Introduction
Congenital cutaneous candidiasis (CCC) is a rare condition in neonates. It is the result of a Candida spp. infection acquired in utero and is thought to occur via an ascending pathway from candida vulvovaginitis1. One of the most accepted risk factors is the presence of an intrauterine foreign body (intrauterine device or cervical cerclage)2. CCC can occur in infants regardless of the type of delivery, duration of rupture of membranes, or maternal diagnosis of candida vaginosis3. It is more common in preterm neonates under 27 weeks of gestational age, as well as in those with extremely low birth weight4. Immature, compromised mucocutaneous barriers and poor systemic host defences are thought to be predisposing factors in preterms. Extensive CCC in term neonates is rare, with only a few published reports in the literature.
Clinical case
A full-term (38 weeks) female infant was born by vaginal delivery with an appropriate birth weight for the gestational age (3044 gm). She was born to a healthy mother with a positive history of vaginal candidiasis before pregnancy. Serologies and prenatal ultrasounds were normal. There was no maternal history of herpes simplex infection, intrauterine device, cervical cerclage or amniocentesis. The mother tested positive for group B Streptococcus and received adequate antibiotic prophylaxis before delivery.
The Apgar score was 9/10/10 at 1, 5 and 10 minutes at birth. She had a regular clinical examination at birth.
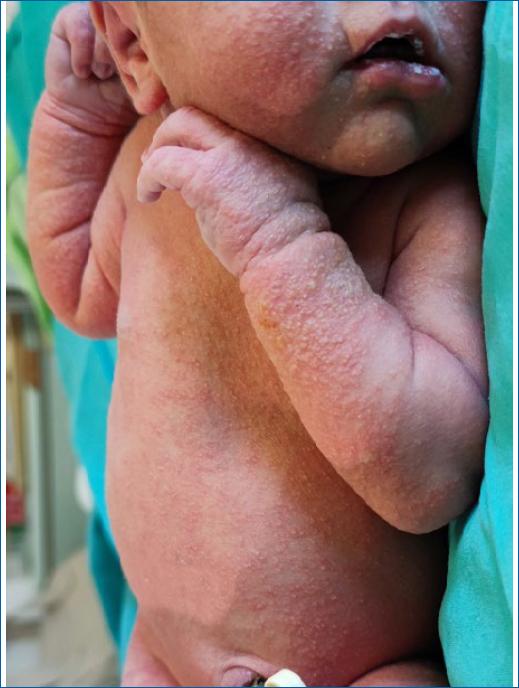
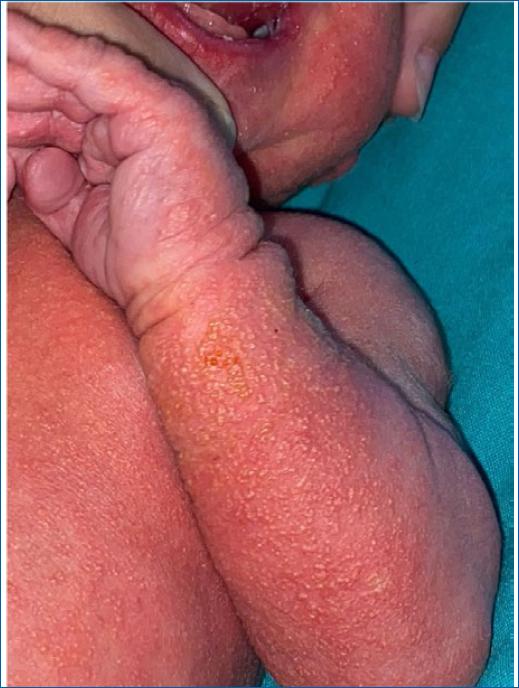
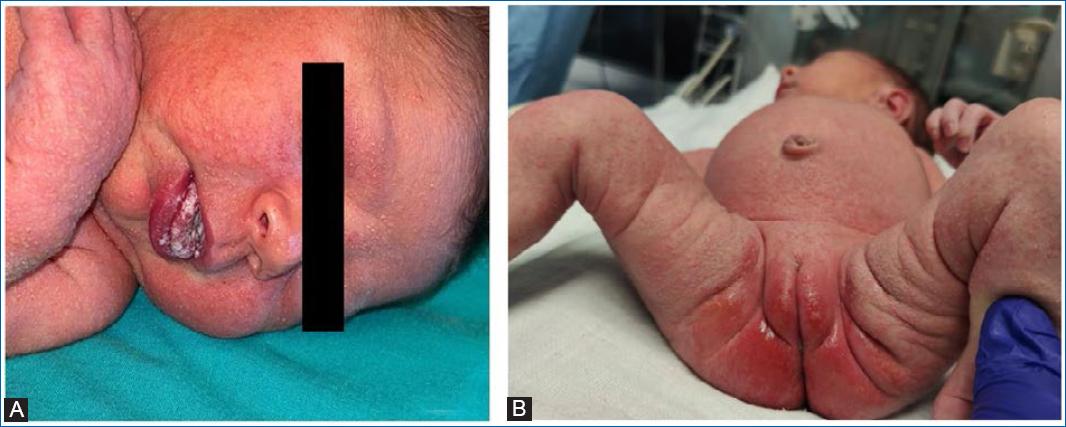
At 24 hours of life, a few papules were noticed on the head and trunk, which rapidly and progressively worsened over the next 48 hours. On the third day, she developed a generalised papulopustular eruption involving the face, neck, trunk and extremities, including palms and soles, as well as diffuse bright erythema with erosions in the perineal area, multiple white plaques on the oral mucosa (Figs. 1 to 3), and purulent ocular exudate. There was no fever or other systemic symptoms. Based on clinical findings and skin examination, the diagnosis of CCC was strongly considered. Laboratory tests showed no evidence of systemic infection and a complete blood count revealed 18100/μL white blood cells (9500/uL neutrophils) and negative serum C-reactive protein concentration (0.03 mg/dL). Skin, ocular and blood cultures were collected. After multidisciplinary discussion, taking into consideration the extensive cutaneous and mucous membranes involvement, in the absence of systemic signs of infection, empiric treatment with intravenous fluconazole (12 mg/kg/day), oral nystatin and clotrimazole cream was started. Cerebrospinal fluid was not collected due to the lack of lesion-free areas at the puncture sites. Considering CCC as the most likely diagnosis and the absence of clinical or laboratory evidence of systemic infection, no antibacterial nor antiviral therapy was added, pending clinical evolution and culture results. After a few days, the culture of pustules and ocular exudate identified Candida albicans confirming the diagnosis. Blood cultures were negative. The mother's vaginal evaluation did not show any signs of active vulvovaginal candidiasis and vaginal swab cultures were negative.
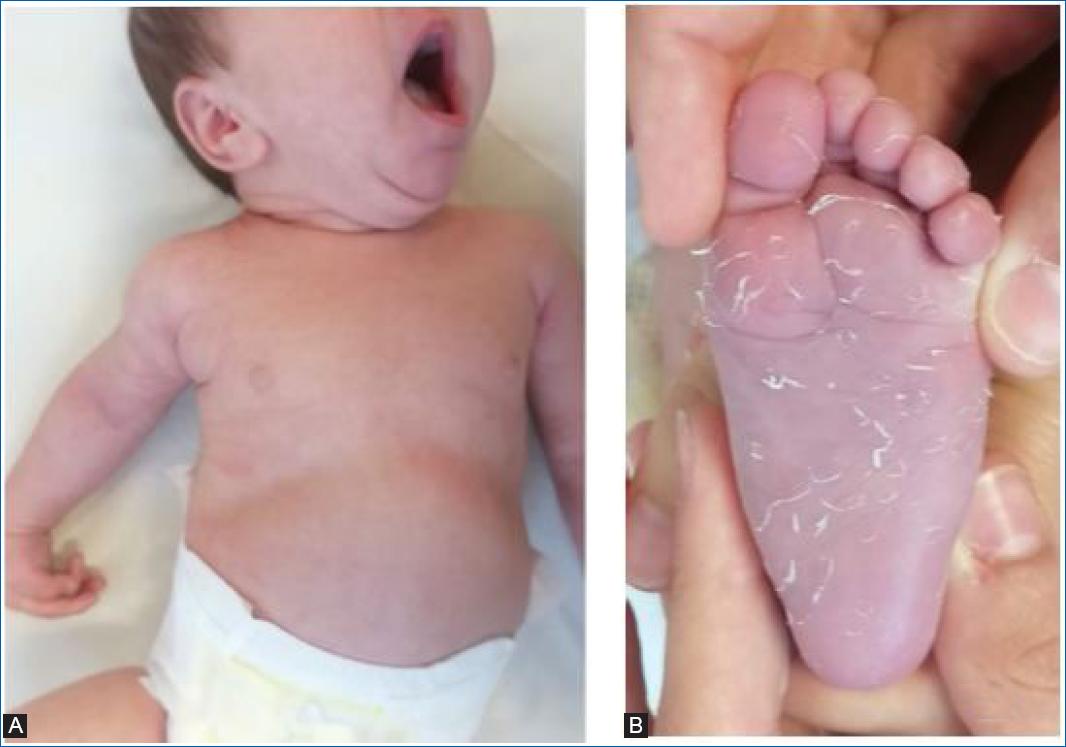
Oral candidiasis resolved on the third day of treatment, and skin lesions progressively resolved with desquamation after fourteen days of intravenous fluconazole and topical clotrimazole (Fig. 4).
There was no evidence of immunodeficiency in the initial investigation, and the patient is currently under clinical follow-up.
Discussion
We present a case of extensive mucocutaneous candidiasis appearing in the first 48 hours of life. The involvement of the palms and soles, as well as the involvement of the oral mucosa and perineal region, were essential for suspecting the diagnosis. There was no evidence of invasive disease, and there was a good evolution under topical and intravenous antifungal agents.
Congenital cutaneous candidiasis (CCC) develops in the 1st week of life and may be present in the first few hours5. It presents a broad spectrum of symptoms, from rash to severe invasive disease. It is clinically characterised by a generalised exanthema, including erythematous macules and papules, vesicles and pustules that later undergo desquamation1,5. The entire body can be affected, often involving the palms and soles, which is an essential feature for the differential diagnosis6. The most affected areas are usually the back, extensor surfaces and skin folds.
It is necessary to consider other differential diagnoses of exanthemas in newborns. Neonatal candidiasis differs from CCC by appearing after the first week of life and usually affects only the perianal region and oropharynx1. Other differential diagnoses of CCC are extensive-erythema toxicum neonatorum, transient neonatal pustular melanosis, several infectious causes (group B Streptococcus, L. monocytogenes, herpes simplex virus, varicella zoster-virus, among others), miliaria or even Langerhans cell histiocytosis5. Some features help in the differential diagnosis—the presence of plaques in the umbilical cord (funisitis) should raise the suspicion of CCC but also of L. monocytogenes infection; lesions on the palms and soles are characteristic of Candida infection. Due to the difficulty in the differential diagnosis, it is imperative to confirm the diagnosis by cultural examination with isolation of Candida spp.
Usually, it is self-limiting but can progress to invasive diseases such as candidemia, pneumonia, arthritis and endocarditis4. The probability of invasive disease and the mortality rate is inversely proportional to birth weight and gestational age.
Treatment of clinically stable term neonates without evidence of disseminated disease can be controversial. Nonetheless, studies also indicate that topical and shorter treatment cycles (< 14 days) are risk factors for the dissemination of Candida into the bloodstream3. In our case, due to the extensive mucocutaneous involvement with a higher risk of invasion, it was decided to initiate systemic therapy. Systemic treatment with fluconazole, amphotericin B deoxycholate and liposomal amphotericin B has been demonstrated to be effective3. In our hospital, amphotericin B deoxycholate is not available. Due to the low rate of fluconazole resistance in our unit and since liposomal amphotericin B is not the first choice in case of urinary tract infection7 (which could not be excluded due to the great mucocutaneous involvement contraindicating aseptic collection), it was decided to use fluconazole as first choice with excellent clinical outcome.