doi: 10.4152/pea.201406417
-->Experimental and Theoretical Evaluation on the Effect of the Terminal Side Chain of a Polymeric Surfactant on the Inhibition Efficiency of Aluminum Corrosion in Acid Medium
S. M. Sayyaha,* , M. M. El-Deeba,b , S. S. Abd El-Rehimc , R.A. Ghanemd and S.M. Mohameds
a Polymer Research Lab., Chemistry Department, Faculty of Science, Beni Suef University, 62514 Beni Suef, Egypt
b Chemistry Department, Faculty of Science, Ha'il University, P. Box 2440, 81451 Ha'il, Saudia Arabia
c Chemistry Department, Faculty of Science, Ain Shams University, Abasia, Egypt
d Department of Chemistry, University of A-al-Bayt,P.O. Box 130040, Mafraq 25113, Jordan
]]> Abstract
Inhibition effect of poly 3-dodecyloxy aniline polymeric surfactant (PC12H) on the corrosion of aluminum in 0.5 M HCl solution was investigated using potentiodynamic polarization technique under different experimental conditions. Inhibition efficiency and thermodynamic functions for dissolution and adsorption of PC12H are compared with Poly 3-(dodecyloxy sulfonic acid) aniline (PC12SO3H) to explain the effect of terminal side chain on the corrosion protection. Data show that the presence of -SO3H as a terminal group in PC12SO3H decreases the inhibition efficiency from 88.7%, in case of PC12H, to 55.1% in case of PC12SO3H. Quantum chemical calculations were performed on PC12H and PC12SO3H to investigate the relationship between the molecular structures and their inhibition efficiencies.
Keywords: Corrosion, Polymers, Electrochemical Techniques, Adsorption, Thermodynamic Properties, Computational Techniques.
Introduction
Corrosion inhibitors play a very important role in protecting metals and alloys. Aluminum has a remarkable economic and industrial importance owing to its low cost, light weight, high thermal and electrical conductivity. The most important feature of aluminum is its corrosion resistance due to the formation of a protective film on its surface upon its exposure to atmosphere or water [1]. Several authors [2-4] studied the corrosion of aluminum and their inhibition by organic inhibitors in acid solutions. Various surfactants as 1, 1-(laurly amido) propyl ammonium chloride are being studied as a corrosion inhibitor for pure aluminum in acid media [5-6]. Many of organic compounds as electroactiveconducting polymers [7-10], carboxylic organic acids [11, 12], fatty acids [13], dicyandiamide and some of its related compounds [14], hydrazine compounds [15], triazole and thiazole derivatives [16], were also found to inhibit the corrosion of aluminum.
Sayyah et al. [17, 18] studied the inhibition effect of the series of the prepared monomeric surfactants and their analog polymers with different hydrophilic side chain length on the corrosion of aluminum in acid medium. Results indicate that the hydrophilic side chain has remarkable influence on the inhibition efficiency of the prepared surfactants and the inhibition efficiency increases in both monomeric and polymeric surfactants as the length of the side chain increases. Effect of 3-(10-sodiumsulfonate decyloxy) aniline monomeric surfactant and its analog polymer on the corrosion of aluminum in 0.5 M HCl was studied [19]. The results indicate that the inhibition occurs through the adsorption of surfactant molecules on the aluminum surface as well as the inhibition efficiency increases with increasing the inhibitor concentrations, and decreases with raising the temperature.
Quantum chemistry calculations have been widely used to evaluate the inhibition performance of corrosion inhibitors, which can quantitatively study the relationship between inhibition efficiency and molecular reactivity [20-22]. With this method, the capability of inhibitor molecules to donate or accept electrons can be predicted with analysis of the global reactivity parameters, such as energy gap between HOMO and LUMO, chemical potential, hardness, softness, dipole moment, and electrophilicity index, etc.
Zhang et al. [23] studied the corrosion inhibition of four 1-R-2-undecylimidazoline compounds for carbon steel by quantum chemistry and molecular mechanics methods. Results indicate that the hydrophilic groups (R) affect on the molecular reactivity, binding strength between self-assembled monolayer and Fe surface, and compactness of the inhibitor.
The present study aims to investigate the inhibition effect of poly 3-dodecyloxy aniline polymeric surfactant (PC12H) on the corrosion of aluminum in 0.5 M HCl solution using potentiodynamic polarization techniques, as well as the inhibition efficiency and thermodynamic functions for dissolution and adsorption of PC12H are compared with our previous data for PC12SO3H to explain the effect of the terminal side chain on the corrosion protection of these two compounds. Quantum chemical calculations were performed on PC12H and PC12SO3H to determine the relationship between the molecular structures and their inhibition efficiencies.
]]>Experimental
Materials
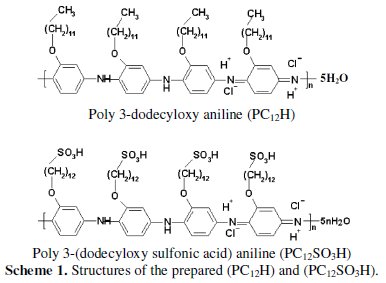
Poly 3-dodecyloxy aniline (PC12H) and poly 3-(dodecyloxy sulfonic acid) aniline (PC12SO3H) have been prepared by Sayyah et al. [18, 24] and the structures are given in scheme (1).
Concentrated hydrochloric acid is chemically a pure grade product provided by Prolabo-Chemical Co., (U.K.). Bi-distilled water is used to prepare all solutions.
Potentiodynamic polarization measurements
Working electrode is made of aluminum metal provided by the Egyptian Aluminum Company, Naga Hammadi, Egypt, which has the following chemical composition (wt%): 99.57% Al, 0.31% Fe, 0.07% Si, 0.015% Ti, 0.0016 %Zn, 0.0003% Cr, 0.0019% Mg, 0.0021% Mn and 0.0007% Cu.
The rod is axially embedded in Araldite holder to offer an active flat disc shaped of surface area 0.785 cm2. Prior to each experiment, the working electrode was polished successively with fine emery paper. The polished metal surface was rinsed with acetone and distilled water before dipping it into the electrolytic cell. A platinum wire was used as the counter electrode and a saturated calomel electrode as a reference electrode to which all potentials are referred. Electrochemical experiments were performed using a Wenking PGS95, Potentiostat /Galvanostat connected to a PC computer. Current potential curves (I-E) are recorded with a computer software (ECT). Experiments were carried out by changing the electrode potential automatically from the starting potential towards more positive values using scan rate of 25 mV s-1 till the end of the experiments.
]]> Computational techniquesQuantum chemical calculations were conducted with hyperchem 8.0 and Chem Bio3D Ultra software. The 3D structure of the inhibitor molecules was constructed with the build module implemented in HyperChem 8.0 software package and subsequently, optimized using molecular mechanics calculations, MM+ force field implemented in both software packages. The MM+ force field is an extension of MM2; force field refers to both an interatomic potential functional form and its relative parameters. Interatomic potentials are mathematical functions used to describe the potential energy of a statistical mechanical model formed by a system of particles. MM2 was developed primarily for conformational analysis of hydrocarbons and other small organic molecules. Calculations of the inhibitor molecules were accomplished by MM+. Frontier orbital's distribution, energy of optimized structure, bond length and bond angle were also obtained. On the other hand, analysis of the electrostatic potential surfaces (ESP) of the two compounds was made using the semi- empirical ZINDO method using Argus Lab 4 software.
Results and discussion
Potentiodynamic polarization measurement
Fig. 1 shows Tafel polarization curves of aluminum in 0.5 M HCl solution in the absence and presence of different concentrations of PC12H at 30 °C with a scan rate of 25 mVs-1.
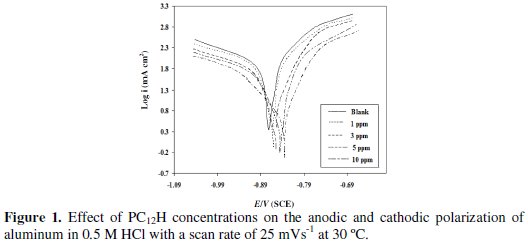
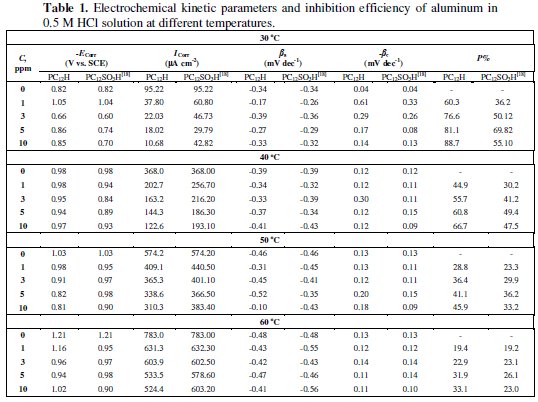
The electrochemical kinetic parameters, including Tafel constants for both cathodic (βc) and anodic (βa) reactions, the corrosion current densities (Icorr) and the corrosion potentials (Ecorr), were obtained and are listed in Table 1, and are compared with the data that we have obtained in our previous work in case of PC12SO3H [18].
 ]]>
]]>
The inhibition efficiency (P%) was calculated according to Eq. (1) and is depicted in Table 1.
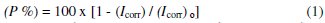
where (Icorr)0 and (Icorr) are the corrosion current densities in the absence and presence of inhibitor surfactants, respectively. Results indicate that the corrosion current density decreases with an increase in the inhibitor concentrations for both polymeric surfactants and the lowest value of 10.68 μA cm-2 (P% = 88.7) is obtained with PC12H, indicating that PC12H is more effective corrosion inhibitor than PC12SO3H.
In acidic solutions, the anodic process of corrosion is the passage of metal ions from the metal surface into the solution, while the principle of cathodic process is the discharge of hydrogen ions to produce hydrogen gas, and the inhibitor may affect either one or both of the anodic and cathodic processes [25]. When the change in the Ecorr value is greater than 85 mV, a compound can be recognized as an anodic or cathodic type inhibitor [26].
Inspections of these data reveal that the presence of PC12H and PC12SO3H shifts the corrosion potential (Ecorr) towards less negative values, indicating that the studied polymeric surfactants act predominantly as an anodic type inhibitor, and thus, the polymeric surfactant inhibitors are probably adsorbed on the anodic part of the aluminum surface and form a barrier layer of adsorbed molecules that separates the metal from direct contact with the corrosion medium. The adsorption behavior is attributed to the presence of electrostatic interaction between the adsorption centers of the surfactant polymer inhibitors and the metal surface.
It is found that the maximum inhibition efficiency of PC12H (P%=88.7) is higher than that of PC12 SO3H (P%=55.1), Although, the two compounds have the same number of carbon atoms in the alkyl chain but the difference in the protection can be attributed to the terminal group; the presence of CH3 as a terminal group in PC12H decreases the repulsion between the inhibitor and the negatively charged aluminum surface and allows a closed layer from the inhibitor to be adsorbed more easily on aluminum surface and hence increasing the inhibition efficiency. These results are in a good agreement with the results obtained from the theoretical calculations.
The effect of temperature on the corrosion of aluminum in 0.5 M HCl solution in the absence and presence of 10 ppm of PC12H and PC12SO3H [18] with scan rate of 25 mVs-1 was studied and the electrochemical kinetic parameters are tabulated in Table 1. It is observed that the corrosion current densities (Icorr) in the absence and presence of both inhibitors increase with increasing the temperature. Such results declared that the rate of corrosion of aluminum enhanced with increasing the temperature. The inhibition efficiency (P%) of both PC12H and PC12SO3H was calculated at different temperatures and is given in Table 1. It has been reported [17 - 19] that, for acid corrosion of metals, the logarithm of the corrosion rate is a linear function with 1/T (following Arrhenius equation type)
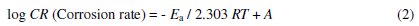 ]]>
]]>
where Ea is the apparent activation energy, R is the universal gas constant and A is the Arrhenius pre-exponential factor. The logarithm of the corrosion rate was plotted against 1/T (c.f. Fig. 2) for each concentration and the values of (Ea) have been calculated and tabulated in Table 2.
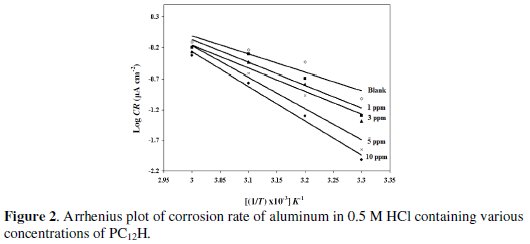
An alternative formula of the Arrhenius equation is the transition state equation:
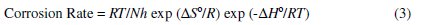
where h is the Planck's constant, N is the Avogadro's number, ΔS0 is the entropy of the activation, and ΔH0 is the enthalpy of activation. The plot of log (Corrosion Rate / T) vs. 1/T gives a straight line with a slope of (-ΔH0/2.303R), from which the values of ΔH0 are calculated and listed in Table 2.
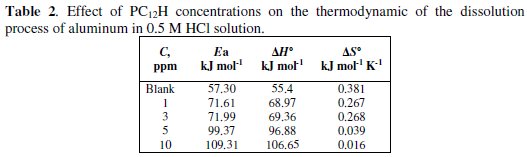
It is obvious that the value of Ea is higher in the presence of both PC12H and PC12SO3H when compared with the blank and the value in case of PC12H (10 ppm) is 109.31 kJ mol-1, while it is 76.2 kJ mol-1 [18] in the case of PC12SO3H (10 ppm), which agrees with our results indicating that PC12H is more effective corrosion inhibitor than PC12SO3H. Furthermore, with increasing the concentrations of both the polymeric surfactant inhibitors, the activation energies increase and the inhibition efficiency decreases; this behavior is attributed to an appreciable decrease in the adsorption process of the inhibitors on the metal surface with increasing the temperature [27,28]. A decrease in inhibition efficiencies (P%) with increasing the temperature, with analogous increase in corrosion activation energy (Ea) in the presence of the inhibitors compared to their absence is a good evidence of the physical adsorption mechanism [27,29]. ΔH0 for the corrosion of Al in the presence of PC12H and PC12SO3H is higher than that of blank solution and increases with an increase in the inhibitors concentrations, indicating that more energy barrier for the corrosion reaction in the presence of the inhibitor is attained and this energy barrier of corrosion reaction increases as the concentration of the inhibitor increases.
]]> Adsorption isothermIn order to get more information about the mode of adsorption of the inhibitors on the metal surface at different temperatures, the obtained data have been tested with several adsorption isotherms. Experimental data were fitted into Langmuir adsorption isotherm as shown in Fig. 3.
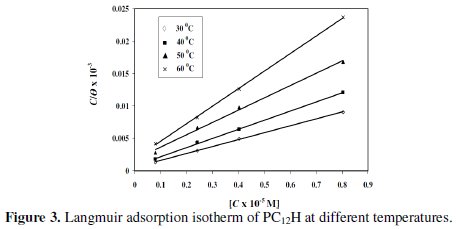
The adsorption isotherm relationship is represented by the following equation [18, 19]:

Values of Ci are the concentration of the inhibitor in bulk solution, θ is the surface coverage (θ = P/100) and kads is the adsorption equilibrium constant.
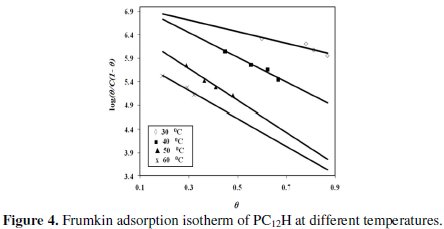
Frumkin isotherm [18, 19] was also found to fit well with the obtained experimental data, as represented in Fig. 4.
 ]]>
]]>
The adsorption isotherm relationship of Frumkin is represented by the following equation:

where (a) is the lateral interaction term describing the molecular interactions in the adsorption layer and the heterogeneity of the surface and is a measure for the steepness of the adsorption isotherm. It may be positive or negative values. The more positive value of (a), the steeper is the adsorption isotherm.
The apparent thermodynamic parameters (ΔHads and ΔSads) for the surfactant adsorption on the aluminum surface in 0.5 M HCl at different temperatures were determined from the slopes and intercepts of the lines of log kads vs. 1/T plots using the following equation [18,19]:
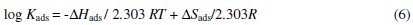
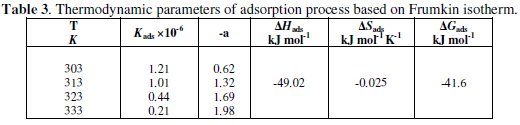
where ΔHads and ΔSads are the enthalpy and entropy of the adsorption process, respectively. Values (kads), (ΔHads), (ΔSads) and (ΔGads) were obtained and listed in Table 3.
 ]]>
]]>
The calculated values of ΔGads in case of PC12H (-41.6 kJ mol-1) and in case of PC12SO3H (-36.45 kJ mol-1[18]) are close to -40 kJ mol-1; this reflects electrostatic interaction between the inhibitor and the charged metal surface and this adsorption process is described as physical adsorption [30]. These data are also in a good agreement with the data obtained from the thermodynamic activation parameters.
Quantum Chemical Calculation
Molecular Reactivity
The optimized geometries of PC12H and PC12SO3H are shown in Fig. 5 as well as bond lengths, bond angles and dihedral angles are tabulated in Table 4.
Electronic behavior of PC12H and PC12SO3H was studied to explain the electron donor/acceptor properties of these compounds. The effectiveness of a particular donor/acceptor can be assessed by computation of certain quantum chemical parameters such as the energy of the highest occupied molecular orbital (EHOMO), the energy of the lowest unoccupied molecular orbital (ELUMO) and LUMO- HOMO gap (ELUMO - EHOMO). EHOMO is a quantum chemical descriptor, which is often associated with the electron donating ability of the molecule. High value of EHOMO indicates the tendency of a molecule to donate electrons to an appropriate acceptor molecule with empty molecular orbitals. Therefore, the energy of ELUMO indicates the ability of a molecule to accept electrons. On the other hand, values of energy gap (ΔE = ELUMO - EHOMO) are the measure of excitation energy to remove an electron from the last occupied molecular orbital. Hence, an increase in the values of EHOMO can facilitate the disposition of the molecule to donate orbital electrons to an appropriate acceptor and decrease in ELUMO is an indication of strong interaction with the metal [27].
Figs. 6 and 7 represent the highest occupied molecular orbital (HOMO) and lowest occupied molecular orbital (LUMO) for PC12H and PC12SO3H, respectively.
]]>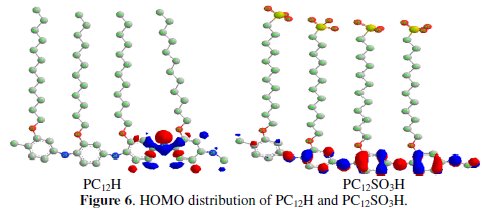
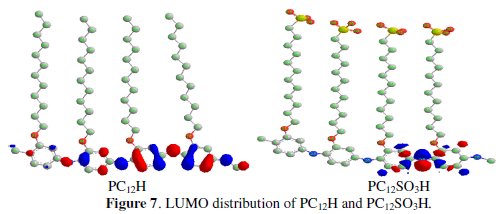
It could be seen that the frontier molecular orbitals of two compounds are generally located on the head group that be preferentially adsorbed onto the metal surface as active sites. It can be observed that HOMO energy of PC12H and PC12SO3H is -6.275 and -6.278 eV, respectively. These results indicate that the electron donating behaviour of these compounds follows the order PC12H > PC12SO3H. On the other hand, the LUMO energies of PC12H and PC12SO3H are -5.016 and -4.864 eV, respectively, which indicates that the capability of accepting electrons of the studied compounds follows the order PC12H > PC12SO3H.
Low value of energy gap (ΔE) leads to easier polarization of the molecule and greater adsorption on the surface. Thus, a small ΔE of PC12H (1.259 eV) facilitates its adsorption on aluminum metal and enhances its inhibition efficiency (P%=88.7) compared with PC12SO3H (P%= 55.1). This behaviour is in a good agreement with the results obtained from the polarization measurements.
Electrostatic potential surface
Electrostatic potential surface generally provides information regarding the chemical reactivity of a molecule. The electrostatic potential generated in the space around a molecule by the charge distribution is helpful to understand how much electrophilic or nucleophilic the molecular species is.
Fig. 8 represents the electrostatic potential map for PC12H and PC12SO3H.
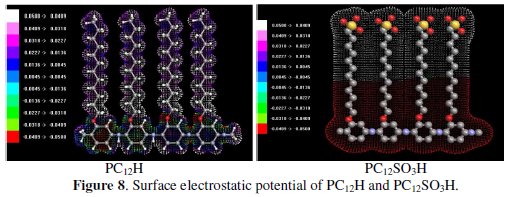
]]> Results show that PC12SO3H has a more negative charge than PC12H, therefore, the electrophilicity of PC12H is higher than that of PC12SO3H. These results explain the good electrostatic attraction between PC12H and the negatively charged aluminum surface and thus, the higher adsorption and inhibition efficiency for PC12H compared with that of PC12SO3H. These theoretical calculations are in a good agreement with the experimental results.
Conclusions
• The terminal side chain has a great effect on the inhibition efficiency of the studied polymeric surfactants.
• Inhibition efficiency of PC12H is higher than that of PC12SO3H.
• Inhibition efficiency increases with increasing the inhibitor concentrations but decreases with raising the temperature.
• Physical adsorption was suggested for both polymeric surfactant inhibitors.
• Langmuir and Frumkin isotherms are found to fit the adsorption of the studied compounds.
• Quantum chemical calculations and the electrostatic potential surface explain the effect of the terminal side chain on the inhibition efficiency for both studied polymeric surfactant compounds.
]]> References
1. Sorkhabi H A, Ghasemi Z, Seifzadeh D. Appl Surf Sci. 2005;247:408. [ Links ]
2. Maayta A K, Al-Rawashdeh N A. Corros Sci. 2004;46:1129. [ Links ]
3. Branzoi V, Golgovici F, Branzoi F. Mater Chem Phys. 2002;78:122. [ Links ]
4. Sherif E M, Park S M. Electrochim Acta 2006;51:1313. [ Links ]
5. Abd El Rehim S S, Hassan H, Amin M. Mater Chem Phys. 2002;78:337. [ Links ]
6. Zhao T, Mu G. Corros Sci. 1999;41:1937. [ Links ]
7. Perucki M, Chandrasekhar P. Synth Met. 2001;119:385. [ Links ]
8. Breslin C, Fenelon A, Conroy K. Mater Des. 2005;26:233. [ Links ]
9. Gelling V J, Wiest M M, Tallman D E, et al. Prog Org Coat. 2001;43:149. [ Links ]
10. Epstein A J, Smallfield J A, Guan H, et al. Synth Met. 1999;102:1374. [ Links ]
11. Muller B. Corros Sci. 2004;46:159. [ Links ]
12. Bereket G, Yurt A. Corros Sci. 2001;43:1179. [ Links ]
13. Foad El-Shebini E E, Abd-El-Wahab S M, Deyab M A. Mater Chem Phys. 2003;82:631. [ Links ]
14. Maitra A, Barua S. Corros Sci. 1974;14:587. [ Links ]
15. Moussa M N, Taha F I, Gouda M A, et al. Corros Sci. 1976;16:379. [ Links ]
16. Zheludkevich M L, Yasakau K A, Poznyak S, et al. Corros Sci. 2004;47:3368. [ Links ]
17. Sayyah S M, Abd El-Rehim S S, El-Deeb M M, et al. Egypt J Chem. 2012;55:583. [ Links ]
18. El-Deeb M M, Sayyah S M, Abd El-Rehim S S, et al. Arab J Chem. In press. http://dx.doi.org/10.1016/j.arabjc.2013.09.018. [ Links ]
19. El-Deeb M M, Mohamed S M. J Appl Polym Sci. 2011;122:3030. [ Links ]
20. Camacho R L, Montiel E, Jayanthi N, et al. Chem Phys Lett. 2010;485:142. [ Links ]
21. Musa A Y, Mohamed A, Kadhum A H, et al. J Ind Eng Chem. 2012;18:551. [ Links ]
22. Danaee I, Ghasemi O, Rashed G R, et al. J Mol Struct. 2013;1035:247. [ Links ]
23. Zhang J, Qiao G, Hu S, et al. Corros Sci. 2011;53:147. [ Links ]
24. Sayyah S M, El-Salam H M A, Azzam E M. Inter J Polym Mat. 2006;55:1075. [ Links ]
25. Saliyana V R, Adhikari A V. Corros Sci. 2008;50:55. [ Links ]
26. Musa A Y, Jalgham R T, Mohamed A.Corros Sci. 2012;56:176. [ Links ]
27. Obot I B, Obi-Egbedi N O. Curr Appl Phys. 2011;11:382. [ Links ]
28. Saleh M M, Atia A A.J Appl Electrochem. 2006; 36:899. [ Links ]
29. Obot I B, Obi-Egbedi N O. Corros Sci. 2010;52:198. [ Links ]
30. Abd-El-Rehim S S, Sayyah S M, El-Deeb M M, et al. Mater Chem Phys. 2010;123:20. [ Links ]
*Corresponding author. E-mail address: smsayyah@hotmail.com
Received 15 October 2014; accepted 25 October 2014
]]>