10mm in 78.3% of the HCWs. The proportionof positive IGRA results increased with the TST diameter. In those with a TST >15mm, 49.2% were IGRA positive. TST was more than twice as often positive than the IGRA. Therefore, TST+/IGRA- results were more often observed than concordant negative or positive results. In none of the HCWs with a TST+/IGRA- result active TB was diagnosed during the study period. Repeated BCG vaccination increased the number of TST+/IGRA- discordance. The smaller the interval after BCG vaccination, the higher was the TST+/IGRA- discordance. In the screened HCWs population, active TB was diagnosed in 9. At the time of diagnosis TST and IGRA were positive in all active TB cases. The study period covers 24 months, therefore the average annual incidence rate was 268/100 000. TB burden in HCWs in Portugal is high. Considering the limitations that TST and IGRA present, the best solution seems to be the use of both, using the IGRA higher specificity for confirming a positive TST, taking advantage of the best characteristics of each test.]]>
10mm em 78.3% dos PS. A proporção de resultados IGRA positivos aumentou com o diâmetro da PT. Nos PS com PT >15mm, 49.2% são IGRA positivos. A PT foi positiva em mais do dobro dos casos do IGRA. Assim, foram observados mais frequentemente resultados PT+/IGRA- do que resultados concordantes negativos ou positivos. Em nenhum PS com resultado PT+/IGRA- foi diagnosticada tuberculose activa durante o período do estudo. A vacinação repetida pelo BCG aumentou o número de casos discordantes PT+/IGRA -. Quanto menor o intervalo após a vacinação pelo BCG, maior a discordância PT+/IGRA -. Na população de PS rastreada, foram diagnosticados 9 casos de tuberculose activa, sendo todos PT e IGRA positivos na altura do diagnóstico. Este estudo durou 24 meses, pelo que a taxa de incidência anual média foi de 268/100 000. A tuberculose é um problema importante nos PS em Portugal. Considerando as limitações que a PT e IGRA apresentam, a melhor solução parece ser o uso de ambos, utilizando a maior especificidade do IGRA para confirmar uma PT positiva, aproveitando as melhores características de cada teste.]]>
José Torres Costa 1,2,3, Rui Silva 1,2,3, Raul Sá 1, Maria João Cardoso 4, Carla Ribeiro 1, Albert Nienhaus 5
1 Occupational Health Division
2 Allergy and Clinical Immunology Division, Hospital São João, Alameda Professor Hernâni Monteiro, PortoPortugal
3 Faculty of Medicine, Porto University, Alameda Professor Hernâni Monteiro, Porto-Portugal
4 Hospital São João, Clinical Pathology Division, Alameda Professor Hernâni Monteiro, PortoPortugal
5 Accidents Insurance and Prevention in the Health and Welfare Services, Hamburg, Germany
]]> Correspondência
Abstract
Healthcare workers (HCWs) have an increased risk of tuberculosis (TB). Screening for latent tuberculosis infection and active TB is therefore essential in infection control programs. Tuberculin skin test (TST) and Interferon-Á Release Assay (IGRA) were used simultaneously in 1686 HCWs between May 2007 and April 2009. A chest X-ray was performed in order to exclude active TB when TST was .10mm or IGRA was positive and in HCWs with TB contact or symptoms. IGRA was positive in 33.1% and TST was >10mm in 78.3% of the HCWs. The proportionof positive IGRA results increased with the TST diameter. In those with a TST >15mm, 49.2% were IGRA positive. TST was more than twice as often positive than the IGRA. Therefore, TST+/IGRA- results were more often observed than concordant negative or positive results. In none of the HCWs with a TST+/IGRA- result active TB was diagnosed during the study period. Repeated BCG vaccination increased the number of TST+/IGRA- discordance.
The smaller the interval after BCG vaccination, the higher was the TST+/IGRA- discordance.
In the screened HCWs population, active TB was diagnosed in 9. At the time of diagnosis TST and IGRA were positive in all active TB cases. The study period covers 24 months, therefore the average annual incidence rate was 268/100 000.
TB burden in HCWs in Portugal is high. Considering the limitations that TST and IGRA present, the best solution seems to be the use of both, using the IGRA higher specificity for confirming a positive TST, taking advantage of the best characteristics of each test.
Key-words: Tuberculosis, healthcare workers, tuberculin skin test, interferon-γ release assay, Portugal.
Comparação do teste de libertação do interferão γ e da prova de tuberculina no rastreio de profissionais de saúde
]]> ResumoOs profissionais de saúde (PS) têm um risco aumentado de tuberculose. O rastreio da tuberculose latente e da tuberculose activa é portanto essencial nos programas de controlo de infecção. Entre Maio de 2007 e Abril de 2009, foram utilizados simultaneamente a prova de tuberculina (PT) e o teste de libertação do interferão-ã (IGRA) em 1686 PS. Quando PT ≥10mm ou IGRA positivo, e em PS com contacto com tuberculose ou sintomáticos, foi realizada uma radiografia torácica para excluir tuberculose activa. O IGRA foi positivo em 33,1% e a PT foi >10mm em 78.3% dos PS. A proporção de resultados IGRA positivos aumentou com o diâmetro da PT. Nos PS com PT >15mm, 49.2% são IGRA positivos. A PT foi positiva em mais do dobro dos casos do IGRA. Assim, foram observados mais frequentemente resultados PT+/IGRA- do que resultados concordantes negativos ou positivos. Em nenhum PS com resultado PT+/IGRA- foi diagnosticada tuberculose activa durante o período do estudo. A vacinação repetida pelo BCG aumentou o número de casos discordantes PT+/IGRA -. Quanto menor o intervalo após a vacinação pelo BCG, maior a discordância PT+/IGRA -.
Na população de PS rastreada, foram diagnosticados 9 casos de tuberculose activa, sendo todos PT e IGRA positivos na altura do diagnóstico. Este estudo durou 24 meses, pelo que a taxa de incidência anual média foi de 268/100 000.
A tuberculose é um problema importante nos PS em Portugal. Considerando as limitações que a PT e IGRA apresentam, a melhor solução parece ser o uso de ambos, utilizando a maior especificidade do IGRA para confirmar uma PT positiva, aproveitando as melhores características de cada teste.
Palavras-chave: Tuberculose, profissionais de saúde, prova de tuberculina, teste de libertação do interferão-γ, Portugal.
Introduction
The increased risk of healthcare workers (HCWs) for tuberculosis is well established1-3.
Screening HCWs for latent tuberculosis infection (LTBI) and active tuberculosis (TB) is therefore fundamental in infection control programs in hospitals4. For about a century, the tuberculin skin test (TST) has been used to detect LTBI. The TST measures the hypersensitive response to purified protein derivative (PPD), a crude mixture of antigens,many of which are shared by M. tuberculosis, M. bovis (source of the Calmette -Guérin bacillus;BCG), and several nontubercular mycobacteria (NTM). Although the TST has proved to be useful in clinical practice, it has known limitations, including crossreactivity with BCG and NTM infections5.
Advances in molecular biology have led to the development of a new in vitro assay that measures interferon (INF)-ã released by sensitized T cells after stimulation with M. tuberculosis antigens. These antigens include early secreted antigenic target (ESAT) -6 and culture filtrate protein (CFP) -10. ESAT -6 and CFP -10 are encoded by genes located within the region of difference (RD) -1 segment of the M. tuberculosis genome. Two new T-cell-based tests for diagnosing LTBI have been developed and licensed for commercial distribution: QuantiFERON (QTF)- TB Gold in Tube® (Cellestis, Victoria, Australia) uses an enzyme-linked immunosorbent assay (ELISA) to measure antigenspecific production of INF -ã by circulating T cells in whole blood; T -SPOT.TB® (Oxford Immunotec, Oxford, United Kingdom) uses the Elispot technique to measure peripheral blood mononuclear cells that produce INF -ã. These tests are more specific than the TST using PPD, because they use antigens not shared by any of the BCG vaccine strains nor by the more common species of NTM (e.g. M. avium)6. Research on test performance has shown that assays using RD1 antigens are more specific than TST, have a better correlation with surrogate measures of exposure to M. tuberculosis in low incidence settings, and exhibit less cross-reactivity due to BCG vaccination than the TST7,8. In the absence of a gold standard for LTBI, active TB is used as a surrogate for LTBI to judge sensitivity. In a meta -analysis, the pooled sensitivity and specificity for the IGRA were higher than for TST9. The IGRA has potential advantages besides its greater specificity, including logistical convenience (no second patient contact for reading the test), easier interpretation of the test results (cut-off point is independent from the risk status of the patient) and the ability to perform serial testing without inducing the boosting phenomenon. A recent study demonstrated that the IGRA has merits in screening close contacts for LTBI in low incidence areas10. Within the first two years after contact, the progression rate to active TB for IGRA -positive was three times higher than with TST positive contacts.
]]> So far only few systematic investigations of LTBI in HCWs using the IGRA have been published11-17. Therefore, we studied the prevalence of LTBI in Portuguese HCWs and compared the performance of the IGRA to the TST.
Materials and methods
Study setting and study subjects
In compliance with EU regulations, the Hospital S. João, Porto Portugal, implemented an Occupational Health Division for the hospital staff. Since May 2007 the workers of this hospital have been offered TB screening with TST and IGRA simultaneously by this Division. Screening follows the Centers for Disease Control and Prevention (CDC) guidelines4. Upon commencement of employment, all workers are examined to exclude active TB and to assess their pre-employment status. Depending on risk assessment, the examination is repeated annually or every other year. HCWs with close patient contact in the infection and TB wards are considered at high risk, workers with regular patient contact in the other wards are considered at medium risk and workers with no regular patient contact or no contact with biological material are considered at low risk. Screening is performed annually for those with contact with TB patients or infectious material. For all other HCWs screening is scheduled biannually. After unprotected contact with na infectious patient or material, an additional screening is performed as well.
The data presented here comprise all HCWs screened between May 2007 and April 2009 using TST and IGRA simultaneously. TST was only performed when the diameter of a previous TST was below 15 mm or when no previous TST result was known. A chest X-ray was performed in order to exclude active pulmonary disease when TST was 10 mm or higher or IGRA was positive and in HCWs with TB contact or symptoms.
BCG vaccination was assessed through the individual vaccination register. If no register was available, vaccination status was verified by scars. According to the Portuguese National Vaccination Plan18, BCG vaccination is administered in newborns, and until January 2000 was repeated if TST diameter was below 5 mm. Therefore, every HCW was considered to have been vaccinated at least once.
Tuberculin skin test
TST was performed by trained personnel following standard procedures. Briefly, 0.1 mL (2 TU) of purified protein derivate (PPD, RT23; Statens Serum Institute, Copenhagen, Denmark) were injected in the volar side of the forearm of the participants and read 72 to 96 hours afterwards. The transverse diameter of the induration was measured by experienced personnel.
]]> Before the TST application, the interview was performed and blood for the IGRA was drawn. For the IGRA, the QuantiFERON- -TB® Gold In -Tube Assay (Cellestis Limited, Carnegie, Australia) was used. This whole blood assay uses overlapping peptides corresponding to ESAT -6, CFP -10, and a portion of tuberculosis antigen TB7.7 (Rv2654).Stimulation of the antigenic mixture occurs within the tube used to collect blood. Tubes were incubated at 37oC overnight before centrifugation, and INF-ã release was measured by ELISA following the protocol of the manufacturer. All assays performed met the manufacturers quality control standards.
The test was considered positive when INF -ã was ≥ 0.35 IU after correction for the negative control. Observers were blinded to the results of the TST results.
Statistical analysis
Chi-square test was used to compare frequencies of test results among different groups of participants. For ordered risks, the proportions of positive test results were compared using the chi-square test of trend.
The agreement between TST and QFT independent from the agreement by chance alone was assessed by calculating Kappa values for TST >10 mm and TST >15 mm. P<0.05 was considered statistically significant. Adjusted odds ratios (OR) and 95% confidence intervals (CI) were calculated for different putative predictive variables using conditional logistic regression. Model building was performed backwards using the chance criteria for variable selection19.
Data analysis was performed with SPSS, Version 14 (SPSS Inc., Chicago, Illinois).
All people gave their informed consent prior to their inclusion in the study. No additional data were collected for the study purpose only and analysis was performed with anonymous data. Therefore, no endorsement by an ethics committee was required.
]]> Results
TST and QFT were performed in 1686 HCWs. The population available for analysis comprises 1682 HCWs since QFT was undetermined in 4 (0.2%). Their characteristics are described in Table I. The TST was >10 mm in 78.3% and the QFT was positive in 33.1%. The proportion of positive QFT results increase with the diameter of the TST (Table II). In those with a TST >15 mm, 49.2% are positive in the QFT. Agreement between TST and QFT was higher with >15 mm instead of >10 mm as cut off for TST (0.18 versus 0.29). For both cut offs agreement decreased with number of BCG vaccination (Table III), but even in those vaccinated at birth only, Kappa was low for both the >10 mm (0.26) and for the >15 mm (0.37) cut off for TST. Moving the cut off for TST from >10 to >15 mm did not yield good agreement between QFT and TST (Table III) and as a drawback increased the number of TST -/QFT+ discordant results from 33 (2%) to 202 (12%) as can be calculated from Table II. Therefore, further analysis were undertaken with TST >10 mm as cut off.
Table I - Study population for comparison of IGRA with TST (n=1682)
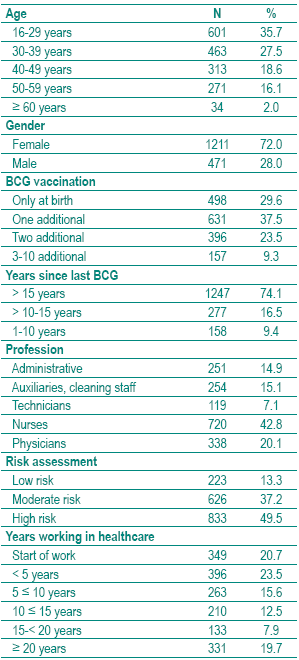
Table II - TST diameter by IGRA results
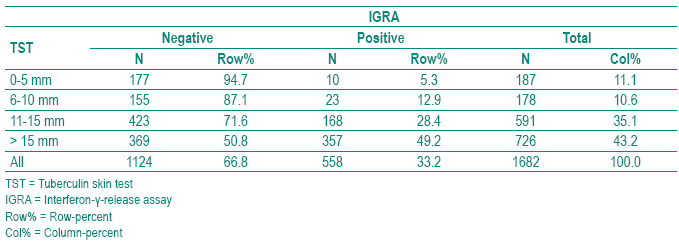
Table III -Agreement assessed by Kappa between TST and QFT depending on number of BCG vaccinations and cut off for TST
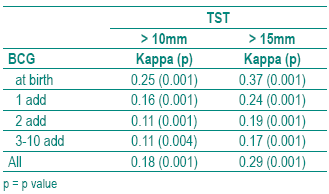
With age the proportion of positive TST or QFT increased (Table IV). In those 60 years or older the proportion of positive TST declined compared to those 40-40 years or 50-59 years old while for the QFT a linear association was seen over all age groups. Repeated BCG vaccination was not associated with the probability of a positive TST but decreased the proportion of positive QFT. When a BCG vaccination was performed during the last 10 years, the odds ratio for a positive TST was increased with a borderline statistically significant confidence interval (OR 1.5; 95%CI 1.0 -2.4) and the odds ratio for a positive QFT was well below 1 (OR 0.4; 95%Ci 0.3 -0.7).
]]>Table IV Proportion of tuberculin skin test (TST) >10 mm and positive interferon-ã-release assay (IGRA) and the respective adjusted odds ratios (OR) and 95% confi dence intervals (CI) for different putative risk factors
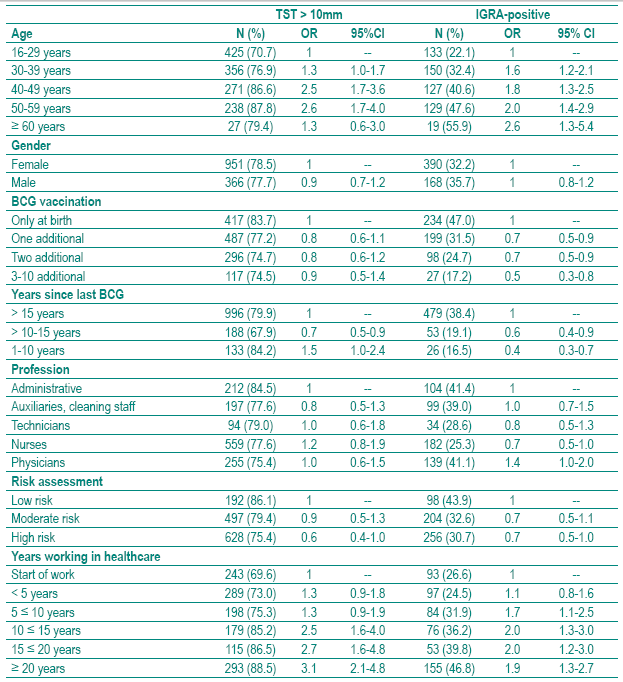
Profession was not associated with the TST, while doctors had a slightly increased odds ratio for a positive QFT (OR 1.4; 95%CI 1.0- 2.0), again with a borderline statistically significant confidence interval. Risk assessment, according to the CDC guidelines4, was neither associated with TST nor with QFT while a positive association was found between years in healthcare and the TST as well as the QFT.
Due to colliniarity problems age and years working in healthcare could not be introduced in the same model. Therefore, the effect of these two variables on TST or QFT could not be distinguished.
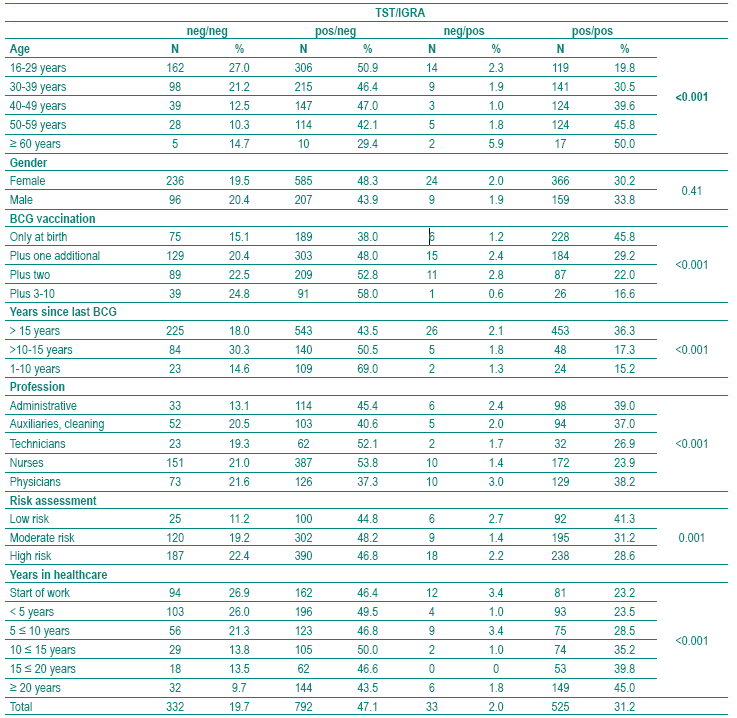
Most often TST+/QFT results (47.1%) followed by concordant positive results (31.2%) occurred (Table V). With age the number of concordant positive results increased while the proportion of TST+/QFT discordant results decreased. Repeated BCG vaccination increased the number of TST+/QFT discordance. The smaller the interval after BCG vaccination the higher was the proportion of TST+/QFT discordance. Nurses had less often positive concordant results than administratives, auxiliaries and physicians. At the same time, they had the highest rate of TST+/QFTdiscordance. Surprisingly, the higher the assumed risk of infection the lower was the proportion of TST+/QFT+ concordance. Years working in healthcare increased the probability of TST+/QFT+ concordance but were not associated with TST+/QFT discordance.
Table V Concordant and discordant tuberculin skin test (TST) >10 mm and interferon-ã-release assay (IGRA) results depending on putative risk factors
In these 1682 HCWs for whom TST and QFT results are available, active TB was dia gnosed in 9 HCWs. Diagnosis was based on culture confirmed positive smear in 7 cases, on culture in 1 and on PCR also in 1.
]]> At the time of diagnosis TST and QFT were positive in all active TB cases. The study period covers 24 month, therefore the average annual incidence rate was 268/100 000.
Discussion
The study presented here is one of the largest European studies analyzing risk factors for LTBI by using QFT and TST simultaneously. It is also the only crosssectional study that observed active TB in HCWs in a cross-sectional design. The agreement between TST and QFT was low and could not be improved by increasing the cut off for the TST from >10 to >15 mm because this increased the proportion of TST -/QFT+ results in the whole population from 2% to 12%. TST was more than twice as often positive than the QFT. Therefore, TST+/ QFT results were more often observed than concordant negative or positive results.
In none of the HCWs with a TST+/QFT result active TB was diagnosed during the study period.
So far little is known about the effect of repeated BCG vaccination on the probability of TST+/IGRA discordant results20,21. According to the Portuguese National Vaccination Plan, BCG vaccination is administered in newborns18. Therefore, the effect of BCG vaccination on TST and QFT could not be studied. Nevertheless it could be shown that repeated BCG vaccination increased the probability of TST+/QFT results. When re -vaccination was performed during the last 10 years the chance for discordant TST+/ QFT results was highest (69%) while at the same time probability of concordant positive results was low (15.2%).
Contrary to our preliminary analysis with a smaller sample not only QFT but also TST showed that years working in healthcare are a risk factor for LTBI22. Surprisingly, neither risk assessment nor profession was associated with TST or IGRA. In the two European fingerprint studies, the majority of workrelated active TB cases occurred when the infection risk was not suspected and preventive measures were not taken23,24. Rotation of the staff is another explanation for the lack of this association, as well as the crosssectional design of our study, which might dilute the expected association.
The high proportion of TST positive HCWs with repeated BCG vaccination corroborates the conclusion of other authors that TST is not very helpful in populations in which most people are vaccinated and in which vaccination is repeated25. In those populations the QFT is a promising alternative even though it is still not clear how the QFT will perform in serial testing.
While unlike with the TST26, the test results cannot be influence by previous QFT, the definition for conversions and reversions of the QFT are not yet well established. Further research will have to elucidate the question about how changes in interferon-ã concentration over time indicate real new infections or clearance of the Mycobacteria can be distinguished from changes in concentration due to natural variation27,28.
Additionally, and despite our increasing knowledge, several key questions about latent infection and reactivation of M. tuberculosis remain unanswered. Particularly, it should be noted that both the TST and the IGRA are designed to identify an adaptive immune response against M. tuberculosis, but not necessarily a latent infection. A positive result of currently available diagnostic tests is primarily a measure of an immunological response to stimulation by mycobacterial antigens that should not, therefore, be equated with the presence of live M. tuberculosis in the human host. The proportion of individuals who truly remain infected with M. tuberculosis after TST or IGRA conversion is unknown. It is also uncertain how long adaptive immune responses towards mycobacterial antigens persist in the absence of live mycobacteria. For these reasons, according to the recently published TBNET consensus statement regarding latent TB, based on the informative value presently derived by IGRA and TST, the term latent infection would at best implicate lasting tuberculosis immune responses and not necessarily identify a true latent infection with viable microorganisms and potential risk of developing active disease29,30. Further studies are needed. Portuguese LTBI treatment guidelines31, which were revised in 2006, need to be updated, in order to address this question. Whether treatment can be monitored by follow-up of the interferon-ã release is another interesting question to be studied in future32.
]]> Considering the limitations that both tests present, the best solution seems to be the use of both, using the IGRA higher specificity for confirming a positive TST. This option allows us to take advantage of the best characteristics of each test30.
Bibliography
1. Saleiro S, Santos AR, Vidal O, Carvalho T, Torres Costa J, Marques JA. Tuberculosis in hospital department health care workers. Rev Port Pneumol 2007; 13(6):789-799. [ Links ]
2. Seidler A, Nienhaus A, Diel R. Review of epidemiological studies on the occupational risk of tuberculosis in low-incidence areas. Respiration 2005; 72(4):431-446.
3. Menzies D, Joshi R, Pai M. Risk of tuberculosis infection and disease associated with work in health care settings. Int J Tuberc Lung Dis 2007; 11(6):593-605.
4. CDC Center for Disease Control and Prevention. Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Healthcare Settings, 2005. MMWR 2005; 54(RR-17):1-141.
5. Menzies D. What does tuberculin reactivity after Bacille Calmette-Guerin vaccination tell us? Clin infect Dis 2000; 31(Suppl 3):S71-S74.
6. Andersen P, Munk ME, Pollock JM, Doherty TM. Specific immune-based diagnosis of tuberculosis. Lancet 2000; 356(9235):1099-1104.
7. Nahid P, Pai M, Hopewell PC. Advances in the diagnosis and treatment of tuberculosis. Proc Am Thorac Soc 2006; 3:103-110.
]]> 8. Pai M, Riley LW, Colford JM. Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis 2004; 4:761-776.9. Menzies D, Pai M, Comstock G. Meta-analysis: New tests for the diagnosis of latent tuberculosis infection: Areas of uncertainty and recommendations for research. Ann Intern Med 2007; 146:340-352.
10. Diel R, Loddenkemper R, Meywald -Walter K, Niemann S, Nienhaus A: Predictive value of a whole blood IFN -{gamma} assay for the development of active TB disease. Am J Respir Crit Care Med 2008; 177:1164-1170.
11. Harada N, Nakajima Y, Higuchi K, Sekiya Y, Rothel J, Mori T. Screening for tuberculosis infection using whole-blood interferon-ã and Mantoux testing among Japanese healthcare workers. Infection Control and Hospital Epidemiology 2006; 27(5):442-448.
12. Soberg B, Andersen AB, Larsen HK, Weldingh K, Andersen P, Kofoerd K, Ravn P. Detecting a low prevalence of latent tuberculosis among health care workers in Denmark detected by M. tuberculosis specific INF-ã whole-blood test. Scandinavian Journal of Infectious Diseases 2007; 39:554-559.
13. Kobashi Y, Obase Y, Fukuda M, Yoshida K, Miyashita N, Fujii M, Oka M. Usefulness of QuantiFERON TB-2G, a diagnostic method for latent tuberculosis infection, in a contact investigation of health care workers. Intern Med 2007; 46(18):1543-1549.
14. Mirtskhulava V, Kempker R, Shields KL, Leonard MK, Tsertsvadze T, del Rio C, Salakaia A, Blumberg HM. Prevalence and risk factors for latent tuberculosis infection among health care workers in Georgia. Int J Tuberc Lung Dis 2008; 12(5):513-519.
15. Nienhaus A, Schablon A, Siano B, le Bacle C, Diel R. Evaluation of the interferon-gamma release assay in healthcare workers. Int Arch Occup Environ Health 2008; 81:295-300.
16. Schablon A, Beckmann G, Harling M, Diel R, Nienhaus A. Prevalence of latent tuberculosis infection among healthcare workers in a hospital for pulmonary diseases. J Occup Med Toxicol 2009; 4:1.
17. Vinton P, Mihrshahi S, Johnson P; Jenkin GA; Jolley D, Biggs BA. Comparison of QuantiFERON-TB gold in-tube test and tuberculin skin test for identification of latent Mycobacterium tuberculosis infection in healthcare staff and association between positive test results and known risk factors for infection. Infection Control and Hospital Epidemiology 2009; 30(3), online first.
]]> 18. Programa Nacional de Vacinação. Direcção-Geral de Saúde Ministério da Saúde. Available at http://www.dgsaude.pt/upload/membro.id/ficheiros/i007442.pdf (accessed May 1st, 2009)19. Hosmer D, Lemeshow S. Applied logistic regression. New York, NY: John Wiley & Sons 2000.
20. Farhat M, Greenaway, Pai M, Menzies D. Falsepositive tuberculin skin tests: what is the absolute effect of BCG and non-tuberculous mycobacteria? Int J Tuberc Lung Dis 2006; 10(11):1192-1204.
21. Nienhaus A, Schablon A, Diel R. Interferon -ã release assay for the diagnosis of latent TB infectionanalysis of discordant results, when compared to the tuberculin skin test. PLoS ONE 2008; 3(7):e2665.
22. Torres Costa Torres Costa J, Sá R, Cardoso MJ, Silva R, Ferreira J, Ribeiro C, Miranda M, Plácido JL, Nienhaus A. Tuberculosis screening in Portuguese healthcare workers using the tuberculin skin test and the interferon-ã release assay. Eur Resp J 2009 (in press).
23. Diel R, Seidler A, Nienhaus A, Rusch-Gerdes S, Niemann S. Occupational risk of tuberculosis transmission in a low incidence area. Respir Res 2005; 6(1):35-45.
24. De Vries G, Sebek MM, Lambregts-van Weezenbeek CS. Health-care workers with tuberculosis infected during work. Eur Respir J 2006; 28(6): 1216-21
25. Lee S S-J, Liu Y-C, Huang T-S, Chen Y-S, Tsai H-C,Wann S-R, Lin H-H. Comparison of the interferon-ã release assay and the tuberculin skin test for contact investigation of tuberculosis in BCG-vaccinated health care workers. Scandinavian Journal of Infectious Disease 2008; 40(5): 373-380.
26. Menzies D. Interpretation of repeated tuberculin tests. Boosting, conversion, and reversion. Am J Respir Crit Care Med 1999; 159:15-21.
27. Pai M, Joshi R, Dogra S, Zwerling AA, Gajalakshmi D, Goswami K, Reddy MVR, Kalantri S, Hill PC, Menzies D, Hopewell PC. T-cell assay conversions and reversions among household contacts of tuberculosis patients in rural India Int J Tuberc Lung Dis 2009; 13(1):84-92.
]]> 28. Yoshiyama T, Harada N, Higuchi K, Nakajima Y, Ogata H. Estimation of incidence of tuberculosis infection in healthcare workers using repeated interferon-ã assays. Epidemiol Infect 2009 (ahead of print).29. Mack U, Migliori GB, Sester M, Rieder HL, Ehlers S, Goletti D, et al, for the TBNET. LTBI: latent tuberculosis infection or lasting immune responses to M. tuberculosis? A TBNET consensus statement. Eur Respir J 2009; 33: 956-973.
30. Duarte R. Teste tuberculínico. Como optimizar? Rev Port Pneumol 2009; 15 (2):295-304. [ Links ]
31. Duarte R, Amado J, Lucas H, Sapage JM, Comissão de Trabalho de Tuberculose da Sociedade Portuguesa de Pneumologia. Tratamento da tuberculose latente: Revisão das normas, 2006. Rev Port Pneumol 2007; 13(3): 397-418 [ Links ]
32. Pai M, Joshi R, Dogra S, Mendiratta DK, Narang P, Dheda K Kalantri S. Persistently elevated T cell interferon -ã response after treatment for latent tuberculosis infection among healthcare workers in India: a preliminary report. Journal of Occupational Medicine and Toxicology 2006; 1:7.
Correspondência/Correspondence to:
José Castela Torres Costa
Estrada de St.ª Luzia 269
4900-408 Viana do Castelo
]]> e-mail: zecatoco@sapo.pt
Recebido para publicação/received for publication:09.06.05
Aceite para publicação/accepted for publication:09.07.23
]]>