Introduction
Disorders of the menstrual cycle are a common problem in ambulatory medicine and ovarian cysts are a common disorder in young women associated with a multitude of etiologies1. Ovarian hyperstimulation induced by inappropriate follicle-stimulating hormone (FSH) oversecretion from pituitary adenoma may manifest as menstrual irregularities and multiple ovarian cysts. The true incidence of this condition remains unclear, but in the literature, around 30 cases are described2. Pituitary adenomas can be classified as functioning or non-functioning depending on hormonal secretion. We report an unusual case of a woman that presented at the gynecology department with menstrual disturbances with five years duration.
Case report
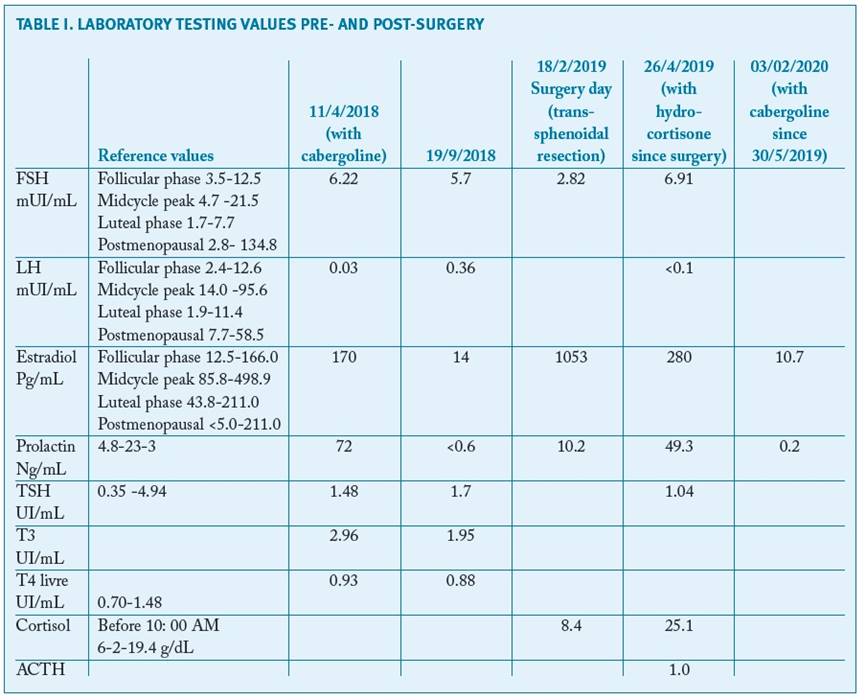
A 29-year-old nulliparous woman presented to the gynecology department in August 2017, with menstrual disturbances and pelvic pain, irrespective of usage of contraceptive pills for the past five years. On the gynecological exam she had galactorrhea with no palpable masses. The transvaginal ultrasound revealed enlarged ovaries, measuring 93x78 mm(left) and 52x30 mm (right), with multiple cysts with regular walls, featuring like an ovarian hyperstimulation syndrome. No ascites or endometrial thickness were detected (Fig. 1).
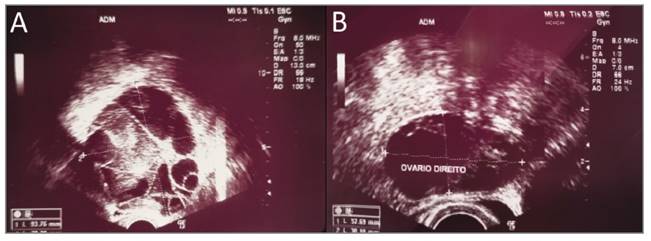
FIGURE 1 Transvaginal ultrasonography showing bilateral enlarged ovaries - (A) left ovary and (B) right ovary
An initial endocrine evaluation showed: estradiol level of 170 pg/mL (slightly elevated), prolactin level (PRL) of 72 ng/mL (mildly elevated), luteinizing hormone (LH) level of 0.03 mIU/mL (low level), serum FSH of 6.22 mIU/mL (normal), thyroid-stimulating hormone (TSH) level of 1.48 UI/mL (normal) and thyroid hormones such as triiodothyronine (T3) and thyroxine (T4) levels were normal (2.96 and 0.93 UI/mL respectively). The pregnancy test was negative. Tumoral markers were negative (CA 125; CA 15.3 and CA 19.9). The pelvic magnetic resonance imaging (MRI) confirmed the previous ultrasound findings (Fig. 2).
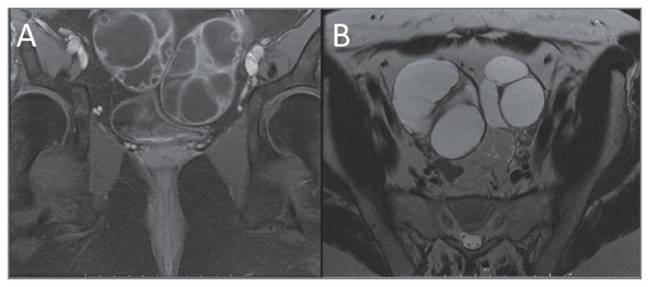
FIGURE 2 Pelvic MRI revealing enlargement of the ovaries (A) Axial T1 FatSat with gadolinium (B) Coronal T2
Pituitary MRI revealed a pituitary macroadenoma, 29x31x24 mm, with enlargement and apparent lateral deviation of the cavernous segment pathway of the internal carotid artery (Fig. 3).
FIGURE 3 MRI of the Sella Turcica: (A) T1-shaped coronal and (B) sagittal, planes with Gadolinium reveal a large expansive capturing lesion in the sella, suprasellar, and bilateral paraselar (38 x 27.3 x 23.3mm of larger transverse x vertical x anteroposterior diameters) compatible with pituitary macroadenoma.
After the diagnosis of the macroadenoma, campimetry revealed left eye hemianopsia and right eye scotoma.
The patient started cabergoline 0.5 mg once a week to control the level of prolactin. Hormonal study after cabergoline revealed low PRL level (< 0.6 ng/mL) and normal levels of estradiol (14pg/mL), LH (0.36 mIU/mL), FSH (5.7mIU/mL) and TSH (1.04 UI/mL).
Endoscopic transsphenoidal surgery was performed and the pituitary macroadenoma was partially removed with uneventful hospitalization. The patient was discharged home four days after the surgery with hydrocortisone (10 mg/ day). Postoperatively, the situation remained stable with occasional headache and minimal visual symptoms.
Histological examination showed a tumor with solid and nested patterns composed of small to medium--sized polygonal cells, without significant atypia, with clear and eosinophilic cytoplasm (Figures 4A and B). The perivascular papillary arrangement was also present. There was neither mitotic figures nor areas of necrosis. The tumor cells were immunoreactive for synaptophysin, CAM 5.2, focal for FSH (Figure 4C), and LH (Figure 4D) with no expression of growth hormone (GH), ACTH, TSH, and prolactin. The Ki-67 proliferation index was 2%.
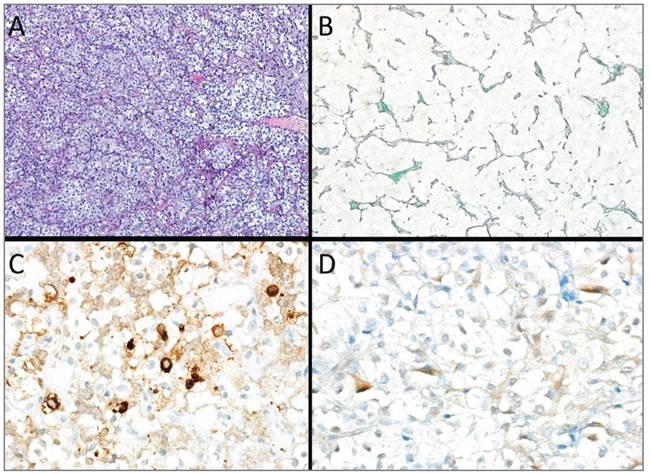
FIGURE 4 Histology of the pituitary tumor. (A) HE, 100x; (B) Reticulin, 200x; (C) FSH 400x; (D) LH 400x
Two months after surgery, the patient menstrual cycles became regular and her vision returned to normal. PRL was slightly elevated (49.3 ng/ml), FSH was normal and LH was suppressed (6.91 mU/ml and < 0.1 mU/ml, respectively). Cortisol was slightly elevated (25 g/dL), but adrenocorticotropic hormone (ACTH) remained normal. Because prolactin still slightly elevated, the hydrocortisone was tapered down, and the patient was medicated with cabergoline (Table I). The transvaginal ultrasound showed a significant decrease in ovary dimensions.
The patient is checked regularly in a neurosurgery department and is symptom-free at the present moment.
Discussion
The management of menstrual irregularities at reproductive age is often challenging. In our case, a transvaginal ultrasound scan revealed an enlargement of both ovaries. Blood tests showed a slightly elevated level of estradiol and PRL and a suppressed LH. The patient seemed to have ovarian hyperstimulation3.
Serum estradiol was elevated in response to FSH secreted in the adenoma cells. Subsequently, the hypothalamus-anterior pituitary gland axis was suppressed due to the negative feedback. As a result, the excessive FSH level was reduced to a normal range. Serum LH was also significantly reduced, probably due to the negative feedback or compression of the normal pituitary gland by the tumor4. Apparent normal FSH, suppressed LH and normal-high estradiol levels can be considered the characteristic endocrinological profile of this FSH-secreting pituitary adenoma5.
The administration of clomiphene citrate or exogenous gonadotropins can potentially simulate such a clinical scenario, but it is usually associated with a systemic increase of vascular permeability, manifesting by an extravasation of fluid into the peritoneal cavity or third space, which does not occur in ovarian hyperstimulation induced by pituitary adenomas1,6.
The Polycystic ovary syndrome (PCOS) also presents with menstrual irregularities and polycystic ovaries. Cysts are smaller and peripherally arrayed. In contrast, in pituitary adenoma, ovaries are markedly enlarged and contain larger cysts with scarce stroma. Furthermore, elevated LH with normal FSH levels are commonly found in PCOS, while suppressed LH, and mildly/high FSH are reported in pituitary adenoma7. Ovarian hyperstimulation has also been in association with primary hypothyroidism, possibly caused by TSH-mediated stimulation of the FSH receptor or by enhanced thyrotropin-releasing hormone (TRH) production stimulating gonadotropin release. This aspect was not apparent in our case8.
In our patient, due to endocrinological findings, a pituitary MRI was performed, and a macroadenoma was detected. The patient was submitted to neurosurgery for removing the mass. The final histological results confirmed the suspicion of a pituitary adenoma with FSH and LH expression.
In rare cases, these adenomas can cause clinical symptoms with variable clinical severity because the biological activity of FSH is not always uniform2,9-11. More commonly, they present with neurological symptoms secondary to mass effects or are incidentally discovered. Visual field defect is a common presentation, and so the study of visual abnormalities is paramount. Our patient initially had no visual complaints, which made the diagnosis more challenging.
Medical treatment of gonadotroph adenomas with bromocriptine or cabergoline improves hormonal status and decreases ovarian volume with resumed ovulatory cycles but does not decrease pituitary tumor size12-15. Accordingly, medical treatment was the first strategy to improve hormonal levels in this patient. Surgery should lead to the resolution of the clinical symptoms and normalization of the biochemical changes. In this case, the resection was partial and consequentially, the patient maintained cabergoline to control tumor size. Patients should also undergo hormonal testing 2-3 months after surgery to assess whether hypopituitarism is present. If it is, hormonal replacement therapy should be initiated. There are limited data on long-term outcomes in patients with functioning gonadotroph adenomas, so long-term follow-up is required due to the risk of recurrence9. For patients that have residual tumor after surgery (as in this case) there is no consensus on the best approach. To date, there is no reliable marker to predict tumor regrowth after surgery. This patient maintained cabergoline to control tumor size. MRI should be performed periodically ( initially 6 months in the first year and annually afterward) to indicate whether there was stabilization/reduction of the lesion (which would justify the maintenance of medication) or tumor progression, wich justify surgery or radiotherapy. If the patient develops visual impairment or the tumor begins to compress visual pathways, further surgery is indicated. Symptoms like menstrual irregularities should be inquired at each appointment, and endocrine evaluation with estradiol, prolactin, LH and FSH level should be performed every 6-12 months. Annual visual field perimetry should be performed in patients with tumors with suprasellar extension16.
Functioning gonadotroph adenomas should be considered in women presenting with ovarian hyperstimulation, as surgical treatment of the tumor can result in the resolution of symptoms and avoid the need for either cystectomy or oophorectomy. Women with ovarian hyperstimulation are likely to be referred to gynecology; therefore, it is crucial gynecologists be aware of this association.