Services on Demand
Journal
Article
Indicators
Related links
Share
GE-Portuguese Journal of Gastroenterology
Print version ISSN 2341-4545
GE Port J Gastroenterol vol.25 no.6 Lisboa Dec. 2018
https://doi.org/10.1159/000485803
ORIGINAL ARTICLE
High Efficacy of Repeating Colonoscopy by an Advanced Endoscopist after an Incomplete Colonoscopy
Elevada eficácia da repetição de uma colonoscopia incompleta por um endoscopista avançado
Ana Ponte, Rolando Pinho, Adélia Rodrigues, Luísa Proença, Joana Silva, Jaime P. Rodrigues, Mafalda Sousa, João Carlos Silva, João Carvalho
Department of Gastroenterology, Centro Hospitalar Vila Nova de Gaia/Espinho, Vila Nova de Gaia, Portugal
* Corresponding author.
ABSTRACT
Background and Aims: This study aims to evaluate the role of an advanced endoscopist to study the entire colon after an incomplete colonoscopy. Methods: All patients with an elective incomplete colonoscopy performed under deep sedation in our department between January 2010 and October 2016 were included. Patients with a colonic stenosis, an inadequate bowel preparation, or a colonoscopy performed without deep sedation were excluded. Included patients were followed up to evaluate if and what type of subsequent examinations (colonoscopy by an advanced endoscopist, single-balloon enteroscopy [SBE], and/or CT colonography) was performed to complete the study of the entire colon. Lesions found during these subsequent examinations were also recorded. Results: Ninety-three patients had an incomplete colonoscopy, with no diagnosis of colorectal cancer (CRC) and a high-risk polyp rate of 5.4% (n = 5). Seventy-seven patients with incomplete colonoscopies underwent subsequent examinations, namely CT colonography in 45.5% (n = 35), colonoscopy by an advanced endoscopist in 53.2% (n = 41), and SBE in 13% (n = 10). In the 49 patients who performed either colonoscopy (n = 39) or SBE (n = 10) by an advanced endoscopist, the cecal intubation rate was 100%, and high-risk polyps were found in 26.5% (n = 13) and CRC in 4.1%. CT colonography revealed findings consistent with polyps and CRC in 22.9% (n = 8) and 2.9% (n = 1) of the cases, respectively. Colonoscopy was further repeated in 6 patients with suspected polyps in CT colonography, confirming the initial diagnosis in 5 patients. Conclusions: Colonoscopy by an advanced endoscopist achieved cecal intubation in all patients, representing a good choice after an incomplete colonoscopy.
Keywords: Colonoscopy, Computed tomography colonography, Single-balloon enteroscopy, Colorectal cancer
RESUMO
Introdução/Objetivo: O presente estudo pretende avaliar o papel de um endoscopista avançado no estudo do cólon após colonoscopia incompleta (CI). Material: Incluíram-se todos os doentes com CI eletiva realizada por especialista, sob sedação profunda entre janeiro de 2010 e outubro de 2016. Excluíram-se doentes com estenose cólica, colonoscopia com preparação inadequada e/ou sem sedação. Avaliaram-se os exames subsequentes (colonoscopia por endoscopista avançado, enteroscopia assistida por monobalão [EAB] e colonografia virtual [CV]) realizados para completar o estudo do cólon e as lesões diagnosticadas. Resultados: Incluíram-se 93 CI, que detetaram pólipos de alto risco em 5.4% (n = 5) e não identificaram carcinoma colorretal (CCR). Realizou-se um segundo exame em 82.8% (n = 77) dos doentes, designadamente, a CV, colonoscopia por endoscopista avançado e EAB em 45.5% (n = 35), 53.2% (n = 41) e 13% (n = 10), respetivamente. Nos 49 doentes que realizaram colonoscopia (n = 39) ou SBE (n = 10) por endoscopista avançado, a taxa de entubação cecal foi de 100%, detetando-se pólipos de alto risco em 26.5% (n = 13) e CCR em 4.1%. Na CV observaram-se achados compatíveis com pólipos em 22.9% (n = 8) e CCR em 2.9% (n = 1) dos casos. A colonoscopia foi subsequentemente realizada em 6 doentes com suspeita de pólipos na CV, confirmando-se o diagnóstico em 5 doentes. Conclusão: A entubação cecal foi conseguida em todos os doentes submetidos a colonoscopia realizada por endoscopista avançado representando uma boa opção no estudo subsequente do cólon após uma CI.
Palavras-Chave: Colonoscopia, Colonografia virtual, Enteroscopia assistida por monobalão, Cancro colorretal
Introduction
Colorectal cancer (CRC) is the second most common cause of cancer-related death in the Western world [1]. Screening for CRC in asymptomatic patients can reduce the incidence and mortality of CRC by allowing the detection of premalignant lesions or CRC in early stages and the risk stratification according to the endoscopic findings leading to different endoscopic surveillance intervals [2–4]. Colonoscopy is considered a first-line screening exam for CRC prevention [4–7]. Quality of screening colonoscopy depends on some indicators, namely cecal intubation, as 30–50% of advanced neoplasias are located in the proximal colon [2, 8]. A standard approach after an incomplete colonoscopy is lacking and several exams to complement the visualization of the entire colon have been described including double-contrast barium enema, computed tomography colonography (CTC), single-balloon enteroscopy (SBE), double-balloon enteroscopy (DBE), and colon capsule endoscopy [9, 10]. Nonetheless, a second colonoscopy may be attempted with the adoption of different maneuvers and in the hands of an advanced endoscopist [9].
This study aims to evaluate the role of an advanced endoscopist to study the entire colon after an incomplete colonoscopy.
Materials and Methods
A retrospective cohort study involving all consecutive patients with an elective incomplete colonoscopy in our department between January 2010 and October 2016 was performed. Incomplete colonoscopy was defined as a colonoscopy without cecal intubation. Patients with a colonic stenosis, an inadequate bowel preparation, or a colonoscopy performed without deep sedation were excluded. The index colonoscopy was performed under deep propofol sedation administered by an anesthesiologist. Moreover, the procedure was performed by an endoscopist with or without a trainee.
Patients with an incomplete colonoscopy were followed up to evaluate if and what type of secondary examinations were performed to visualize the remaining colon not reached at the index colonoscopy. Secondary examinations included CTC, colonoscopy by an advanced endoscopist, and SBE, which are the procedures currently available in our center. An advanced endoscopist was defined as a gastroenterologist with expertise in advanced therapeutic endoscopy. Patients with an inadequate preparation in second colonoscopy or SBE were excluded. All patients provided informed consent for all investigations performed.
Data regarding demography, past medical and surgical history, indication for the index colonoscopy, depth of intubation at the index colonoscopy, reason for an incomplete examination, findings at the index colonoscopy, secondary investigations, and findings in the secondary investigations in the nonvisualized part of the colon at the index colonoscopy were recorded. Indication for the index colonoscopy was categorized in surveillance (inflammatory bowel disease surveillance, previous cancer or polyp follow-up, family history screening) and symptoms (iron deficiency anaemia, abdominal mass, abdominal pain, change in bowel habits, inflammatory bowel disease assessment, diarrhoea, rectal bleeding, and abnormal CTC, barium enema, or flexible sigmoidoscopy) [8].
Recorded findings consisted of diverticulosis, polyps, and CRC, possibly leading to more than one simultaneous finding for each exam. Polyps were further categorized as low risk if 1–2 tubular adenomas <10 mm with low-grade dysplasia; serrated polyps <10 mm and no dysplasia, and high risk if adenoma with villous histology or high-grade dysplasia or ≥10 mm in size, or ≥3 adenomas; serrated polyps ≥10 mm or with dysplasia [3]. In cases where more than one polyp was observed, the most advanced lesion was included in the analysis. Moreover, if a lesion was already diagnosed at the index colonoscopy, it was excluded from the findings of a second examination.
A descriptive statistics was performed with continuous variables given as mean and standard deviation and categorical variables expressed as proportions. The Statistical Package for Social Sciences version 20.0 (IBM Corp., Armonk, NY, USA) was used for data entry and data analysis.
Results
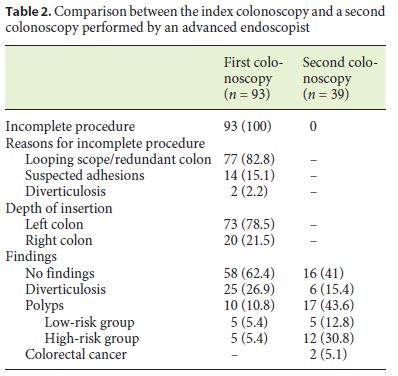
Between January 2010 and October 2016, 93 patients out of a total of 6,196 exams had an incomplete colonoscopy; 67.7% were women with a mean age of 70.6 years (Table 1). The rate of incomplete exams was 1.5%. Colonoscopy was performed for surveillance in 52.7% of patients and for diagnostic purposes in 47.3% (Table 1). Main reasons for an incomplete colonoscopy included looping of the scope and redundant colon (82.8%) and suspected adhesions (15.1%) (Table 2). Moreover, the majority of the exams were ceased in the left colon (78.5%). Most incomplete colonoscopies had no findings (62.4%) and no CRC was identified. Diverticulosis and polyps were observed in 26.9 and 10.8%, respectively (Table 2).
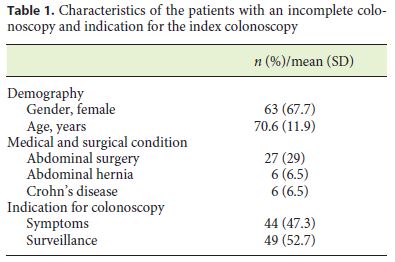
A second examination was performed in 82.8% (n = 77) of the patients, with a mean time between the two exams of 6.73 ± 11.88 months. Additionally, in 11.7% (n = 9) patients, a third examination was further performed.
CTC, colonoscopy by an advanced endoscopist, and SBE were performed in 45.5% (n = 35), 53.2% (n = 41), and 13% (n = 10) of patients, respectively (Table 3). CTC was normal in 68.6% (n = 24) of the cases and findings consistent with polyps and CRC were identified in 22.9% (n = 8) and 2.9% (n = 1) of the exams, respectively (Table 3). Colonoscopy was further repeated in 6 patients with suspected polyps in CTC, confirming the initial diagnosis in 5 patients. SBE was also further performed in 3 patients with diverticulosis and suspected polyps in CTC, confirming the initial diagnosis of polyps in 1 patient. The suspected CRC was confirmed by surgery.
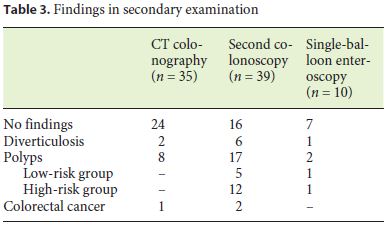
The second colonoscopy was incomplete in 4.9% (n = 2) of the cases due to poor bowel preparation, with stools preventing the progression of the colonoscope and the completeness of the exam. Considering only colonoscopies whose preparation did not limit the progression of the colonoscope, the cecal intubation rate of the second colonoscopy was 100%. This second colonoscopy performed by an advanced endoscopist revealed more significant lesions when compared to the index colonoscopy, namely high-risk polyps (n = 12; 30.8%) and CRC (n = 2; 5.1%) (Table 2). The adenoma detection rate was 32.4%.
SBE was performed in 13% (n = 10) of the patients and was able to reach the cecum in all cases. Polyps were identified in 20% (n = 2) of the procedures, half of them categorized as high risk (Table 3).
In the 49 patients who performed either colonoscopy (n = 39) or SBE (n = 10), the cecal intubation rate was 100%, and high-risk polyps were found in 26.5% (n = 13) and CRC in 4.1% (n = 2).
In patients with an initial incomplete colonoscopy (n = 93), no CRC was diagnosed and high-risk polyps were diagnosed in 5.4% (n = 5). In patients submitted to a subsequent examination (n = 77), CRC was diagnosed in 3.9% (n = 3) and high-risk polyps were diagnosed and removed in 16.9% (n = 13).
Discussion
The cecal intubation rate is a quality indicator for colonoscopy because it is associated with an increased detection rate of advanced neoplasia, as 30–50% of advanced neoplasias are located in the proximal colon [1, 2, 7, 8, 11]. Quality guidelines currently recommend a cecal intubation rate ≥90% in all colonoscopies in routine clinical practice and ≥95% in screening colonoscopies [2, 7, 9–14]. Nevertheless, reported rates of cecal intubation are suboptimal ranging from 76.9 to 89.1% in clinical practice and from 91 to 97.7% in screening colonoscopy [1, 7, 12]. Various patient-related factors may contribute to an incomplete colonoscopy, including colonic redundancy or tortuosity, low body mass index, female sex, prior abdominal or pelvic surgeries resulting in adhesions, severe diverticular disease, colonic stenosis, poor bowel preparation, and patient intolerance [2, 6, 9, 10, 12, 15–17]. Some technical-related factors may also contribute to incomplete colonoscopy, such as the use of sedation, the type of colonoscope, and the skill of the endoscopist.
Recommendations regarding the subsequent approach after an incomplete colonoscopy are lacking and malignant and premalignant lesions may be missed if further investigation is not pursued [1, 8, 9]. In our study, 17.2% of patients had no documented recommendation for follow-up to complete colonic evaluation, similar to other studies reporting a rate of 19% [9].
Several techniques have been described to allow cecal intubation in difficult colonoscopies, namely minimizing air insufflation, use of carbon dioxide instead of air for insufflation, abdominal pressure to prevent looping of the colonoscope, changing patient positioning, use of more flexible endoscopes or variable stiffness endoscopes, water immersion, or fluoroscopy [2, 9, 18]. In cases of an incomplete colonoscopy despite the adoption of the described methods, alternative endoscopic approaches as SBE or DBE and alternative nontherapeutic imaging or endoscopic approaches as double-contrast barium enema, CTC, magnetic resonance colonography, and colon capsule have been described [2, 7, 9, 11, 12, 15].
Double-contrast barium enema has a low sensitivity (48%) for polyps ≥10 mm and CTC has been proven to be substantially more effective with a sensitivity of 90% for polyps ≥10 mm and a positive predictive value of >90% for clinically important neoplasia [1, 2, 6–8, 10]. Moreover, it provides extracolonic examination [6]. In our study, CTC revealed polyps in 22.9% (n = 8) patients, which were further confirmed by a subsequent endoscopic method in 6 patients. Previous reports of CTC after an incomplete colonoscopy revealed lower rates of polyp detection ranging from 11 to 13.2% [2, 6]. Main disadvantages of CTC include need for bowel preparation with the addition of contrast agents for stool and fluid tagging, radiation exposure, inability for tissue sampling or therapeutic techniques, and relatively low sensitivity for the detection of polyps ≤5 mm and flat lesions [2, 10]. Moreover, a recent study concluded that the detection rate of high-risk sessile serrated polyps was significantly higher with colonoscopy than CTC [19].
Colonoscopy performed by an advanced endoscopist was the subsequent examination more frequently adopted after an incomplete colonoscopy. Cecal intubation was achieved in all patients after excluding two patients with inadequate bowel preparation. This is in line with the quality indicators for nonselected screening colonoscopies and with previous reports of performance of a second colonoscopy by an experienced endoscopist after failure of a standard colonoscopy, which have published cecal intubation rates ranging from 96 to 99% [2, 11]. Moreover, in our study, the reasons for inability to reach the cecum resulted from poor bowel preparation. The expertise of the endoscopist appears to be the most important factor that determines success at colonoscopy and it avoids the need for subsequent examinations for diagnostic and therapeutic approaches [2, 11, 20]. Moreover, bleeding and perforation are the most feared complications in colonoscopy and the reported factors for an incomplete colonoscopy are known risk factors for perforation [21]. Current guidelines recommend a ratio lower than 1: 1,000 of diagnostic or therapeutic colonoscopies which complicate with a perforation requiring surgical repair. In our study, no complications were reported in the second colonoscopy [22].
Deep enteroscopy was originally developed to facilitate deep insertion of the endoscope into the small-bowel. SBE and DBE were then adapted to difficult colonoscopy, mainly in cases of redundant colons, severe diverticulosis, and sharp angulations due to surgical adhesions [2, 23]. Cecal intubation rates for DBE and SBE in cases of incomplete colonoscopies are reported to be 88–100% and 93–100%, respectively [2, 10, 15, 16, 24, 25]. Studies comparing DBE and SBE showed no significant differences in cecal intubation rates, mean cecal intubation times, and diagnostic and therapeutic rates [2, 16]. In our study, SBE resulted in a successful cecal intubation in all cases, and polyps were identified in 20% of the procedures. Previous studies, report a similar polyp detection rate in SBE of 27.3% [26].
The retrospective nature of the study and the small sample size of each group constitute limitations of our study. Moreover, the completeness of the second procedure may sometimes be related to other factors independent from the skill of the endoscopist, such as bowel changes over time or better bowel preparation.
In conclusion, in our study, a subsequent examination after an incomplete colonoscopy led to the diagnosis of CRC in 3.9% of cases and high-risk polyps in 16.9%. This addresses the importance of subsequent examinations after an incomplete colonoscopy. Taking into account the results of our study, all 3 modalities for subsequent examinations available in our center are quite effective in the completeness of the study of the colon. Second colonoscopy performed by an advanced endoscopist achieved cecal intubation in all patients in our study. Moreover, it does not require additional equipment, which may not be widely available in every center, as CTC or SBE. Finally, it avoids unnecessary exams since, after a significant finding in CTC, an endoscopic procedure should always be performed to confirm the diagnostic findings and perform therapy.
References
1 Neerincx M, Terhaar sive Droste JS, Mulder CJ, et al: Colonic work-up after incomplete colonoscopy: significant new findings during follow-up. Endoscopy 2010;42:730–735. [ Links ]
2 Villa NA, Pannala R, Pasha SF, et al: Alternatives to incomplete colonoscopy. Curr Gastroenterol Rep 2015;17:43. [ Links ]
3 Hassan C, Quintero E, Dumonceau JM, et al: Post-polypectomy colonoscopy surveillance: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2013;45:842–851. [ Links ]
4 Lieberman DA, Rex DK, Winawer SJ, et al: Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology 2012;143:844–857. [ Links ]
5 Rex DK, Boland CR, Dominitz JA, et al: Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc 2017;86:18–33. [ Links ]
6 Pullens HJ, van Leeuwen MS, Laheij RJ, et al: CT-colonography after incomplete colonoscopy: what is the diagnostic yield? Dis Colon Rectum 2013;56:593–599. [ Links ]
7 Spada C, Hassan C, Barbaro B, et al: Colon capsule versus CT colonography in patients with incomplete colonoscopy: a prospective, comparative trial. Gut 2015;64:272–281. [ Links ]
8 Britton EJ, Sidhu S, Geraghty J, et al: The 5-year outcome of patients having incomplete colonoscopy. Colorectal Dis 2015;17:298–303. [ Links ]
9 Gawron AJ, Veerappan A, McCarthy ST, et al: Impact of an incomplete colonoscopy referral program on recommendations after incomplete colonoscopy. Dig Dis Sci 2013;58:1849–1855. [ Links ]
10 Keswani RN: Single-balloon colonoscopy versus repeat standard colonoscopy for previous incomplete colonoscopy: a randomized, controlled trial. Gastrointest Endosc 2011;73:507–512. [ Links ]
11 Bick BL, Vemulapalli KC, Rex DK: Regional center for complex colonoscopy: yield of neoplasia in patients with prior incomplete colonoscopy. Gastrointest Endosc 2016;83:1239–1244. [ Links ]
12 Morini S, Zullo A, Hassan C, et al: Endoscopic management of failed colonoscopy in clinical practice: to change endoscopist, instrument, or both? Int J Colorectal Dis 2011;26:103–108. [ Links ]
13 Rex DK, Schoenfeld PS, Cohen J, et al: Quality indicators for colonoscopy. Gastrointest Endosc 2015;81:31–53. [ Links ]
14 Kaminski MF, Thomas-Gibson S, Bugajski M, et al: Performance measures for lower gastrointestinal endoscopy: a European Society of Gastrointestinal Endoscopy (ESGE) Quality Improvement Initiative. Endoscopy 2017;49:378–397. [ Links ]
15 Hotta K, Katsuki S, Ohata K, et al: Efficacy and safety of endoscopic interventions using the short double-balloon endoscope in patients after incomplete colonoscopy. Dig Endosc 2015;27:95–98. [ Links ]
16 Hotta K, Katsuki S, Ohata K, et al: A multicenter, prospective trial of total colonoscopy using a short double-balloon endoscope in patients with previous incomplete colonoscopy. Gastrointest Endosc 2012;75:813–818. [ Links ]
17 Jia H, Wang L, Luo H, et al: Difficult colonoscopy score identifies the difficult patients undergoing unsedated colonoscopy. BMC Gastroenterol 2015;15:46. [ Links ]
18 Vemulapalli KC, Rex DK: Water immersion simplifies cecal intubation in patients with redundant colons and previous incomplete colonoscopies. Gastrointest Endosc 2012;76:812–817. [ Links ]
19 Je IJ, Tutein Nolthenius CJ, Kuipers EJ, et al: CT-colonography vs. colonoscopy for detection of high-risk sessile serrated polyps. Am J Gastroenterol 2016;111:516–522. [ Links ]
20 Gawron AJ, Veerappan A, Keswani RN: High success rate of repeat colonoscopy with standard endoscopes in patients referred for prior incomplete colonoscopy. BMC Gastroenterol 2014;14:56. [ Links ]
21 Levy I, Gralnek IM: Complications of diagnostic colonoscopy, upper endoscopy, and enteroscopy. Best Pract Res Clin Gastroenterol 2016;30:705–718. [ Links ]
22 Rembacken B, Hassan C, Riemann JF, et al: Quality in screening colonoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE). Endoscopy 2012;44:957–968. [ Links ]
23 Nemoto D, Isohata N, Utano K, et al: Doubleballoon colonoscopy carried out by a trainee after incomplete conventional colonoscopy. Dig Endosc 2014;26:392–395. [ Links ]
24 Kobayashi K, Mukae M, Ogawa T, et al: Clinical usefulness of single-balloon endoscopy in patients with previously incomplete colonoscopy. World J Gastrointest Endosc 2013;5:117–121. [ Links ]
25 Sulz MC, Frei R, Semadeni GM, et al: The role of single-balloon colonoscopy for patients with previous incomplete standard colonoscopy: is it worth doing it? Surg Endosc 2016;30:1876–1882. [ Links ]
26 Teshima CW, Aktas H, Haringsma J, et al: Single-balloon-assisted colonoscopy in patients with previously failed colonoscopy. Gastrointest Endosc 2010;71:1319–1323. [ Links ]
Statement of Ethics
This study did not require informed consent or review/approval by the appropriate ethics committee.
Disclosure Statement
The authors declare no conflict of interest for this article. The authors declare no source of funding for this article.
* Corresponding author.
Dr. Ana Ponte
Department of Gastroenterology, Centro Hospitalar Vila Nova de Gaia/Espinho
Rua Conceição Fernandes
PT–4434-502 Vila Nova de Gaia (Portugal)
E-Mail ana.ilponte@gmail.com
Received: September 4, 2017; Accepted after revision: November 28, 2017














