Introduction
Technological advances in digital impression methods and production systems have led to the increasing use of CAD/CAM (computer‑aided design/computer‑aided manufacturing) systems in the manufacture of indirect prosthetic restorations.1,2,3
Several materials with different compositions and physical properties are currently available to use with these systems.3
Ceramics (glass or polycrystalline), characterized as resistant, rigid yet brittle, and having excellent esthetics and high wear resistance, are examples of this development.3,4
Composite resin‑based materials were later introduced and nowadays serve as an alternative to conventional ceramics.3 These materials have gained popularity due to their mechanical properties, high esthetic potential, wear similar to tooth enamel’s, and easy repair of minor defects induced by function.3‑5
Composite resins for use with CAD/CAM systems usually come in blocks or disks consisting of a polymeric matrix reinforced by inorganic fillers (porcelains, glasses, ceramics, or glass ceramics), differing in type, size, and amount of filler.6,7
In an attempt to combine the favorable characteristics of ceramics with those of composite resins, new materials were developed and classified as resin nanoceramics.7,8These materials contain a polymeric matrix with the addition of refractory inorganic compounds (>50% by weight).7 They are recommended for indirect restorations, such as full‑contour crowns, inlays, onlays, and veneers.9 These materials have shown a modulus of resilience greater than dental ceramics’, and although they are less resistant to wear, they cause less abrasion to the opposing dentition.9,10 Furthermore, manufacturers claim they are less susceptible to fracture and chipping because their modulus of elasticity is close to that of dentin.3
In general, polymer‑based materials developed for CAD/CAM use performed better in flexural testing than analogous ceramic materials due to the combination of a relatively high flexural strength with a low modulus of elasticity.4,9 This results in an increased ability to withstand occlusal loading by undergoing more elastic deformation before fracture, being more flexible and less brittle than purely ceramic materials.4,9,10
Additionally, they exhibited flexural properties similar to those of dentin, making them an acceptable choice for single‑unit prosthetic restorations.9,10
Although no single property can be used to predict a material’s clinical success or failure, parameters such as flexural strength, fracture resistance, modulus of elasticity, and modulusof resilience provide important information about the dynamic behavior of these materials.11 Previous studies have shown that the type of cement significantly affects the fracture resistance of crowns, but the materials tested were mostly feldspathic and glass ceramics.12,13 Other studies have evaluated the influence of cement on the fracture resistance of different indirect composite resins.5,14‑17 However, until now, there is a lack of available literature regarding its influence on the fracture resistance of full‑contour crowns milled from some of the new resin nanoceramic CAD/CAM blocks; this justifies the relevance of this pilot study.
The objective of this study was to evaluate the influence of the type of cement on the fracture resistance of full‑contour resin nanoceramic crowns cemented over dental preparations with knife‑edge margins. The null hypothesis was as follows: the type of cement does not influence the fracture resistance of full‑contour resin nanoceramic crowns cemented over preparations with knife‑edge margins.
Material and methods
In this pilot study, 20 full‑contour resin nanoceramic crowns were milled from Cerasmart270™ CAD/CAM blocks and randomly divided into two experimental groups. This material is a high‑density composite resin containing a polymeric matrix with the addition of silica nanoparticles of up to 1 μm18 and barium glass (77% by weight).
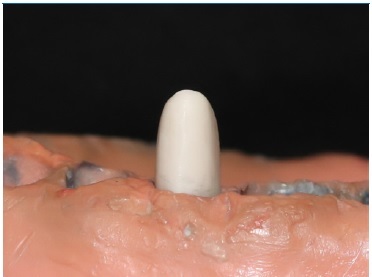
A dental preparation for a full‑contour crown was performed on a lower right second premolar typodont tooth mounted on a dental anatomical model (ANA‑4, Frasaco GmbH®, Germany) (Figure 1). Axial guiding grooves were made 1‑mm deep with a round end taper diamond bur (FG G850.010, Edenta AG. Hauptstrasse, Switzerland). The axial reduction of 1.5 mm was made with a needle diamond bur (FG G859L.016, Edenta AG. Hauptstrasse, Switzerland) and the occlusal reduction of 2.0 mm with a football diamond bur (FG G368.023, Edenta AG, Hauptstrasse, Switzerland) while maintaining the tooth morphology. The 45º‑angle bevel of the functional cusp and the knife‑edge margin were also performed with the needle Diamond bur. The dental preparation was finished and polished with an Arkansas stone (Abrasive 025, Edenta AG. Hauptstrasse, Switzerland) followed by a fine‑grain polishing rubber point (Exa Intrapol 050, Edenta AG. Hauptstrasse, Switzerland) to smooth the preparation’s surface. The whole procedure was executed using rotary instruments with constant water cooling (Bora LK 1600732‑001, Bien‑Air Dental, Bienne, Switzerland; CA 1:1 Standard 1600424‑001, Bien‑Air Dental, Bienne, Switzerland).
The dental preparation was digitally scanned (Zirkonzahn S600 ARTI, Zirkonzahn GmbH, Austria) to obtain its three‑dimensional model. An anatomical crown of a lower second premolar was designed using dedicated CAD software (Zirkonzahn. Modellier, Zirkonzahn GmbH, Austria).
Twenty full‑contour crowns with a 0.2‑mm margin thickness were milled from resin nanoceramics CAD/CAM blocks (Cerasmart270™, GC Corporation, Tokyo, Japan; Lot 1901101/1902071; Val: 01/2024) (Figure 2) using a milling unit (Zirkonzahn M5 Heavy Metal, Zirkonzahn GmbH, Austria), as well as a replica of the dental preparation in a cobalt-chrome alloy (Zirkonzahn Sintermetall, Zirkonzahn GmbH, Austria), which was used for mechanical testing (Figure 3). The internal surface of each crown was sandblasted with 50‑μm aluminum oxide powder (1.5 bar) for 10 seconds at a distance of 10 mm (Basic classic 2945‑2026, Renfert GmbH, Hilzingen, Germany). Before each test, the cobalt-chrome alloy die was cleaned with a wet compress and dried with an oil‑free airstream to obtain a clean surface, thus preventing contamination.
Figure 2 Full‑contour crown milled from Cerasmart270™ resin nanoceramic CAD/CAM blocks (GC Corporation, Tokyo, Japan).
Group 1 crowns were luted with dual‑cure self‑adhesive resin cement (G‑CEM LinkAce™, GC Corporation, Tokyo, Japan; Lot 180611B; Val: 07/2020) according to the manufacturer’s instructions. The self‑adhesive resin cement was dispensed homogeneously using a self‑mixing syringe. A uniform layer of cement was placed on the crown’s inner surface with a spatula (Heidemann Spatula n.º 3 TD4173, Dental Market, Lucca, Italy). The crown was seated onto the die with finger pressure and then with a 5‑kg weight, equivalent to a 49‑N seating pressure, for 1 minute.19 During cementation, each surface was light‑cured for 1 second (Bluephase® Style 20i, Ivoclar Vivadent, Schaan, Liechtenstein), and then the excess cement was removed with a straight explorer (Explorers No. 6 TD4006, Dental Market, Lucca, Italy). Each surface was light‑cured again for 20 seconds each, with the device directed to the interface between the crown and the die to improve marginal integrity. In the end, all excesses were cleaned up as necessary.
After cementation, the die and crown set was placed in a circumferential putty base (Turboflex Putty Soft Normal Set, R&S, France; Lot 286323; Val: 11/2020) with a 50‑mm diameter and 5‑mm thickness and fixed to the load frame base of the testing machine. The fracture resistance test was performed in a universal testing machine (Instron, model 4502, Instron Ltd, Bucks, England) with a 5000‑N load cell and a 6‑mm stainless steel spherical tip at a crosshead speed of 1 mm/minute.
The set was positioned so that the spherical tip was at the center of the crown’s occlusal surface. Then, vertical load was applied toward the crown until fracture occurred (Figure 4).
The maximum breaking load was recorded in Newtons (N). After fracture, the remaining crown fragments and cement remnants were removed from the die using a straight explorer and a 50‑μm aluminum oxide sandblast (Basic classic 2945‑2026, Renfert GmbH, Hilzingen, Germany). The whole process was repeated for each tested specimen.
Group 2 crowns were luted with resin‑modified glass ionomer cement (RMGI) (FujiCEM™ 2, GC Corporation, Tokyo, Japan; Lot 180804A; Val: 08/2020), according to the manufacturer’s instructions. The RMGI was hand‑mixed for 10 seconds until a homogeneous mixture was obtained. A uniform layer of cement was placed on the crown’s inner surface using the spatula. The seating protocol was the same as in group 1. Excess cement was removed with the straight explorer as soon as the cement achieved a rubbery consistency. The manufacturer’s recommended setting time of 4.5 minutes was applied. In the end, the remaining excesses were cleaned up as necessary.
The processes of placing the die and crown set on the putty base, carrying out the fracture resistance test, removing the remaining crown fragments and cement from the die, and repeating the procedure for all crowns were executed in the same way as for group 1. A single operator carried out the cementation of all crowns. The sample size (n=20) was selected based on the study of Sorrentino et al., who tested the fracture resistance of Cerasmart™ full‑contour crowns (predecessor of Cerasmart270™), using a sample of ten specimens per experimental group.20
The statistical power of the present study was 93%, with a significance level of 0.05. The data were analyzed using IBM SPSS Statistics, version 25.0 (SPSS Inc., Chicago, Illinois, USA). Since the Shapiro‑Wilk and Levene’s tests revealed normality and homogeneity (p>0.05), respectively, the data were analyzed with a parametric Student’s t‑test. A statistical significance of α=0.05 was considered.
Results
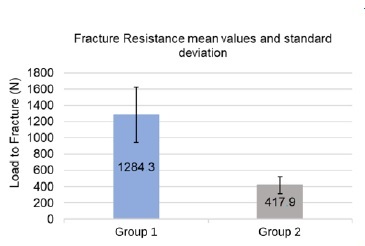
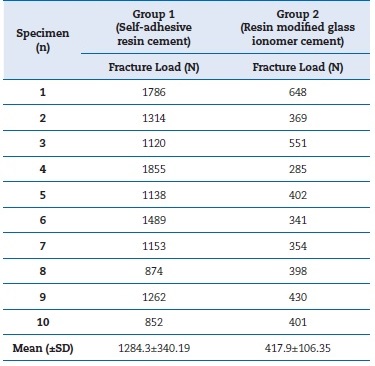
In group 1, the fracture resistance ranged from 852 N to 1855 N, with a mean value of 1284.3±340.19 N. In group 2, it ranged from 285 N to 648 N, with a mean value of 417.9±106.35 N (Table 1). These values are depicted in a graphic (Figure 5). Statistically significant differences were observed between the two groups (p<0.001), as group 1 registered a mean value higher than group 2, with a difference of 866.4 N, which translates into a 207.3% increase in the fracture resistance.
Table 1 Load at fracture in Newtons (N). Descriptive statistics by group: mean and standard deviation (SD).
All crowns failed and were not repairable. All fractures were confined to the crowns, and none progressed to the die. Group 1 crowns fractured at the crown‑die interface, with
crown fragments completely separating from the die. Group 2 crowns failed by chipping without separation of the crown material from the die.
Discussion
A higher mean value and greater standard deviation were obtained in group 1. Cementation with self‑adhesive resin cement showed significantly higher results of fracture resistance than cementation with RMGI. Thus, the null hypothesis was rejected. The mode of failure also differed between the two groups.
The mean values of fracture resistance were 1284.3±340.19 N for group 1 and 417.9±106.35 N for group 2. Ferrario et al. noted that the average maximum bite force in premolars was 206.01 N in women and 291.36 N in men.21 The present study obtained a minimum fracture resistance value of 285N, corresponding to a crown cemented with RMGI. Therefore, almost every crown withstood higher loads than the average bite forces in the premolar area.
Notwithstanding the lack of studies regarding the influence of the type of cement on the fracture resistance of crowns made from the resin nanoceramic featured in this study, it is possible to verify concordant results in similar papers.14,15 Lim and Lee’s study14 demonstrated that a self‑adhesive cement increased the fracture resistance of an indirect composite resin by 29%, compared to zinc phosphate. However, they registered mean values significantly lower than those of the presente study, presumably due to using rectangular blocks as specimens instead of crowns with adequate marginal adaptation.
Attia et al.15 demonstrated that full‑contour crowns milled from indirect composite resin CAD/CAM blocks and cemented with RMGI showed superior fracture resistance than those cemented with zinc phosphate. The mean values of fracture resistance reported were higher than those in the present study.
The differences in composition between the composite resin studied by Attia et al.22 and Cerasmart270™ affect the modulus of elasticity and flexural strength of these materials, which are properties that influence their fracture resistance. It should also be noted that Attia et al. used dentin as a substrate as opposed to a cobalt‑chrome alloy.
Wheyrauch et al.16analyzed the influence of the type of cement on the fracture resistance of full‑contour crowns milled from CAD/CAM blocks of various ceramics and a nano‑hybrid indirect composite resin. Several types of cement were used, and they found no correlation between the two variables.
Differences between the materials’ compositions may be responsible for the incompatibility of their results with those of the present study. Nakamura et al.23 also studied the influence of various types of cement on the fracture resistance of full‑contour zirconia crowns and found no significant diferences between groups. This result may be due to the high strength and hardness inherent to that material.24
There are limitations to this in vitro study. Instead of prepared natural teeth, a single cobalt‑chrome alloy die was used for the cementation of all 20 crowns tested. The sandblasting can be speculated to have caused dimensional alterations in the die, increasing the cement gap for the next cementations and, therefore, influencing the fracture resistance and reducing standardization. Considering a potential actual increase of cement gap, recent studies using CAD/CAM milled crowns from both conventional ceramics and resin nanoceramics showed that different cement thicknesses had no significant effect on fracture resistance.25,26 Furthermore, studies with similar methodology also had standard deviations similar to the ones of the present study.15,16,20,23 Thus, there is no clear evidence that the standardization was reduced.
Nevertheless, the sandblasting caused micro‑mechanical retentions on the die’s surface. In addition, the moduli of elasticity of cobalt‑chrome and dentin are different, which may have led to results different than expected. Thus, it would be pertinent to use dentin as a substrate and one die per cemented crown.
Another possible limitation is the use of putty to stabilize the crown and die set. Being a flexible material, it may have allowed some movement even though the crown and die set was firmly fixed to the test machine. However, the paper chart from the software of the testing machine, in all specimens, demonstrated a linear curve until fracture occurred. Therefore, there is no indication that the putty elastomer impacted the applied force or influenced the results obtained, but less flexible materials, such as gypsum or epoxy resin, could have been a more suitable alternative.
The crowns cemented with self‑adhesive resin cement were tested immediately after light‑curing. Thus, there was no post‑curing time, which is described as increasing resin cement’s properties, namely surface hardness.27,28,29
Another limitation of this study is the lack of cyclic and/or thermomechanical load since restorations in the clinical environment fracture mainly due to fatigue.30,31,32,33 Given the aforementioned limitations, the direct transfer of results to in vivo conditions is limited.
For this study, a knife‑edge margin was selected. Available literature relates this preparation design to crowns with lower resistance values but still higher than the occlusal loads described.34 Although the type of margin was not one of the variables studied in this study, it is also expected to affect the evaluated material. Therefore, studies focused on the relationship between margin design and fracture resistance are needed. Moreover, future studies should replicate the conditions of the oral environment by applying cyclic and/or thermodynamic loads to mimic the fatigue suffered by restorations.