António Murinello1, Ana Carvalho2, A. Manuel Figueiredo1, Helena Damásio1, M. Jesus Murillo2, Garção Nunes3, Marta Baptista1, A. Raquel Martins1
1Unit of Internal Medicine, 2Unit of Histopathology, 3Unit of Urology, Hospital Curry Cabral, Lisbon, Portugal
Abstract
Malignant peritoneal mesothelioma mesothelioma is associated with a long a exposure to asbestos and usually has a poor prognosis. Short survival is due to late diagnosis, as patients are frequently pauci-symptomatic until advanced stage. Since the tumor is usually confined to the peritoneal cavity, extensive peritonectomy and hypertermic intraoperative intraperitoneal chemotherapy are associated with increased survival in selected patients. The synchronous occurrence of primary malignant peritoneal mesothelioma and renal cell carcinoma has not yet been described. A concise literature review of symptomatology of peritoneal mesothelioma, paraneoplastic syndromes (dysfunctional biochemical hepatopathy, wasting syndrome); role of asbestos fibers in pathogenesis; mechanisms of intraperitoneal dissemination; CT scan evaluation; relevance of immunohistochemistry for histopathologic diagnosis; staging of tumors; multidisciplinary approach for treatment of these malignancies is performed.
KEYWORDS: Malignant peritoneal mesothelioma, Asbestos, Paraneoplastic hepatopathy, Renal cell cancer, Cytoreductive surgery, Hyperthermic intraoperative intraperitoneal chemotherapy.
]]> Mesotelioma maligno primário do peritoneu associado a carcinoma de células renais - Revisão concisa baseada num caso clínico
Resumo
O Mesotelioma peritoneal maligno é um tumor maligno relacionado frequentemente com exposição prolongada a fibras de amianto, de mau prognóstico, de diagnóstico geralmente tardio, face à pouca expressão clínica na fase inicial da doença.
Como o mesotelioma evolui geralmente só na cavidade peritoneal, doentes seleccionados poderão ter maior sobrevivência se for possível a peritonectomia extensa e quimioterapia hipertérmica intraperitoneal intraoperatória. Os autores referem a sincronicidade ainda não descrita, de mesotelioma peritoneal maligno primário e carcinoma de Grawitz. São revistos concisamente: a clínica destes tumores, síndromes paraneoplásicos (disfunção bioquímica hepática, emagrecimento extremo); etiopatogenia da acção cancerígena das fibras de amianto; mecanismos de disseminação intraperitoneal; avaliação tomodensitométrica; importância da imunohistoquímica no diagnóstico histopatológico; estadiamento; importância do tratamento multidisciplinar destes tumores.
PALAVRAS-CHAVE: Mesotelioma peritoneal maligno, Amianto, Hepatopatia paraneoplásica, Carcinoma de células renais, Cirurgia cito-redutora, Quimioterapia hipertérmica intra-peritoneal intra-operatória.
INTRODUCTION
Malignant Peritoneal Mesothelioma (MPM) is a rare malignancy, occurring at any age, although more commonly in men in the 5th/6th decades of life due to increased occupational exposure to asbestos. MPM has a poor prognosis. In untreated cases, median survival ranges from 5 - 12 months. Even in patients receiving traditional multimodality treatment (surgery + chemotherapy), only exceptionally longterm survival cases were mentioned1. The incidence of MPM began to increase after 1930 with the frequently industrial use of asbestos, and a peak in incidence is expected within the next two decades due to the long latency period from asbestos exposure2. Exposure to asbestos dust/fibers occurs in a variety of ways. Washing clothes of a family member who worked with asbestos may be sufficient. Genetic predisposition appears to influence the greater occurrence of mesotheliomas in certain families.
MM arises from the mesothelial cells lining the pleura and peritoneum (30%), and rarely in the pericardium or tunica vaginalis3. Primarily exposure to the crocidolite variety has been implicated in the pathogenesis of this malignancy. However,about half of the reported cases do not have a history of asbestos exposure. Smaller particles of asbestos are more dangerous than the larger fiber, remaining suspended in the air where they can be inhaled, penetrating more easily and deeperinto the lungs. MPM usually has a shorter latency period but can vary from 10 to 50 years, with symptoms presenting in 20 - 30 years after asbestos exposure as compared to 30 – 40 years for pleural mesothelioma4. Probably inhaled asbestos fibers into the lung induce MPM through transport to the abdomen via the lymphatic system. Asbestos fibers also may be deposited in the gut after ingestion of asbestos contaminated sputum. Other possible risk factors are familial Mediterranean fever and ports of prior radiation1, or after thorium, talc, erionite or mica exposure, and also patients with diffuse lymphocytic lymphoma5.
Clinical history can be elusive, sometimes weight loss being the only clinical symptom present for a long time, delaying the diagnosis often until an advanced stage at the initial presentation. Besides abdominal pain/abdominal distention, MPM may manifest atypically with unusual paraneoplastic syndromes (dysfunctional hepatopathy, wasting syndrome), thrombocytosis, venous thrombosis, and hypoglycemia1.
]]> MPM is usually confined to the surfaces of the abdomen/pelvis, rarely penetrating through the diaphragm and involving pleural serosa, causing respiratory symptoms. In the majority of patients, hematogenous or lymphatic metastases to other sites usually do not occur, but metastases to the parasternal, retroperitoneal, mediastinal, axillary, supraclavicular and cervical lymph nodes, lung, bone, liver, and umbilical areas (Sister Mary Joseph’s nodule), have been reported5. Mortality is usually caused by intestinal obstruction, fistula formation, and starvation6.In addition to mesothelioma, exposure to asbestos increases the risk of lung cancer in smokers, asbestosis (a noncancerous, chronic lung ailment), and other cancers such as tumors of the larynx, and kidney4.
CASE REPORT
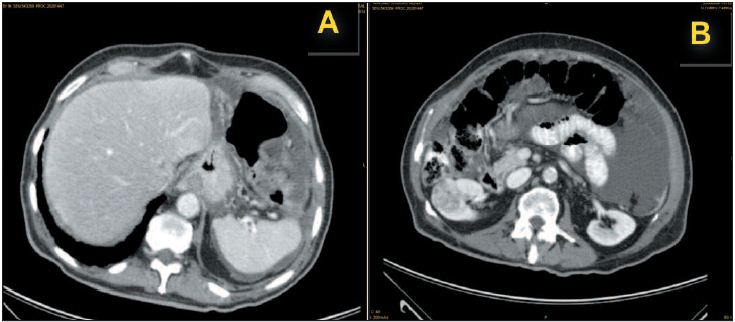
A 78-year-old man was admitted on August 8, 2005 due to massive ascites. He was apparently well until five months prior to admission (PTA), when he started to develop anorexia, asthenia, easy fatigability, progressive weight loss (15 kg), postprandial fullness and abdominal swelling. Several days PTA, he noticed ankle edema, decreasing urine output, frequent borborygmi and intermittent lower abdominal colic. One day PTA, he had one episode of vomiting. He denied any other symptom. During these months the following exams were performed: a chest R-ray revealing bilateral randomly distributed pleural calcifications, normal barium swallow and barium enema, and a non-informative abdominal ultrasonography. On questioning, he admitted to have been exposed to asbestos 40 years ago when he handled a lot of industrial bags that contained asbestos. On physical exam the patient was afebrile, BP 130/70 mmHg, HR 115 bpm, with no respiratory distress. His abdomen was very distended and tense with ascites. A vague mass on the right flank was identified. Liver and spleen did not appear to be enlarged and there was no abdominal collateral venous circulation or hernias. There were no signs of liver failure. Prostate was slightly enlarged. Blood tests revealed: Hb 10.0 g/dl, MCHC 33.2%; leukocytes 8.1x109/L (84.8% N); platelets 5.27x109/L; normal PT and APTT; reticulocytes 37.0x109/L (20.0 - 100.0); ESR 86 mm; PCR 14.5 mg/dL (<1.00); iron 14μg/dL (49 - 181); transferrin 129 mg/dL (190-375); ferritin 383 ng/mL (17.9 - 464.0); normal folic acid and vit. B12, albumin2.36 g/dL; alkaline phosphatase 165 U/L (38-126); γ-GT 91 U/L (15-73); other tests like glucose, BUN, creatinine, protein electrophoresis and immunoglobulins, bilirrubin, alanine ami notrans ferase and alanine aspartate transferase, LDH, amylase, cholesterol, triglycerides, calcium, phosphorus, ionogram, magnesium were normal. Serology for HBV, HCV and HIV were negative. Anti-nuclear, anti-smooth muscle and anti-mitochondrial antibodies were negative. Several malignant markers: PSA; CEA; CA 19.9; CA 15.3; α-FP were negative. Parentesis yielded 6000 ml of yellow citrine non-turbid peritoneal fluid revealing parameters of non-hypertensive portal ascites (SAAG < 0.737), with 0.2x109/L leukocytes (neutrophils 39.9%, lymphocytes 29%), erythrocytes 0.01x1012/L, absence of malignant cells, protein 3.20 g/dL, glucose 62 mg/dL, LDH 1.118 U/L. The thoracic CT scan revealed bilateral thickening and calcifications involving the anterior, posterior and diaphragmatic pleura, with neither pleural fluid nor tumoral nodules, and lungs were normal. The abdominal-pelvic CT scans showed massive ascites with contrast-enhancement of several peritoneal folds, together with heterogeneity and hypervascularity of the great omentum and of the left perigastric ligaments (Fig. 1A). These findings were in favor of peritoneal carcinomatosis.
There was evidence of a solid tumor with a diameter of 4 cm at the level of the lower third of the right kidney, with low-contrast enhancement relative to the normal adjacent parenchyma (Fig. 1B), and without evidence of either vascular invasion or of regional involvement.

Fig. 1. Intravenous contrast-enhanced abdominal-pelvic axial CT-scans, revealing: A) mesentery nodularity. B) Grawitz tumor of the right kidney and ascites.
]]> Ascites re-accumulated quickly necessitating a repeated paracentesis, which again showed the same laboratory characteristics. Nephrectomy of the right kidney (August 27 2008) revealed two nodular lesions characterized by microscopy as renal cell carcinoma, conventional type, nuclear anaplasia degree 1 (Fuhrman‘s classification) (Fig. 2), without invasion of the peri-renal fat and of the hilar vessels and adrenal gland. The pyelocaliceal tree and the ureter were normal and there was no thrombosis of the renal vein. Postoperatively there was a transient worsening of the renal function. A week after nephrectomy, there was re-accumulation of ascites and paracentesis was again performed, with the ascitic fluid showing the same laboratory characteristics. After the third paracentesis, hepatomegaly was noted through inspection and by abdominal palpation. Due to the abnormalities of the liver tests suggesting the possibility of intrahepatic cholestasis, a liver biopsy was undertaken, which did not reveal any abnormality. The patient was discharged symptomatically improved, but after ten days he was re-admitted due to re-accumulation of ascites. The peritoneal fluid was then suspicious of malignancy. We proceed to peritoneoscopy on September 16, 2008, which revealed “tumor” implants on the abdominal wall, specifically in the right hypochondrium and the right inguinal regions (it was not possible to see the liver due to adherence of the colon and great omentum). Although these findings were more in favor of peritoneal carcinomatosis, the microscopic examination showed MPM, epithelial type, characterized by tubular structures and gland-like spaces lined by cuboidal cells with vesicular nuclei and eosinophilic cytoplasm (Fig. 3 A/B). Immunohistochemistry study revealed the following data: CAM 5.2 +, EMA + (membranous pattern), Ck 7 +, Ck 20 -, calretinin +, CEAp -, CD15 -) (Fig. 4 A/B).
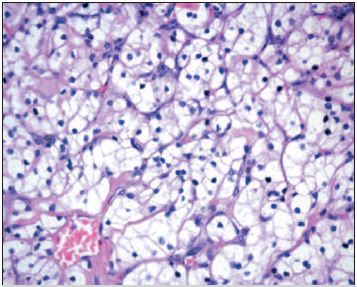
Fig. 2. HEx400: Clear renal cell renal carcinoma.
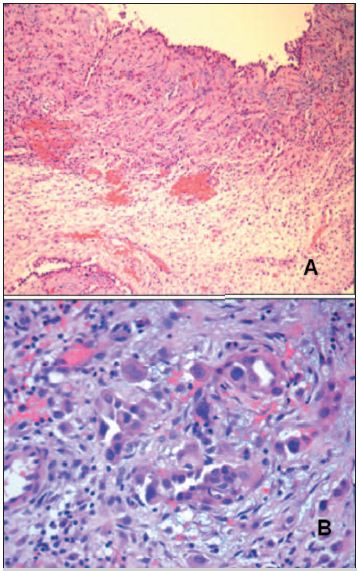
Fig. 3. Peritoneal biopsy - Malignant peritoneal mesothelioma: A) HEx100: tubular structures and gland-like spaces. B) HEx400: the cells are cuboidal with vesicular nuclei and eosinophil cytoplasm.
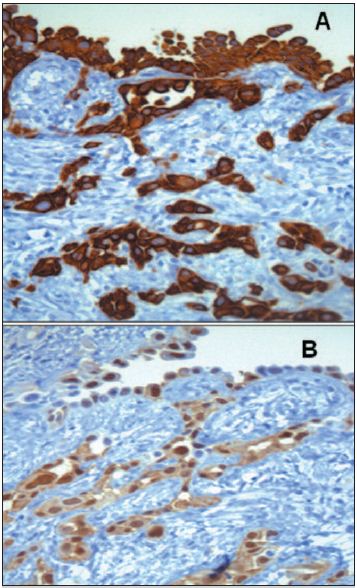
Fig. 4. Peritoneal biopsy – Malignant peritoneal mesothelioma: Immunohistochemistry study: A) 400x: CAM5.2 +. B) 400x: calretinin +
]]>With the diagnosis already confirmed, and with the patient’s age and general state of health in mind, the patient was considered not suitable to either traditional chemotherapy or cytoreductive surgery, and even less for hyperthermic intraoperative intraperitoneal chemotherapy. Apart from intermittent paracentesis for symptomatic relief of ascites, we tried to ameliorate anorexia and asthenia by administering thalidomide in increasing doses. However, we were not able to surpass a dosage of 200 mg per day, as the patient become progressively worse and died due to acute bronchopneumonia. The patient’s family did not consent to a necropsy.
DISCUSSION
Asbestos appears to act as a complete carcinogen with development of mesothelioma in sequential stages of initiation/ promotion. Hypothetically, asbestos fibers act through direct physical interactions with the mesothelium cells, in conjunction with effects in interaction with inflammatory macrophages cells, altering its function and secretory properties, ultimately creating conditions favoring the development of mesothelioma, through the generation of increased amounts of free radicals which promote asbestos carcinogenicity.
Asbestos also possess immunosuppressive properties. The interactions between asbestos fibers and chromosomic DNA, induce complex genetic abnormalities, most commonly on chromosome 22. In experimental mesothelioma cell lines deletion of tumor suppressor genes were identified4. Milky spots in the greater omentum and pelvic peritoneum have a role in tumor cell spread in the peritoneal cavity, being the sites to which tumor cells migrate preferentially from the peritoneal cavity, forming clusters within. Milky spots are minute organelles containing accumulations of macrophages, T and B lymphocytes around postcapillary venules, connected by lymphatics, covered by leaky mesothelial cells, and may be associated with mesothelioma metastases. Special peritoneal lymphatic orifices (lymphatic stomata) connect with subperitoneal lymphatic channel and milky spots. Intraperitoneal free cancer cells specifically deposit in the lymphatic stomata and proliferate in the submesothelial lymphatic space. Numerous stomata are detected on the undersurface of the diaphragm, small bowel mesentery, greater omentum, appendix epiploicae of the large bowel and the pelvic peritoneum. In contrast, there are no lymphatic stomata on the liver capsule, the surface of the spleen and the serosal surface of the small bowel and stomach. Accordingly, the serosal surface of these organs is involved only at the late phase of peritoneal dissemination7.
In MPM dissemination by cancer seeding and peritoneal fluid production, result in disease progression. It seems that fluid production is due to maintenance of a functional property of normal mesothelial cells in malignant cells. As the peritoneal fluid produced by mesothelial nodules increased, dissemination to sites of peritoneal resorption occurs, resulting in widespread progression of malignant cells on peritoneal surfaces and copious fluid production. In these patients the peritoneal space becomes a free conduit for mesothelioma cells to migrate from place to place. In producing of ascites fluid, the cancer cells provide themselves with a carrier solution to disseminate throughout the abdominal/pelvic spaces, explaining why patients with MPM often present with large volume of ascites8.
Sugarbaker et al6 described three patterns of clinical presentation of MPM: abdominal pain, abdominal distention, and a combined presentation. These patterns appear to correlate with the CT scan manifestations of MPM. The commonest manifestation is the “dry-painful” type - CT revealing enhancing soft-tissue multiple small masses in the peritoneum, omentum or peritoneal folds, or a single dominant mass in one quadrant of the abdomen, with little or no ascites. Less common is the “wet type”, associated with abdominal distention and ascites – the CT showing ascites associated with widespread small nodules and plaques, without a dominant mass (“debilitating ascites”). The third type is a combination of the previous two, in which the patients may present with pain and ascites. MPM has a tendency to spread along serosal surfaces and for direct invasion of both solid and hollow intra-abdominal organs, most commonly the colon and the liver. As the disease evolves, the tumoral nodules become more confluent plaque-like masses or looking omental “caking”. Occasionally, the initial presentation may be fever of unknown origin, night sweats, dysphagia, bowel obstruction, new onset hernia, deep vein thrombosis and arterial occlusion. However, as MPM is rare, we have to consider a radiologic differential diagnosis with other malignant disease of the abdomen, and also with diffuse benign diseases of the peritoneum3. Routine laboratory tests are not useful in making the diagnosis. The most useful serum marker for diagnosis and followup is the serum mesothelin-related protein (SMRP), which is elevated in more than 84% of mesotheliomas with a sensitivity of 60% at diagnosis9. SMRP is also helpful for screening asbestos-exposed individuals for early evidence of mesothelioma.
Confirmatory accurate diagnosis of MPM can be challenging, even though the radiologic CT scan imaging features can suggest this entity. Cytologic diagnosis of aspirated fluid obtained from paracentesis usually reveals an exudative process, but with low yield of malignant cells (slightly more than 50% of the cases), due to combination of marked difficulty in differentiating malignant mesothelial cells from reactive mesothelium and relatively small number of malignant cells within the fluid. Diagnostic accuracy increases with core sample size, which can be done through peritoneoscopy, and with immune-histochemistry study of the biopsy which is fundamental for the differential diagnosis1,5. However, laparoscopy can complicate the management by facilitating tumor dissemination to port sites, so limiting the puncture sites to along the “linea alba” are recommended6.
Mesotheliomas have three base histological forms: (1) epithelial (the most frequent), (2) sarcomatoid and (3) mixed (biphasic), according to the review by Weiss10. Positive stain markers for MPM are cytokeratin, vimentin, EMA (membranous pattern), Ck 5, calretinin, and thrombomodulin. The best negative markers are CEA, Ber-EP4, CD15 (Leu-M1), MOC-3. TTF1 and CA19.910. Electron microscopy may help in the diagnosis, revealing ultra-structural features of MM, as tall and thin microvilli on the cell surface, whose length exceeds the width by a margin of 15:111.
]]> Although there is no established staging system for MPM, these tumors may be staged in four categories using the TNM system: 1) a localized lesion able to be completely resected; 2) disease contained within the abdominal cavity on peritoneal and organ surfaces where debulking (the removal of as much, but not all of the tumor) is possible, 3) disease contained in the abdominal cavity with invasion of organs (e.g. colon, liver); 4) disease extending outside the abdominal cavity. Sebbag et al12 proposed other staging system for MPM – the TGM staging system, based on: 1) primary tumor burden, before resection, evaluated by Peritoneal Cancer Index (PCI), in which the sum of each peritoneal region lesion size scores gives the PCI. Lesion sizes scores are classified from 0: no tumor to 3.Prognosis of MPM varies with the tumor type, the epithelial one having a slightly longer survival than biphasic and sarcomatoid types. Nevertheless, the most reliable predictor of survival is the overall distribution of the disease. The prognosis is better for patients with solitary tumors compared with those with diffuse intra-abdominal disease6. Other significant predictive factors of survival are age and completeness of cytoreduction after treatment. Regarding treatment of MPM, a large majority of patients with MPM are first diagnosed with a large volume of disease diffusely spread throughout the abdomen/pelvis, mostly at sites of peritoneal fluid resorption and at gravity dependent sites. The small bowel surfaces and mesenteries are common sites of peritoneal implants. But as the tumor is usually confined to the peritoneal cavity at the time of initial diagnosis, remaining there for much or every of the subsequent clinical course, multimodality therapeutic regimens combining early aggressive local/regional cytoreductive surgery of as much tumor as possible and intraperitoneal chemotherapy should be pursued to obtain a better prognosis, and complemented by early postoperative chemotherapy and immunotherapy2. Cisplatin is the most frequently chemotherapeutic agent used, usually in association with pemetrexed8. The score of residual peritoneal seeding after cytoreductive peritonectomy is the principal prognostic indicator, but usually the results are disappointing5.
In selected patients able to support it, hyperthermic intraoperative intraperitoneal chemotherapy (HIPEC), followed by early postoperative intraperitoneal chemotherapy, is a good strategy for a much longer survival - 50% of patients alive at 5 years, in cases of epithelial type of MPM8,13. The mortality rate of HIPEC ranges between 0% and 11%, and most cases of death are due to bowel perforation, bone marrow suppression, respiratory failure, methicillin resistant Staphylococcus aureus infection, and pulmonary embolism. Those patients not suitable for curative resection have a median survival of only 6 to 9 months.
Targeted therapies are monoclonal antibodies and drugs targeting vascular endothelial growth factor, platelet-derived growth factor and epidermal growth-factor, disturbing the growth of MPM2. Thalidomide is one of such drugs administered at a daily dose of 100 mg per day, increasing the daily dose weekly by 100 mg to a maximum dose of 500 mg per day, as tolerated. Thalidomide appears to be useful in controlling the distressing profuse sweating that often accompanies end stage cancer, but without obtaining tumor shrinking14. In our patient thalidomide was not useful. Stauffer’s syndrome, one of several frequently associatedprecursor paraneoplastic syndromes of primary or recurrent disease in renal cell cancer (RCC), characterized by hepatosplenomegaly, fever and weight loss, biochemical abnormalities of liver function tests (alkaline phosphatase and γ-glutamyltransferase), and pathologic changes consistent with non-specific hepatitis and absence of liver metastases, was considered in our patient. Notwithstanding, ascites was not described in Stauffer’s syndrome and in our patient biochemical abnormalities persisted after nephrectomy15. Paraneoplastic cholestatic hepatic dysfunction, frequently associated with fever, sweating, wasting and severe weight loss, has been described in MPM2. In our patient “apparent” hepatomegaly was noted through inspection and by palpation only after massive drainage of ascites. Biochemical abnormalities were in favor of liver dysfunction characterized by cholestasis. However, imaging by CT scan did not revealed the presence of liver metastases, which was later confirmed by a normal liver biopsy. To our knowledge, the synchronous occurrence of MPM and RCC has not yet been described. The prior exposure to asbestos in our patient is in favour of a common oncogenic etiology in his case.
ACKNOWLEDGMENTS
To Drs JM Coutinho and P Tavares for the realization of peritoneoscopy; to Dr Cidália Eira for the realization of abdominalpelvic CT scan; to Dr M Abecassis for the surgical advise about hyperthermic intraoperative intraperitoneal chemotherapy.
REFERENCES
1. Bani-Hani KE, Gharaibeh KA. Malignant peritoneal mesothelioma. J Surg Oncol 2005;91:17-25.
]]> 2. Garcia-Carbonero R, Paz-Ares L. Systemic chemotherapy in the management of malignant peritoneal mesothelioma. Eur J Cancer Surg 2006;32:676-681.3. Busch JM, Kruskal JB, Wu B. Best cases from the AFIP – Malignant peritoneal mesothelioma. Radiographics 2002;22:1511-1515. [ Links ]
4. http://en.wikipedia.org/wiki/Mesothelioma Acesseed on 25 September 2008
5. Brida A, Padoan I, Mencarelli R, et al. Peritoneal mesothelioma: a review. Medscape General Medicine 2007;9:32. Published on line 2007 May 10. http://www.pubmedcentral.nih.gov/articlerenderFcgi?artid=1994863
6. Sugarbaker PH, Acherman YI, Gonzalez-Moreno S, et al. Diagnosis and treatment of peritoneal mesothelioma: the Washington Cancer Institute Experience. Seminars in Oncology 2002;29:51-61.
7. Hassan R, Alexander R, Antman K, et al. Current treatment options and biology of peritoneal mesothelioma: meeting summary of the first NIH peritoneal mesothelioma conference. Annals Oncol 2006;17:1615-1619.
8. Sugarbaker PH. Sugarbaker Oncology Associates. Specialty Section for the Treatment of Peritoneal Mesothelioma. http://www.surgicaloncology.com/meso.htm (Accessed in August 2010)
9. Robinson BW, Creanej J, Lake R, et al. Mesothelin-family proteins and diagnosis of mesothelioma. Lancet 2003;362:1612-1616.
10. Weiss SW, Goldblum JR. Mesothelioma. In Weiss S, Goldblum JR, ed. Enzinger and Weiss Soft Tissue Tumors 4th ed. St Louis: Mosby 2001:1063-1110.
11. Ordonez NG. The diagnostic utility of immunohistochemistry and electron microscopy in distinguishing between peritoneal mesotheliomas and serous carcinomas: a comparative study. Mod Pathol 2006;19:34-38.
]]> 12. Sebbag G, Sugarbaker PH. Peritoneal mesothelioma proposal for a staging system. Eur J Surg Oncol 2001;27:223-224.13. Stewart JH 4th, Shen P, Levine EA. Intraperitoneal hyperthemic chemotherapy for peritoneal surface malignancy: current status and future directions. Ann Surg Oncol 2005;12:765-777.
14. Deaner PB. The use of thalidomide in the management of severe sweating in patients with advanced malignancy: Trial report. Palliative Medicine 2000;14:429-431.
15. Giannakos G, Papanicolaou X, Trafalis D, et al. Stauffer’s syndrome variant associated with renal cell carcinoma. Int J Urol 2005;12:757-759.
Correspondência: António Murinello; Avª Engº Antº Azevº Coutinho, Lte 8 r/c dto, 2750 Cascais. Portugal; E-mail: amurinello@gmail.com; Tel: +351 214 865 061; Tlm: +351 918 626 874.
Recebido para publicação: 12/05/2009 e Aceite para publicação: 28/12/2009.
]]>