombosis. Medical management with head positioning, restrictive intravenous fluid therapy, adequate analgesia, and intravenous dexamethasone was started. Dexamethasone was maintained for 13 days. Intravenous antibiotic therapy (ceftriaxone and ciprofloxacin) and acyclovir were also initiated. The child was transferred to a main hospital.
On admission, within 24 hours after brain CT, brain magnetic resonance imaging (MRI) was performed, showing findings consistent with meningoencephalitis and no signs of hydrocephalus. Imaging revealed slight thickening of the left frontobasal cortex, with restricted diffusion of the left cortical-subcortical frontobasal region. It also showed a 3-mm-thick left frontotemporal subdural empyema, an 8-mm-thick small epidural abscess on the left anterior frontobasal region, and left-sided frontal, ethmoid, and maxillary sinusitis. Non-traumatic LP was performed, revealing cloudy CSF with high white blood cell count (895/mm3, with 83% of polymorphonuclear cells), rare red blood cells/mm3, CSF/serum glucose ratio of 0.55, and protein content of 94 mg/dL. CSF Gram stain, culture, and multiplex polymerase chain reaction were negative. Nine hours after the procedure, the patient complained of urinary retention and lower back pain.
On day two of hospital stay, antimicrobial therapy was changed to intravenous ceftriaxone, vancomycin, and metronidazole. Empiric therapy was initiated to provide antimicrobial coverage to the most commonly identified pathogens in sinusitis and subdural empyema (anaerobes, Gram-negative bacteria, Staphylococcus aureus). Surgical treatment was required for management of sinusitis and subdural empyema, with no significant complications. Streptococcus viridans (susceptible to penicillin, clindamycin, and vancomycin) was isolated from maxillary sinuses.
After surgery, the patient was admitted to the Pediatric Intensive Care Unit (PICU) for recovery. During PICU stay, she complained of severe headaches, progressive lower back pain, constipation, and urinary retention. On examination, she was conscious but became acutely agitated with tactile allodynia. She was hemodynamically stable, with no motor impairment.
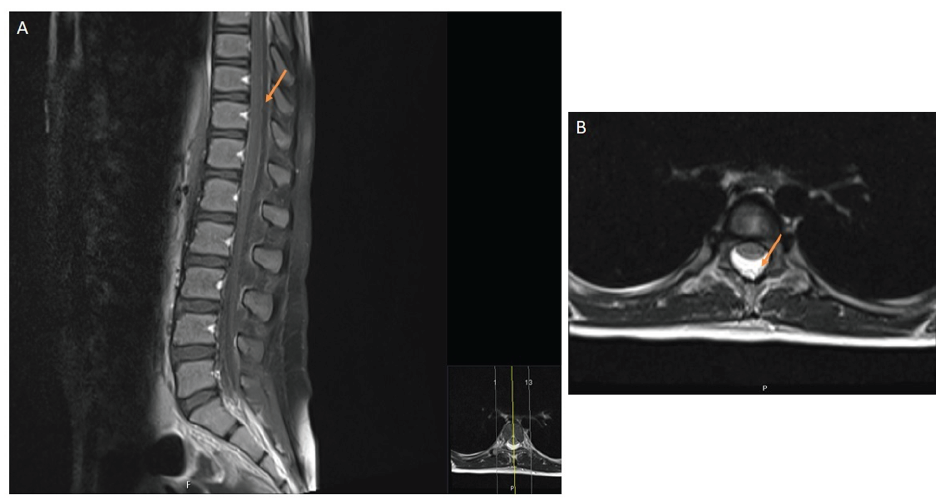
Spinal MRI was performed, revealing extensive dorsal and lumbar spinal epidural fluid collection and anterior displacement of the thecal sac, with subarachnoid space reduction (Figure 1). Imaging findings were consistent with post-LP complication.
During the first 24 hours, pain was not responsive to medical management, including nonopioid analgesics (paracetamol, metamizole, ketorolac) and opioid therapy (morphine).
Due to severe refractory pain and psychomotor agitation, the patient received an intravenous morphine infusion (maximum 30 µg/kg/h), fentanyl (maximum 2.5 µg/kg/h), and midazolam (maximum 3.1 µg/kg/min), and was mechanically ventilated for two days. She also required a Foley catheter for three days.
During PICU stay, conservative measures with hydration and strict bed rest were enforced. Patient’s symptoms improved within seven days, and she recovered without complications, being discharged forty days after admission. At three-month follow-up, the patient had complete resolution of clinical and imaging findings.
Discussion/conclusions
LP is a procedure commonly performed in pediatric age,6 mostly for the diagnosis of central nervous system infections. PDPH is its most frequent complication, potentially caused by transient intracranial hypotension due to the CSF leakage, although the exact underlying mechanism is still unknown.7,8 Needle characteristics (diameter and type of spinal needle), puncture technique (needle orientation), number of attempted dural punctures, and patient’s positioning after LP are factors influencing the occurrence of PDPH.7
Uncommon LP complications, such as epidural and subdural hematoma, symptomatic epidural CSF collection, subarachnoid haemorrhage, cranial nerve palsy, meningitis, and cerebral herniation, have rarely been reported in children.2,5
Although uncommon in infants and children, epidural CSF collection after LP has long been acknowledged among the Pediatric population, from neonates to adolescents.5,9
The incidence of post-LP epidural CSF collection is unknown, but it is believed to be higher than suspected. Koch et al. retrospectively analysed 25 MRIs performed in children with new symptoms following LP.5 All MRI scans showed abnormal dorsal spinal epidural collection and exhibited significant anterior dura displacement.2 Kiechl-Kohlendorfer et al. conducted a prospective study including 33 newborns who underwent ultrasound evaluation after LP to determine the occurrence of abnormal fluid collection. The authors demonstrated that CSF leakage is a frequent finding after LP, developing in 64% of all neonates enrolled in the study.9
Although predisposing risk factors and pathophysiology remain unknown, neonates and children with underdeveloped connective tissue in the epidural space may be at higher risk of this complication.4
These collections are usually dorsally located on MRI and extensive, and may compromise the thecal sac, resulting in transient cauda equina syndrome (CES).2,10
In the present case, typical MRI findings were identified. These included abnormal epidural CSF collection with anterior thecal sac displacement, resulting in decreased subarachnoid space.
Epidural CSF collection may be a misdiagnosed complication and only detected in the presence of significant symptoms, such as severe headache, lower back pain, constipation, and urinary retention, as occurred in this patient. Epidural CSF collection can be severe enough to result in CES. Amini A et al. reported a case of epidural CSF leak after LP causing CES in a four-year-old girl.4
CES is uncommon in pediatric age and results from compression or inflammation of nerve roots of cauda equina. This condition commonly comprises motor and sensory abnormalities of the lower limbs, along with bladder and bowel dysfunction. There are numerous CES classifications, based on location, disease type, and time of onset, and the condition is associated with various disorders, potentially occurring after LP.4,11
According to Koch et al., the extent of epidural CSF collection does not seem to be related to the number of LP attempts.2 Typically, approximately 90% of symptoms start within the first 72 hours after LP and resolve within ten days, as observed in the present case.12,13
In Pediatric patients, conservative management with hydration, analgesia, and bed rest represents the first treatment choice, with patients usually recovering without neurological deficits.4,5
Epidural blood patch (EBP) therapy is indicated if conservative treatment is ineffective, as in patients with severe and persistent pain, with a reported success rate of 90%.12,13 In EBP, a small volume of autologous blood is injected into the epidural space between the ligamentum flavum and dura.14 The proposed mechanism is that clotted blood adheres to dura, resolving the leak and allowing CSF pressure to normalize.14,15 In the present case, although the adolescent achieved complete recovery and symptomatic relief in the expected period, autologous EBP could have been considered to attain earlier recovery.
In conclusion, symptomatic epidural CSF collection can occur following LP and should be considered in the differential diagnosis in children if headache and back pain emerge after LP. Patients with persistent severe back pain and/or signs of spinal cord disease (backpain, urinary retention, loss of bowel and bladder control, weakness of lower limbs, sensory loss) after LP should undergo spine MRI. Close follow-up is required to ensure complete recovery.