Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Jornal Português de Gastrenterologia
versão impressa ISSN 0872-8178
J Port Gastrenterol. vol.19 no.5 Lisboa set. 2012
Acute hepatitis induced by fosfomycin: A case report and review of the literature
Hepatite aguda induzida por fosfomicina: relato de um caso clínico e revisão da literatura
Rosa Ferreiraa,∗, Joana Torresa, Joana Raposob, Margarida Ferreiraa, Sofia Mendesa, Cláudia Agostinhoa, Mário Júlio Camposa
a Gastroenterology Service, Hospitalar Center of Coimbra, Portugal
b Anatomic Pathology Service, Hospitalar Center of Coimbra, Portugal
*Corresponding author
Abstract
Fosfomycin is a broad spectrum antibiotic, belonging to the group of phosphonic acid antibiotics that is active against most urinary tract pathogens. It is an effective and safe antibiotic for the treatment of uncomplicated urinary tract infections, which makes it a frequently used drug in our country in this clinical setting. In general, it is well-tolerated and does not appear to cause serious reactions. Most reactions are gastrointestinal. Hepatotoxicity with this drug has been rarely described.We report a case of acute hepatitis induced by a single 3 g dose of fosfomycin, in a previously healthy female adult.
Keywords Acute hepatitis; Fosfomycin; 3 g Single dose; Adverse drug effect; Acute cystitis
Resumo
A fosfomicina é um antibiótico de largo espectro, que pertence ao grupo do ácido fosfónico e é eficaz contra a maioria dos microorganismos do tracto urinário. Pela sua eficácia e segurança no tratamento de infecções do tracto urinário não complicadas é amplamente usado no nosso país nesta situação clínica. É um fármaco geralmente bem tolerado e que não parece causar efeitos adversos graves. A maioria dos efeitos laterais relatada é gastrointestinal. A hepatotoxicidade induzida com a fosfomicina tem sido raramente descrita.Os autores descrevem um caso de hepatite aguda induzida por uma dose única de 3 g de fosfomicina, numa doente adulta previamente saudável.
Palavras-chave Hepatite aguda; Fosfomicina; Dose única de 3 g; Efeitos adversos; Cistite aguda
Introduction
Fosfomycin is an oral antibiotic derived from phosphonic acid that has been widely used in the treatment of uncomplicated urinary tract infections.1,2 Its potent and enduring activity against urinary pathogens has been confirmed in Europe3 and USA.4 Since 1988 fosfomycin has been extensively used in several European countries for single-dose therapy of uncomplicated urinary tract infections. After a single 3 g dose, fosfomycin exhibits very high and sustained urinary concentrations that rapidly kill pathogens reducing the opportunity for mutant selection. The resistance rates of fosfomycin remain, therefore, extremely low (about 1%) worldwide.5 Furthermore, fosfomycin is well tolerated, with a low incidence of adverse events. These consist mainly of gastrointestinal symptoms that are ordinarily transient, mild and self-limiting.1,6
The authors present a case of a 24-year-old woman with acute hepatitis induced by a single 3 g dose of fosfomycin for acute cystitis.
Case report
A 24-year-old woman, with no significant past medical history, presented to the emergency department with nausea, fatigue, increasing muscle weakness, gradually worsening jaundice and dark urine, for four weeks. The symptoms started one week after taking a single 3 g dose of fosfomycin for acute cystitis. She denied any accompanying symptoms, such as rash, arthralgias, fever or adenopathies.
She also denied taken any other medications including over-the counter medications, herbal or traditional medicines. There was no history of drugs or alcohol abuse, past administration of blood products or blood transfusion, or previous hepatitis. She denied recent travels. There was no family history of liver diseases.
On physical examination, the vital signs were normal and there were no remarkable findings except for icteric skin and sclera. Abdominal and neurological examinations were normal.
The hematological data revealed hemoglobin of 12.6 g/dL; total white cell count of 11, 8×103/L (3.3% of lymphocytes and 0.5% of eosinophils); platelet count of 437×103/L and prothrombin time of 14 s (INR 1.2). The results of liver tests were: total bilirubin 195_mol/L (NR: <22) with 124_mol/L of conjugated, alanine aminotransferase (ALT) 1833 IU/L (NR: 10-66), aspartate aminotransferase 1467 IU/L (NR: 15-46), alkaline phosphatase (ALP) 86 IU/L (NR 38-136), gamma-glutamyl transferase 68 IU/L (NR: 12-58) and LDH 1531 IU/L (NR: 313-618).
Electrolytes, serum albumin, iron and transferrin saturation were normal. IgM anti-HAV, HBsAg, IgM anti-HBc and anti-HCV antibodies were negative. Anti-HIV, anti-CMV, anti- EBV and anti-HSV were also negative. Auto-antibodies (ANA, ANCA, Anti-LKM, AMA and ASMA) were negative. 24 h-urinary copper, ceruloplasmin, _-fetoprotein and _-1 antitrypsin were within normal range. Liver ultrasonography showed no significant abnormality except for increased echogenicity.
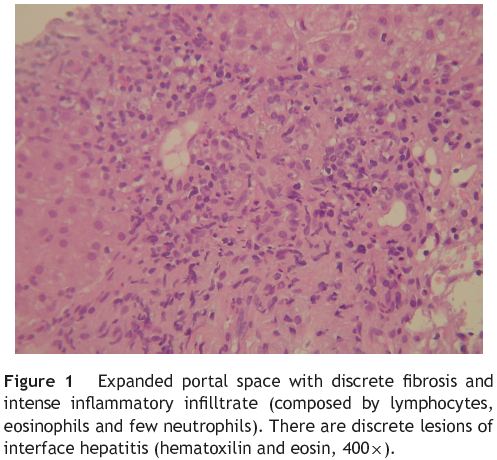
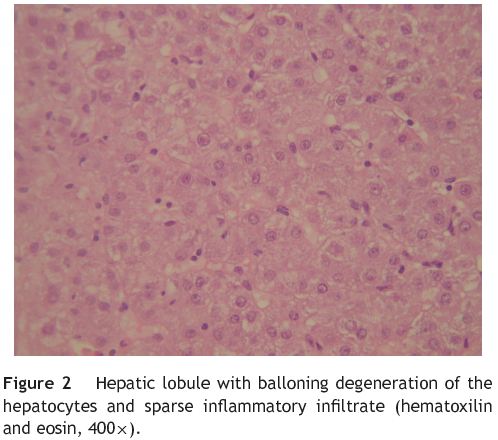
A liver biopsy by percutaneous route was then performed without complications. The biopsy material was fragmented and had lesions located in the portal spaces and hepatic lobules. The histological examination showed expansion of the portal spaces with scant fibrosis and intense inflammatory infiltrate composed by lymphocytes, eosinophils and few neutrophils. There were also focal lesions of interface hepatitis (Fig. 1). The hepatic lobules showed moderate inflammatory infiltrate, similar to the one noticed in the portal spaces, ballooning degeneration of the hepatocytes (Fig. 2) and isolated apoptotic bodies throughout the entire studied material. It was observed focal hepatic necrosis with collapse of the parenchyma, more severe in the perivenular zone, along with discrete fibrosis. There was also focal hepatic steatosis. No granulomas were detected. These findings were consistent with acute hepatitis and were highly compatible with toxic/pharmacological etiology.
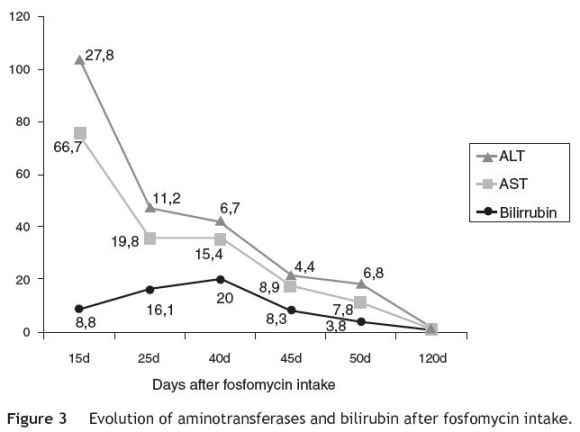
A gradual decrease in liver enzymes was seen; total bilirubin continued to rise and reached a peak 40 days after the intake of fosfomycin, and then it also started to decline (Fig. 3). The patient improved symptomatically, in parallel with the decline in aminotransaminases. Three months after fosfomycin intake, all liver function tests had normalized (Fig. 3).
Two years after, the patient remains asymptomatic and without alteration of the liver enzymes.
Discussion
Fosfomycin is a widely used, broad-spectrum antibiotic, which exhibits a rapid bactericidal activity against a large number of aerobic Gram positive and Gram-negative bacteria.1,2 It is approved as a single-dose therapy (3-g oral dose) for the treatment of uncomplicated urinary tract infections (acute cystitis) in women.7
Usually, it is a well-tolerated drug and does not appear to cause serious reactions.
Reported adverse events are usually mild and last only 1-2 days, resolving without treatment. The overall incidence of side-effects is 6% with oral therapeutic dosing and 7% with parenteral administration. Gastrointestinal complaints, mostly diarrhea, are the most frequent. Dizziness, eadache and vaginitis have also been reported.6 In terms of liver adverse events, only rare cases of asymptomatic and mild liver enzyme abnormalities have been described.8
To our knowledge there are three cases in the literature of fosfomycin induced-liver injury: a case of recurrent fosfomycin-induced hepatic toxicity with drug rechallenge in a 30-year-old woman with cystic fibrosis in France,9 a case of acute severe hepatitis in Japan10 and a case of fosfomycininduced liver disease in a 50-year-old woman in China.11 The authors have no additional information about the case reported in Japan because this article was published in Japanese. In the other two cases, liver enzymes increased 3-4 days after fosfomycin administration and returned to normal levels within a week to a month after the withdrawal of the drug. In our patient, the normalization of all liver function tests took three months to occur. In these cases, fosfomycin induced hepatotoxicity was observed with higher doses during a longer period of time than in our patient (3 g single dose), suggesting that its hepatotoxic effect may be dose independent. While the liver injury observed in our case can be classified as hepatocellular (ALT >2 upper limits of normal (ULN) or ALT/ALP ratio ≥5), in the Chinese case it was mixed (ALT >2 ULN and 2< ALT/ALP ratio >5) and in the French case it cannot be classified because, despite the elevation of ALT, the ALP value was not mentioned.
In our patient, acute hepatitis appears to be causally related to the administration of fosfomycin, based on a temporal relationship, negative serology for acute viral infection, negative autoantibody markers, exclusion of other drugs or other potentially hepatotoxic agents, and suggestive histological alterations in the biopsy. Liver biopsy was not performed in none of the two previously reported cases, and so these histological findings cannot be compared. Proof is not possible in the absence of a second exposition to the drug for ethical reasons.
Based on the Roussel Uclaf Causality Assessment Method (RUCAM) scale,12 a score of 9 was obtained in our patient, indicating that the association between fosfomycin and the liver injury was highly probable. According to the Maria and Vitorino scale,13 this causality was considered to be possible (score of 11).
Drug-induced liver injury (DILI) diagnosis remains a challenge for physicians and it is usually based on exclusion of other possible causes of hepatic dysfunction and on the temporal association between drug administration and the onset of liver disease.14,15 In our case, hepatic biopsy was a helpful tool to establish the diagnosis.
Although not performed in our institution, the drug lymphocyte stimulation test (DLST) is a laboratory test that can be useful to ascertain the diagnosis of DILI and to identify a single causative drug. It consists in culturing a patients lymphocytes in the presence of the suspected drug. Lymphocyte proliferative response is determined by monitoring 3H-thymidine uptake.16 It is most frequently positive on immune mediated liver injury.17 However, recent findings of associations between specific HLA haplotypes and DILI,18-20 which does not have hypersensitivity features, have highlighted the DLSTs potential value.21 In fact, a recente diagnostic scale, the Digestive Disease Week-Japan, already includes DLST.17 Nevertheless, low sensitivity (around 50%), lack of causality between a positive result and liver injury, lack of standardization and restricted availability outsider Japan limit its use.21,22 And so, some authors advocate that it should be considered in selected cases, such as those in which a single causative agent cannot be determined.22 We considered that DSLT was not mandatory in our patient since fosfomycin was the only drug used.
In a prospective study, drug-induced liver injury was caused by a single prescription medication in 73% of the cases and antibiotics were the single largest class of hepatotoxic agents.15
In summary, we report a potential case of acute hepatocelular lesion caused by fosfomycin. Being a commonly used antibiotic, physicians should be aware of this rare but potentially serious adverse drug reaction.
References
1. Patel SS, Balfour JA, Bryson HM. Fosfomycin tromethamine. A review of its antibacterial activity, pharmacokinetic properties and therapeutic efficacy as a single-dose oral treatment for acute uncomplicated lower urinary tract infections. Drugs. 1997;53:637-56. [ Links ]
2. Guay DR. Contemporary management of uncomplicated urinary tract infections. Drugs. 2008;68:1169-205. [ Links ]
3. Kahlmeter G. An international survey of the antimicrobial susceptibility of pathogens from uncomplicated urinary tract infections: the ECO SENS project. J Antimicrob Chemother. 2003;51:69-76. [ Links ]
4. Fuchs PC, Barry AL, Brown SD. Fosfomycin tromethamine susceptibility of outpatient urine isolates of Escherichia coli and Enterococcus faecalis from ten North American medical centres by three methods. J Antimicrob Chemother. 1999;43: 137-40. [ Links ]
5. Schito GC. Why fosfomycin trometamol as first line therapy for uncomplicated UTI? Int J Antimicrob Agents. 2003;22 Suppl. 2:79-83. [ Links ]
6. Stein GE. Comparison of single-dose fosfomycin and a 7-day course of nitrofurantoin in female patients with uncomplicated urinary tract infection. Clin Ther. 1999;21:1864-72. [ Links ]
7. Stein GE. Single-dose treatment of acute cystitis with fosfomycin tromethamine. Ann Pharmacother. 1998;32:215-9. [ Links ]
8. Koeda T, Odaki M, Sasaki H, Yokota M, Watanabe H, Kawaoto H, et al. Toxicological studies on fosfomycin-Na salt. Jpn J Antibiot. 1979;32:61-105. [ Links ]
9. Durupt S, Josserand RN, Sibille M, Durieu I. Acute, recurrent fosfomycin-induced liver toxicity in an adult patient with cystic fibrosis. Scand J Infect Dis. 2001;33:391-2. [ Links ]
10. Matsumori A, Yoneda S, Kobayashi Y, Takeda K, Andoh M, Yamane Y, et al. A case of acute severe hepatitis induced by fosfomycin. Nippon Shokakibyo Gakkai Zasshi. 2005;102:1207-11. [ Links ]
11. Wang YP, Shi B, Chen YX, Xu J, Jiang CF, Xie WF. Drug-induced liver disease: an 8-year study of patients from on gastroenterological department. J Dig Dis. 2009;10:195-200. [ Links ]
12. Danan G, Benichou C. Causality assessment of adverse reactions to drugs-I. A novel method based on the conclusions of international consensus meetings: application to drug-induced liver injuries. J Clin Epidemiol. 1993;46:1323-30. [ Links ]
13. Maria VA, Vitorino RM. Development and validation of a clinical scale for the diagnosis of drug-induced hepatitis. Hepatology. 1997;26:664-9. [ Links ]
14. Danan G, Bénichou C. Causality assessment of adverse reactions to drugs-I. A novel method based on the conclusions of international consensus meetings: application to drug induced liver injuries. J Clin Epidemiol. 1993;46:1323-30. [ Links ]
15. Lee WM. Drug-induced hepatotoxicity. N Engl J Med. 2003;349:474-85. [ Links ]
16. Takikawa H, Takamori Y, Kumagi T, Onji M, Watanabe M, Shibuya A, et al. Assessment of 287 Japanese cases of drug induced liver injury by the diagnostic scale of the International Consensus Meeting. Hepatol Res. 2003;27:192-5. [ Links ]
17. Maria VA, Vitorino RM. Diagnostic value of specific T cell reactivity to drugs in 95 cases of drug induced liver injury. Gut. 1997;41:534-40. [ Links ]
18. Kindmark A, Jawaid A, Harbron CG, Barratt BJ, Bengtsson OF, Andersson TB, et al. Genome-wide pharmacogenetic investigation of the hepatic adverse event without clinical signs of immunopathology suggests an underlying immune pathogenesis. Pharmacogenomics J. 2008;8:186-95. [ Links ]
19. Hirata K, Takagi H, Yamamoto M, Matsumoto T, Nishiya T, Mori K, et al. Ticlopidine-induced hepatotoxicity is associated with specific human leukocyte antigen genomic subtypes in Japanese patients: a preliminary case---control study. Pharmacogenomics J. 2008;8:29-33. [ Links ]
20. Daly AK, Donaldson PT, Bhatnagar P, Shen Y, Peer I, Floratos A, et al. DILIGEN Study; International SAE Consortium. HLA-B*5701 genotype is a major determinant of drug-induced liver injury due to flucloxacillin. Nat Genet. 2009;41:816-9. [ Links ]
21. Watkins PB. Biomarkers for the diagnosis and management of drug-induced liver injury. Semin Liver Dis. 2009;29:393-9. [ Links ]
22. Tajiri K, Shimizu Y. Practical guidelines for diagnosis and early management of drug-induced liver injury. Worl J Gastroenterol. 2008;14:6774-85. [ Links ]
Funding
The authors declare that there is any financial support for this manuscript.
Conflicts of interest
The authors have no conflicts of interest to declare.
*Corresponding author
E-mail address: rosa.l.ferreira@gmail.com (R. Ferreira).
Received 2 December 2010; accepted 3 April 2011













