doi: 10.4152/pea.201603213
-->Synthesis, Characterization and Corrosion Protection Properties of Imidazole Derivatives on Mild Steel in 1.0 M HCl
O. Krima , S. Jodehb,* , M. Messalic , B. Hammoutid,e , A. Elidrissic , K. Khaleda , R. Salghief and H. Lgazg
a LCAE-URAC18, Faculte des Sciences, Universite Mohammed Premier, B.P. 717, 60000 Oujda, Morocco
b Department of Chemistry, An-Najah National University, P.O. Box 7, Nablus, Palestine
c Chemistry Department, Faculty of Science, Taibah University, 30002, Al-Madinah Al-Mounawwara, Saudi Arabia
d Electrochemistry Research Laboratory, Ain Shams University, Faculty of Education, Chemistry Department, Roxy, Cairo, Egypt
e Materials and Corrosion Laboratory, Taif University, Faculty of Science, Chemistry Department, Taif, Hawiya 888, Saudi Arabia
]]> f Department of Chemistry, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabiag Laboratory of Environmental Engineering and Biotechnology, ENSA, Universite Ibn Zohr, PO Box 1136, 80000 Agadir, Morocco
Abstract
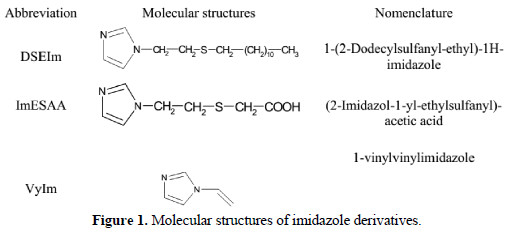
1-(2-Dodecylsulfanyl-ethyl)-1H-imidazole (DSEIm) and 2-Imidazol-1-yl-ethylsulfanyl)- acetic acid (ImESAA) were synthesized via radical catalysis method and characterized using 1H NMR and 13C NMR spectroscopy. The corrosion performances of mild steel specimens were studied by three imidazole derivatives include: 1vinylvinylimidazole (VyIm), DSEIm and ImESAA, which were investigated in 1.0 M HCl using weight loss measurements, potentiodynamic polarization and electrochemical impedance spectroscopic (EIS) method. The results obtained show that DSEIm is the best corrosion inhibitor; its inhibition efficiency (E %) increases with increasing the inhibitor concentration, but decreases with the raise of temperature. Potentiodynamic polarization studies clearly revealed that the inhibitors changed the mechanism of hydrogen evolution, and that they acted as mixed inhibitors, but most effectively in the cathodic range. The higher values of activation energy (Ea) in the inhibited solution can be correlated with the increased thickness of the double layer; this is interpreted with physical adsorption of the inhibitor onto the metal surface resulting in the formation of a surface film. Adsorption of imidazole derivatives have been studied with Monte Carlo simulations.
Keywords: Adsorption, Aluminium, Corrosion Inhibition, Cefuroxime axetil, SEM.
Introduction
The phenomenon of corrosion in mild steel represents a fundamental problem both in basic research in industry. The prevention of corrosion is vital not only for increasing the lifetime of equipment, but also in decreasing the dissolution of toxic metals from the components into the environment [1]. Mild steel is one of the most widely used materials of construction. Corrosion of mild steel has an enormous economic impact. The understanding of the corrosion problem and the solution to tackle it is a very active field of research. In corrosion of mild steel, acids like HCl and H2SO4 are the most important pickling acids, which are widely used in steel and ferrous alloy industry [2].
However, the main problems in using mild steel in these applications are due to the uniform corrosion attack. At this juncture, there is a need to improve the corrosion performance of mild steel. This can be achieved by adding appropriate concentrations of the organic compounds as inhibitors.
]]> The effect of organic compounds containing heteroatoms on the corrosion behavior of iron and steel in acidic solutions has been recognized for a long time [3-7].Most of the effective organic corrosion inhibitors contain multiple bonds and heteroatoms such as oxygen, nitrogen, sulfur, with which the inhibitors can adsorb on the metal surface [8-11]; well-known inhibitors are benzaldehydes [12], furans [13], isoxazolidines [14,15], triazoles [16-20], pyridines [21-23], thiadiazole [24,25], oxadiazoles [26], and imidazolines [27-30]. As expected, the physical/chemical adsorption of the inhibitor at the metal surface is influenced by various factors including electron density and л-character of donor atom, types of functional groups, steric effect, etc., [11, 12].
Moreover, nitrogen-containing heterocyclic compounds are considered to be effective corrosion inhibitors [7]. Among these, imidazole and its derivatives have been found to be the most effective and widely used organic compounds for corrosion inhibition of mild steel in acid medium [31-32] due to the presence of the electron donating heteroatom namely, nitrogen. In addition, the compounds containing both nitrogen and sulphur can provide excellent inhibition compared with compounds containing only nitrogen or sulphur [33-35]. Their adsorption is generally explained by the formation of an adherent film on the metal surface [36].
Despite several experimental and computational tools that have been designed to study the structural characteristics of the inhibitor molecules, little is known about the interaction between the adsorbed inhibitor molecules and the corroding metal surface. A practical route to study these complex processes is computer simulations using suitable models [37-38].
In continuation of our effort in developing corrosion inhibitors with high effectiveness and efficiency, the present paper describes the synthesis of imidazole derivatives using radical catalyzed addition of mercaptoacetic acid and 1-dodecanethiol to 1-vinylvinylimidazole, as described elsewhere [39-41]. The properties of these compounds, DSEIm and ImESAA, as corrosion inhibitors for mild steel in molar HCl were studied by gravimetry, potentiokinetic polarization, electrochemical impedance spectroscopy (EIS) and Monte Carlo simulation methods.
Materials and methods
Materials
The chemical composition (wt.) of the mild steel used in the present study was 0.09 wt.% P, 0.38 wt.% Si, 0.01 wt.% Al, 0.05 wt.% Mn, 0.21 wt.% C, 0.05 wt.% S. The mild steel specimens were mechanically grounded and polished using different grades of abrasive SiC papers, then washed with distilled water, degreased with acetone and subsequently dried at room temperature. Corrosion media were prepared from HCl and double-distilled water.
Synthesis
]]> Initially, 1-(2-Dodecylsulfanyl-ethyl)-1H-imidazole (DSEIm) was synthesized according to a previously reported procedure using the method of telomerization [39, 40]. In a 250 mL three-necked flask equipped with a condenser and a nitrogen flux, 18.6 g (9.2 10-2 mol) of 1-dodecanethiol dissolved in 100 mL of acetonitrile was introduced. A solution containing 4 g (4.2 10-2 mol) of 1vinylimidazole (VyIm) and 0.07 g (4.2 10-4 mole) of azobisisobutyronitrile (AIBN) as initiator was then added dropwise. The mixture was stirred and heated using oil bath at 353K under a continuous flow of nitrogen for 3 h. The solvent and excess thiol were then evaporated off under vacuum and the product isolated by trituration with methanol. The crude sample was purified by column chromatography on silica using ethyl acetate as eluent, giving the final monoadduct as a viscous orange liquid in 84% yield. The reaction with mercaptoacetic acid was realized under the same conditions using 4 g (4.2 10-2 mol) of 1-vinylimidazole, 8.8 g (9.2 10-2 mol) of mercaptoacetic acid, and 0.07 g (4.2 10-4 mol) of AIBN.The monoadduct, (2-Imidazol-1-yl-ethylsulfanyl)-acetic acid (ImESAA) was again obtained as a viscous, orange liquid (yield 91%). Molecular structures of DSEIm and ImESAA were presented in Fig. 1.
The characterization of DSEIm and ImESAA was performed using 1H NMR and 13C NMR recorded for CDCl3 solutions with tetramethylsilane (TMS) as reference. The letters s, d, t, q, and m denote respectively singlet, doublet, triplet, quadruplet, and multiplet.
Measurements
Weight loss technique
Gravimetric experiments were carried out in a double-walled glass cell. The test pieces were cut nearly (1.5 cm × 1.5 cm × 0.04 cm), weighed and suspended in the test solution (1.0 M HCl) with and without the addition of inhibitors at a temperature of 308K in air atmosphere without bubbling.
After specified periods of time (6 h), the test pieces were taken out of the test solution, rinsed in doubly distilled water, dried as before and weighed again.
Polarization measurements
]]> Stainless steel strips with an exposed area of 1 cm2 were used. Measurements were carried out in a standard glass three-electrode electrolysis cylindrical Pyrex glass cell. The auxiliary and the reference electrodes are platinum and saturated calomel (SCE), respectively. 352 Soft CorrTM III software was used to control an EG&G Instruments potentiostat-galvanostat model 263A at a scan rate of 20 mV/min. The working electrode was initially kept at the corrosion potential for 30 min before recording the cathodic curves up to the -800 mV. The test solutions were de-aerated with pure nitrogen and the flow was maintained throughout the experiments. The inhibition efficiency E% was calculated using the following equation:
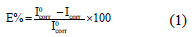
where Icorr and I0corr are the corrosion current densities of mild steel in the presence and absence of inhibitor, respectively.
Electrochemical impedance spectroscopy (EIS)
EIS experiments were conducted using computer controlled simulations with a Tacussel electrochemical system for calculation of polarization resistance (Rp) and double layer capacitance (Cdl) values. All experiments were performed with a frequency ranging from 100 mHz to 10 kHz (10 mV peak-to-peak). The impedance diagrams were plotted in the Nyquist representation. The inhibition efficiency Ew was calculated using the following equation:
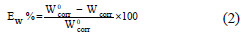
where Wcorr and W0corr are the corrosion rates of mild steel in the absence and presence of the inhibitor, respectively.
]]> Results and Discussion
Characterization
Characterization of DSEIm
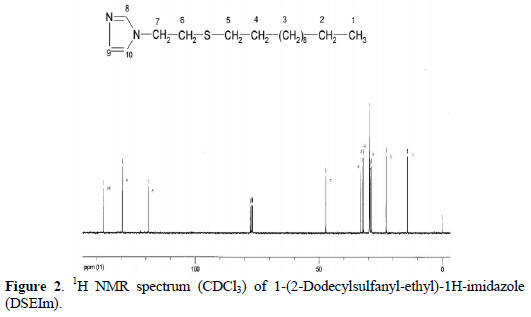
1H NMR spectrum of DSEIm, Fig. 2, shows two new triplets at (δ 2.38, 2.82) assigned to the methylene groups adjacent to sulfur, and the absence of three signals at 4.7, 5.1 and 6.8 confirms the loss of the vinyl unit.
The proton decoupled 13C NMR spectrum of DSEIm was also used to examine the chemical structure of the monoadduct and it is shown in Fig. 3.
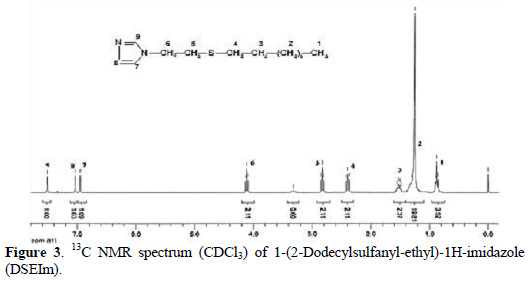
The signals at 177.05 and 175.34 ppm were due to the adjacent carbons to sulfur atom of the monomer.
]]> Characterization of ImESAA1HNMR (CDCl3/TMS): 2,92 (t, 2H); 3,38(s, 2H); 4,10 (t, 2H); 6,97(d, 1H); 7,01(d,1H); 7,54 (s,1H) 13CNMR (CDCl3): 32, 3; 38; 49, 4; 122,6; 125,5; 138,6; 177.
Inhibitive effect of imidazole derivatives
Gravimetric measurements
The effect of the addition of VyIm, DSEIm and ImESAA at different concentrations on the corrosion of mild steel in 1.0 M HCl solution was studied by measuring the weight loss after 6 h of immersion. Table 1 gathers the values deduced for Wcorr and E%.
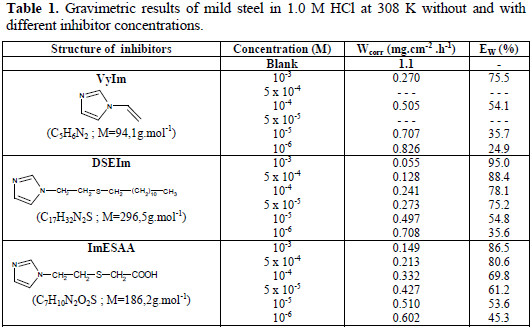
Gravimetric measurements showed that the corrosion decreased in the presence of VyIm, ImESAA and DSEIm. The inhibition efficiency increased with the inhibitor's concentration to reach 95% for DSEIm, 86.5% for ImESAA and 75.5% for VyIm at 10-3M (Table 1).
Thus, all derivatives studied inhibited the corrosion, but DSEIm was the most effective.
Polarization curves
]]> Anodic and cathodic polarization curves for mild steel in 1.0 M HCl with and without various concentrations of the inhibitors are shown in Fig. 4, 5 and 6.
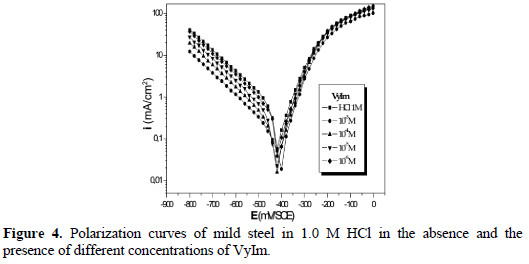
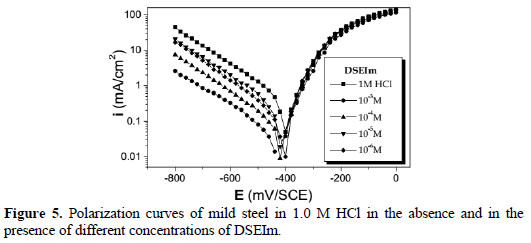
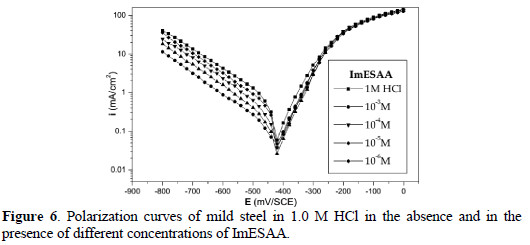
Values of the corrosion potential (Ecorr), corrosion current density (Icorr), and the inhibition efficiency (E%) were determined by the Tafel extrapolation method, and are given in Table 2.
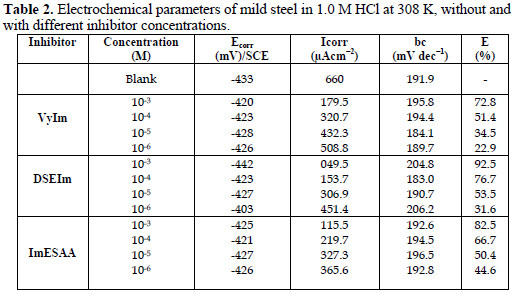
Addition of the various inhibitors decreased Icorr significantly for all the studied concentrations, due to the increase in the fraction of the electrode surface blocked by adsorption. The cathodic current versus potential curves gave rise to Tafel lines indicating that the hydrogen evolution reaction was activation controlled, bc values being slightly modified, and due to modification of the mechanism of the proton discharge reaction.
The anodic branches were only slightly affected in the presence of these inhibitors, the highest effect being observed with DSEIm. The inhibitors thus act as mixed inhibitors, but predominantly in the cathodic domain. The long side chain of DSEIm slightly, -S-(CH2)11-CH3, presumably plays a major role in the adsorption, and the presence of the acid function -S-CH2COOH in ImESAA must diminish the inhibition protection [42].
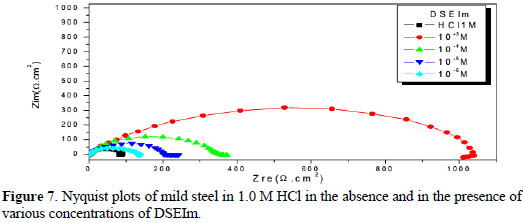
]]> Electrochemical impedance spectroscopic studiesFig. 7 presents the Nyquist diagrams obtained in the absence and the presence of DSEIm at different concentrations.
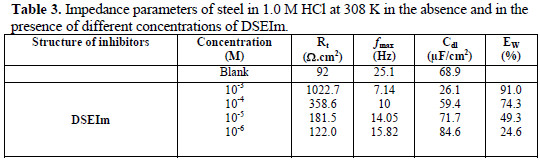
The impedance parameters calculated are given in Table 3.
The charge-transfer resistance values (Rct) were calculated from the difference in impedance at lower and higher frequencies [42] .To obtain the double-layer capacitance (Cdl), the frequency fmax at which the imaginary component of the impedance is maximum (-Zimax) was found, and Cdl values were obtained from the following equation:
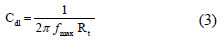
]]> In this case, the inhibition efficiency was calculated using the charge transfer resistance from:
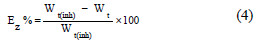
where Rt(inh) and Rt are the charge transfer resistance in the presence and absence of DSEIm, respectively.
The results show that Rt increased, and Cdl tended to decrease when the concentration of the inhibitor increased. A decrease in the Cdl values, which can result from a decrease in the local dielectric constant and/or an increase in the thickness of the electrical double layer, suggests that DSEIm functions by adsorption at the metal solution/interface [43].
The high values of inhibition efficiency observed in the case of DSEIm and ImESAA molecules are due to the presence of the sulfur atom and its highest molecular size.
The nitrogen and sulfur atoms are the major adsorption center for their interaction with the metal surface. The extent of inhibition is found to be more for DSEIm than for ImESAA, and efficiency reaches 95% and 86.5%, respectively at 10-3M (Table 1). This difference in the inhibition efficiency is attributed to the presence of an electron donor group of DSEIm (C12H25); this last group increases and enriches the electron density on the sulfur atom and, consequently, increases the density of the molecule (DSEIm) and provides an active adsorption center. On the other hand, the presence of an electron attractor group (-COOH) in the molecule ImESAA decreases its inhibition efficiency.
Effect of temperature
In acidic media, the corrosion of metal is generally accompanied with the evolution of H2 gas. This is usually followed by an increase in temperature and acceleration of the corrosion reaction which results in higher dissolution rate of the metal [22].
The results retired from Table 4 and Fig. 8 show that DSEIm inhibits the corrosion of steel in 1 M HCl solution.
]]>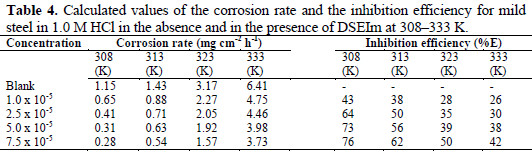
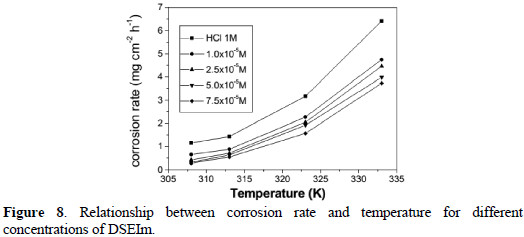
The corrosion rate was found to depend on the concentration of DSEIm and experimental temperature. It is evident from Table 4 and Fig. 8 that the corrosion rate decreased with increasing inhibitor concentration, but increased with a rise in temperature.
Table 4 also shows that inhibition efficiency (%E) decreased with rise in temperature, and reached its maximum value of 76% at a concentration of 7.5×10-5 M. A decrease in inhibition efficiency with an increase in temperature may be due to the desorption of inhibitor molecules at higher temperatures. This has been attributed to physical adsorption of the inhibitor molecule on the mild steel surface [44].
The apparent activation energy, Ea, for the corrosion reaction of mild steel in 1.0 M HCl in the presence and the absence of DSEIm was calculated from the Arrheniustype equation:
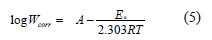
where Wcorr is the corrosion rate, Ea is the apparent activation energy, R is the molar gas constant (8.314 J K-1 mol-1), T is the absolute temperature, and A is the frequency factor.
For the corrosion of steel in acid medium, the natural logarithm of the corrosion rate (Wcorr) is a linear function with 1/T [45]. The plot of log Wcorr against 1/T for mild steel corrosion in 1.0 M HCl in the absence and presence of different concentrations of DSEIm is presented in Fig. 9.
]]>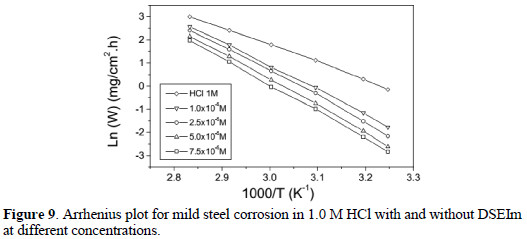
The activation energies calculated for HCl only and in the presence of 1×10-5 M, 2.5×10-5 M, 5×10-5 and 7.5×10-5M DSEIm were 53.43 kJ mol-1, 67.35 kJ mol-1, 71.21 kJ mol-1 and 84.51 kJ mol-1, respectively. It is observed that the apparent activation energy is higher in the presence of the inhibitor than in its absence.
The higher values of Ea in the inhibited solution can be correlated with the increased thickness of the double layer, which enhances the activation energy of the corrosion process [46]. This is interpreted with physical adsorption of the inhibitor onto the metal surface, resulting in the formation of a surface film. This type of inhibitor retards the corrosion at ordinary temperatures, but the inhibition is diminished especially at temperatures higher than 313K [23].
However, the adsorption phenomenon of an organic molecule is not considered only as a physical or as a chemical adsorption phenomenon, because a wide spectrum of conditions, ranging from the dominance of chemisorption or electrostatic effects may arise due to the complex nature of the corrosion inhibiting process [48].
Monte Carlo simulation adsorption studies
In the current study, selected imidazole derivatives have been simulated as adsorbates on an iron (Fe) (111) surface substrate to find the low energy adsorption sites and to investigate the preferential adsorption of the studied imidazole derivatives. To calculate the adsorption density as well as the binding energy of the studied inhibitors, the Monte Carlo method has been used. In this computational work, possible adsorption configurations have been identified by carrying out Monte Carlo searches of the configurational space of the iron/imidazole derivatives system as the temperature was gradually decreased. Imidazole derivatives were constructed and their energy was optimized using Forcite classical simulation engine [48-49]. The geometry optimization process is carried out using an iterative process, in which the atomic coordinates are adjusted until the total energy of a structure is minimized, i.e., it corresponds to a local minimum in the potential energy surface. Geometry optimization is based on reducing the magnitude of the calculated forces until they become smaller than defined convergence tolerances [50]. The forces on the atoms in the studied inhibitors are calculated from the potential energy expression and will, therefore, depend on the force field that is selected [5].
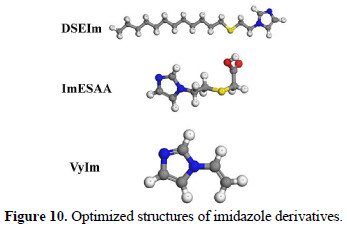
It was shown from chemical and electrochemical studies that the substitution of the hydrogen atom of amino group (-NH-) of imidazole by different substituents - dodecyl(ethyl)sulfane (DSEIm), ethylsulfanyl acetic acid (ImESAA) and vinyl (vyIm) - leads to an increase of the inhibition efficiency as given in Figures 5-7 and Tables 1-3. Each imidazole derivative contains imidazole ring, and the rest of the molecule can affect adsorption by its influence on the electron density of the side chains. Geometric and electronic structures of the imidazoles were calculated by optimization of their bond lengths and bond angles. The optimized structures are given in Fig. 10.
]]> The molecular dynamic (MD) simulations were performed using the software, Materials Studio [50]. The MD simulation of the interaction between the imidazole derivatives molecules and Fe (111) surface was carried out in a simulation box (17.38 Å × 17.38 Å × 44.57 Å) with periodic boundary conditions to model a representative part of the interface devoid of any arbitrary boundary effects [50]. The Fe (111) was first built and relaxed by minimizing its energy using molecular mechanics, then the surface area of Fe (111) was increased and its periodicity is changed by constructing a super cell, and then a vacuum slabwith 15 Å thicknesses was built on the Fe (111) surface [50]. The number of layers in the structure was chosen so that the depth of the surface is greater than the non-bond cutoff used in calculation. Using six layers of iron atoms gives a sufficient depth, so that the inhibitor molecules will only be involved in non- bond interactions with iron atoms in the layers of the surface, without increasing the calculation time unreasonably.
This structure is then converted to exhibit 3D periodicity. As 3D periodic boundary conditions are used, it is important that the size of the vacuum slab is enough (15 Å), and that the non-bond calculation for the adsorbate does not interact with the periodic image of the bottom layer of atoms in the surface. After minimizing the Fe (111) surface and the imidazole derivatives molecules, the corrosion system will be built by a layer builder to place the inhibitor molecules on the Fe (111) surface, and the behaviors of these molecules on the Fe (111) surface are simulated using the COMPASS (condensed phase optimized molecular potentials for atomistic simulation studies) force field. The adsorption locator module in Materials Studio 6.0 [50, 51] has been used to model the adsorption of the inhibitor molecules onto the Fe (111) surface, and therefore provides access to the energy of the adsorption and its effects on the inhibition efficiencies of the studied imidazole derivatives [52-57]. The binding energy between the imidazole derivatives and the Fe (111) surface was calculated using the following equation [58, 59]:
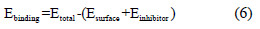
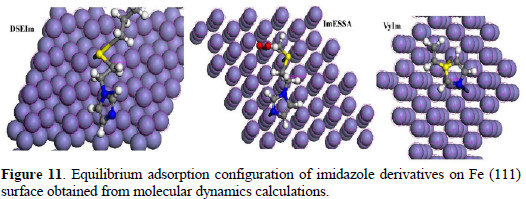
where E is the total energy of the surface and imidazole derivative, E is total surface the energy of the surface without the imidazole derivative, and E is the inhibitor energy of the imidazole derivative without the steel surface. The Metropolis Monte Carlo method in Adsorption Locator provides four step types for a canonical ensemble: conformer, rotation, translation and re-growth [60]. Fig. 11 shows the most suitable imidazole derivatives conformation adsorbed on Fe (111) substrate obtained by adsorption locator module.
The adsorption density of imidazole derivatives on the Fe (111) substrate is presented in Fig. 12 using Materials studio software.
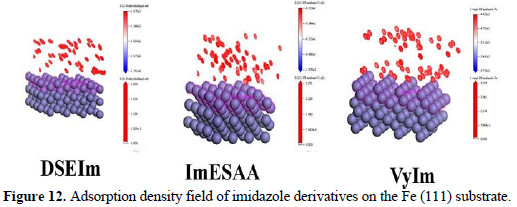
]]> As can be seen from Fig. 11 and 12 the imidazole derivatives molecules showthe ability to adsorb on a Fe (111) surface. Also, it has high binding energy to the Fe surface as seen in Table 5.
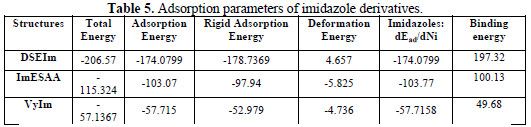
The outputs and descriptors calculated by the Monte Carlo simulation are presented in Table 5.
The parameters presented in Table 5 include total energy, in kcal mol-1, of the substrate-adsorbate configuration. The total energy is defined as the sum of the energies of the adsorbate components, the rigid adsorption energy and the deformation energy. In this study, the substrate energy (iron surface) is taken as zero. In addition, adsorption energy in kcal mol-1, reports energy released (or required) when the relaxed adsorbate components (imidazole derivatives in HCl) are adsorbed on the substrate. The adsorption energy is defined as the sum of the rigid adsorption energy and the deformation energy for the adsorbate components. The rigid adsorption energy reports the energy, in kcal mol-1 , released (or required) when the unrelaxed adsorbate components (i.e., before the geometry optimization step) are adsorbed on the substrate. The deformation energy reports the energy, in kcal mol-1, released when the adsorbed adsorbate components are relaxed on the substrate surface. Table 5 also shows that (dEads/dNi), which reports the energy, in kcal mol-1, of substrate-adsorbate configurations where one of the adsorbate components has been removed.
Fig. 13 shows the adsorption energy distribution of the imidazole derivatives on Fe (111).
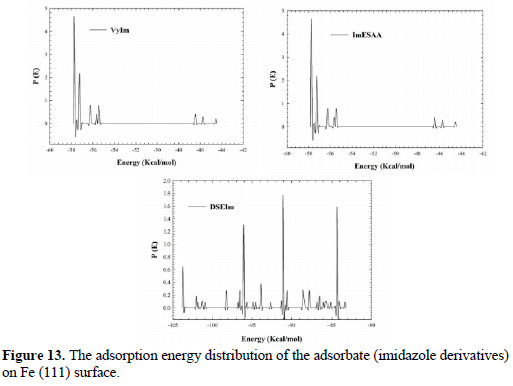
As can be seen in 13, the adsorption energy of DSEIm reaches (174 Kcal mole-1), ImESAA reaches (-103.7 Kcal mole-1) and VyIm reaches (-57.7 Kcal mole-1) which shows the adsorption power for DSEIm molecules on Fe (111) surface.
]]> Conclusions
In this research, we have prepared various monoadducts with Telomerisation method based on 1-vinylimidazol, and have studied their corrosion inhibition properties as a function of concentration and temperature. The results can be summarized as follows.
1) DSEIm and ImESAA were synthesized by Telomerisation method, purified and characterized by 1HNMR and 13CNMR spectroscopy.
2) The monoadducts were evaluated for its inhibitive characteristics for mild steel in 1.0 M HCl by gravimetric and electrochemical methods, and it was found that all inhibitors could be used to protect the mild steel in 1.0 M HCl. However, DSEIm is most suited to protect mild steel in acidic chloride environment.
3) Different results show that inhibition efficiency (%E) increased with increasing inhibitor concentration, but decreased with temperature.
4) The increase of the alkyl chain length attached to the inhibitor molecule (DSEIm) increases its inhibition efficiency compared to at the presence of a functional group (ImESAA).
5) The trend of a greater inhibition efficiency with a higher temperature, and the increase in the activation energy in the presence of DSEIm, suggest a physical adsorption mechanism, although chemisorption may also play a part in the inhibiting process.
6) Monte Carlo simulation studies help to find the most stable imidazole derivative conformation and adsorption sites. This information can help to gain further insight into the corrosion system, such as the most likely point of attack for corrosion on a surface, the most stable site for inhibitor adsorption, adsorption density of the inhibitor and the binding energy of the adsorbed layer.
References
]]>1. Solmaz R. Corros Sci. 2010;52:3321. [ Links ]
2. Srikanth A P, Lavanya A, Nanjundan S, et al. Appl Surf Sci. 2006;253:1810. [ Links ]
3. Cizek A. Mater Perform. 1994;33:56. [ Links ]
4. Batros M, Hakerman N. J Electrochem Soc. 1992;139:3429. [ Links ]
5. Kertit S, Hammouti B. Appl Surf Sci. 1996;93:59. [ Links ]
]]>6. Zucchi F, Trabnelli G, Brunoro G. Corros Sci. 1992;33:1135. [ Links ]
7. Quraishi M A, Jamal D. Mater Chem Phys. 2001;71:202. [ Links ]
8. Quraishi M A, Ansari F A. J Appl Electrochem. 2006;36:309. [ Links ]
9. Rafiquee M Z A, Kahn S, Saxena N, et al. Port Electrochim Acta. 2007;25:419. [ Links ]
10. Bentiss F, Traisnel M, Chaibi N, et al. Corros Sci. 2002;44:2271. [ Links ]
]]>11. Lebrini M, Lagrenee M, Venzin H, et al. Corros Sci. 2005;47:485. [ Links ]
12. Emregul K C, Hayvali M. Mater Chem Phys. 2004;83:209. [ Links ]
13. Machnikova E, Whitmire K H, Hackerman N. Electrochim Acta. 2008;53:6024. [ Links ]
14. Ali S A, Al-Muallem H A, Saeed M T, et al. Corros Sci. 2008;50:664. [ Links ]
15. Ali S A, Saeed M T, Rahman S U. Corros Sci. 2003;45:253. [ Links ]
]]>16. Yildirim A, Cetin M. Corros Sci. 2008;50:155. [ Links ]
17. Hassan H H, Abdelgahani E, Amin M A. Electrochim Acta. 2007;52:6359. [ Links ]
18. Khaled K F. Electrochim Acta. 2008;53:3484. [ Links ]
19. Lebrini M, Traisnel M, Lagrenee M, et al. Corros Sci. 2008;50:473. [ Links ]
20. Rammelt U. Koehler S. Reinhard G. Corros Sci. 2008;50:1659. [ Links ]
]]>21. Abd El-Maksoud S A, Fonda A S. Mater Chem Phys. 2005;93:84. [ Links ]
22. Obi-Egbedi N O, Obot I B, Eseola A O. Arab J Chem. 2014;7:197. [ Links ]
23. Ergun U, Yuzer D, Emreguel K C. Mater Chem Phys. 2008;109:492. [ Links ]
24. Bentiss F, Lebrini M, Vezin H, et al. Mater Chem Phys. 2004;87:18. [ Links ]
25. Lebrini M, Bentiss F, Venzin H, et al. Corros Sci. 2006;48:1279. [ Links ]
]]>26. Bentiss F, Traisnel M, Lagrenee M. Corros Sci. 2000;42:127. [ Links ]
27. Ramachandran S, Tsai B-L, Blanco M, et al. Langmuir. 1996;12:6419. [ Links ]
28. Villamizar W, Casales M, Martinez L, et al. J Solid State Electrochem. 2008;12:193. [ Links ]
29. Yoo SH, Kim YW, Chung K, et al. Corros Sci. 2012;59:42. [ Links ]
30. Tuken T, Demir F, Kicir N, et al. Corros Sci. 2012;59:110. [ Links ]
]]>31. Lagrenee M, Mernari B, Bouanis M, et al. Corros Sci. 2002;44:73. [ Links ]
32. Bentiss F, Traisnel M, Lagrenee M. Br Corros J. 2000;35:315. [ Links ]
33. Wang H L, Liu RB, Xin J. Corros Sci. 2004;46:2455. [ Links ]
34. Saliyan V R, Adhikari A V. Corros Sci. 2008;50:55. [ Links ]
35. Fouda A S, Ellithy A S. Corros Sci. 2009;51:868. [ Links ]
]]>36. Abed Y, Arrar Z, Aouniti A, et al. J Chem Phys. 1999;95:1347. [ Links ]
37. Khaled K F, Sherik AM. Int J Electrochem. 2013;8:10022. [ Links ]
38. Khaled K F. J Solid State Electrochem. 2009;13:1743. [ Links ]
39. Boyer C, Boutevin G, Robin JJ, et al. Polymer. 2004;45:7863. [ Links ]
40. Senhaji O, Robin J, Achchoubi M, et al. Macromol Chem Phys. 2004;205:951. [ Links ]
]]>41. Ameduri B, Boutevin B, Guida-Pietrasanta F, et al. J Fluorine Chem. 2001;107:397. [ Links ]
42. Aouniti A, Hammouti B, Kertit S, et al. Bull Electrochem. 1998;14:193. [ Links ]
43. Anuradha K, Vimala R, Narayanasamy B, et al. Chem Eng Comm. 2008;195:352. [ Links ]
44. Obot LB, Obi-Egbedi NO. Curr Appl Phys. 2011;5:382. [ Links ]
45. Soltani N, Behpour M, Ghoreishi SM, et al. Corros Sci. 2010;52:1351. [ Links ]
]]>46. Behpour M, Ghoreishi SM, Gandomi-Niasar A, et al. J Mater Sci. 2009;44:2444. [ Links ]
47. Solmaz R, Kardas G, Yazici B, et al. Colloids Surf A: Physicochem Eng Asp. 2008;312:7. [ Links ]
48. Barriga J, Coto B, Fernandez B, Tribol. Int., 2007;40:960. [ Links ]
49. Khaled K F. J Electrochem Soc. 2010;157:C116. [ Links ]
50. Khaled K F. J Solid State Electrochem. 2009;13:1743. [ Links ]
]]>51. Khaled K F. J Appl Electrochem. 2011;41:423. [ Links ]
52. Khaled K F, Al-Mobarak N A. Int J Electrochem Sci. 2012;7:1045. [ Links ]
53. Khaled K F, Abdelshafi N S, Elmaghraby A A, et al. Int J Electrochem Sci. 2012;7:12706. [ Links ]
54. Khaled K F, Abdel-Shafi N S. Int J Electrochem Sci. 2011;6:4077. [ Links ]
55. Khaled K F, Abdel-Shafi N S, Al-Mobarak NA. Int J Electrochem Sci.2012;7:1027. [ Links ]
]]>56. Khaled K F. Electrochim Acta. 2008;53:3484. [ Links ]
57. Tan Y. Corros Sci. 2011;53:1845. [ Links ]
58. Tamura H. Corros Sci. 2008;50:1872. [ Links ]
59. Soares C G, Garbatov Y, Zayed A, et al. Corros Sci. 2008;50:3095. [ Links ]
60. Gudze M T, Melchers R E. Corros Sci. 2008;50:3296. [ Links ]
]]>61. Accelrys to Release Enhanced Suite of Chemicals and Materials Modeling and Simulation Tools with Materials Studio(R) 4.1. in: Business Wire. New York; 2006. [ Links ]
62. Ermer O. Calculation of molecular properties using force fields. Applications in organic chemistry. in: Bonding forces. Vol. 27. Berlin Heidelberg: Springer, 1976. Pp 161-211. [ Links ]
63. Khaled K F. Appl Surf Sci. 2008;255:1811. [ Links ]
*Corresponding author. E-mail address: sjodeh@hotmail.com
Received 31 March 2016; accepted 20 April 2016
]]>