0.30 (85 vs 5 days, respectively in BPD and no BPD patients, p<0.001). Comments: The most relevant risk factors were low birth weight, severe hyaline membrane disease, duration of respiratory support and oxygen therapy, and nosocomial sepsis. The implementation of potentially better practices to reduce lung injury in neonates must be addressed to improve practices to decrease these risk factors.]]>
0,30 (85 vs 5 dias, respectivamente nos doentes com e sem DBP, p<0,001). Comentários: Os factores de risco de DBP mais relevantes foram o baixo peso, a doença da membrana hialina grave, a duração da ventilação mecânica e da oxigenoterapia e a sépsis. A implementação das boas práticas para reduzir a lesão pulmonar nos RN deve ser dirigida para melhorar as práticas que reduzem estes factores de risco.]]>
Hercília Guimarães1 , Gustavo Rocha1, Gabriela Vasconcellos1 , Elisa Proença2 , Maria Luísa Carreira3, Maria do Rosário Sossai4 , Benvinda Morais4, Isabel Martins5, Teresa Rodrigues5, Milton Severo5
1 Maternidade Júlio Dinis (Director: Dr. José Pombeiro), Porto
2 Hospital de Santo António (Director Drª. Paula Cristina Fernandes), Porto
3 Hospital Fernando Fonseca (Director Drª Rosalina Barrosos), Lisboa
4 Hospital Pedro Hispano (Drª Agostinha Souto), Porto
5 Serviço de Epidemiologia (Director Professor Henrique de Barros), Faculty of Medicine of Porto University, Portugal
]]> Hospital de S. João (Director: Professora Doutora Hercília Guimarães), Porto
e-mail: hercíliaguimaraes@gmail.com
Abstract
The pathogenesis of bronchopulmonary dysplasia (BPD) is clearly multifactorial. Specific pathogenic risk factors are prematurity, respiratory distress, oxygen supplementation, mechanical ventilation (MV), inflammation, patent ductus arteriosus (PDA), etc.
Aim: To evaluate BPD prevalence and to identify risk factors for BPD in five Portuguese Neonatal Intensive Care Units in order to develop better practices the management of these newborns.
Material and methods: 256 very low birth weight infants with gestational age (GA) <30 weeks and/or birthweight (BW) <1250 g admitted in five Portuguese NICUs, between 2004 and 2006 were studied. A protocol was filled in based on clinical information registered in the hospital charts. BPD was defined as oxygen dependency at 36 weeks of postconceptional age.
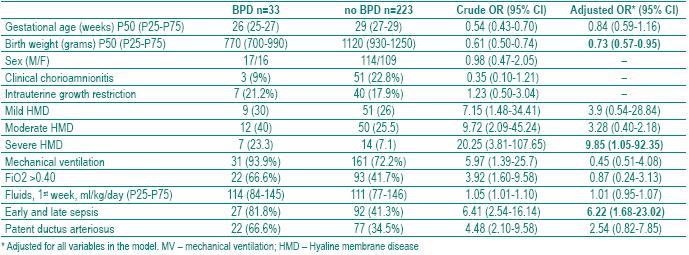
Results: BPD prevalence was 12.9% (33/256). BPD risk decreased 46% per GA week and of 39% per 100g BW. BPD risk was significantly higher among newborns with low BW (adj OR= 0.73, 95% CI=0.57- 0.95), severe hyaline membrane disease (adj OR= 9.85, 95% CI=1.05-92.35), and those with sepsis (adj OR=6.22, 95% CI=1.68-23.02), those with longer duration on ventilatory support (42 vs 3 days, respectively in BPD and no BPD patients, p<0.001) and longer duration of FiO2>0.30 (85 vs 5 days, respectively in BPD and no BPD patients, p<0.001).
Comments: The most relevant risk factors were low birth weight, severe hyaline membrane disease, duration of respiratory support and oxygen therapy, and nosocomial sepsis. The implementation of potentially better practices to reduce lung injury in neonates must be addressed to improve practices to decrease these risk factors.
Key-words: Bronchopulmonary dysplasia, preterm infants, neonatal intensive care, prematurity, hyaline membrane disease, mechanical ventilation, oxygen therapy, risk factors, better practices.
]]>Factores de risco de displasia broncopulmonar em cinco unidades portuguesas de cuidados intensivos neonatais
Resumo
A displasia broncopulmonar (DBP) é multifactorial. Prematuridade, doença da membrana hialina, oxigénio, ventilação mecânica, inflamação e canal arterial são alguns dos factores na sua patogénese.
Objectivo: Avaliar a prevalência da DBP e seus factores de risco em cinco unidades portuguesas, para implementar boas práticas no tratamento deste doentes.
Material e métodos: 256 recém-nascidos (RN) com idade gestacional (IG) <30 semanas e/ou peso <1250 g internados em cinco unidades portuguesas, entre 2004 e 2006, foram estudados. Foi recolhida a informação clínica dos processos. A DBP foi definida como a necessidade de oxigénio às 36 semanas de idade pós-conceptional.
Resultados: A prevalência da DBP foi de 12,9%. O seu risco diminuiu de 46% por semana de IG e de 39% por 100g de peso. O risco de DBP foi maior entre os RN com baixo peso (OR adj = 0,73, 95% CI=0,57-0,95), doença da membrane hialina grave (OR adj = 9,85, 95% CI=1,05-92,35), com sépsis (OR adj = 6,22, 95% CI=1,68-23,02), com maior duração de ventilação (42 vs 3 dias, respectivamente nos RN com e sem DBP, p<0,001) e maior duração de FiO2>0,30 (85 vs 5 dias, respectivamente nos doentes com e sem DBP, p<0,001).
Comentários: Os factores de risco de DBP mais relevantes foram o baixo peso, a doença da membrana hialina grave, a duração da ventilação mecânica e da oxigenoterapia e a sépsis. A implementação das boas práticas para reduzir a lesão pulmonar nos RN deve ser dirigida para melhorar as práticas que reduzem estes factores de risco.
Palavras-chave: Displasia broncopulmonar, recém-nascidos pré-termo, unidades de cuidados intensivos neonatais, doença da membrana hialina, ventilação mecânica, oxigenoterapia, factores de risco, boas práticas.
]]> Introduction
The pathogenesis of Bronchopulmonary dysplasia (BPD) is clearly multifactorial and specific pathogenic known risk factors are prematurity, hyaline membrane disease (HMD), oxygen supplementation, mechanical ventilation (MV), inflammation and infection, patent ductus arteriosus (PDA), among others1. Despite extensive research aimed at identifying risk factors of BPD and devising preventative therapies, many questions about the aetiology and pathogenesis of BPD remain2.
With the advent of surfactant, prenatal steroids and improving technology, the survival rate of extremely low birth weight (ELBW) infants has improved dramatically. Despite these improvements, however, the incidence of BPD in ELBW infants has remained stable over last decades and contributes significantly to the morbidity and mortality seen in these preterm infants. Rates of BPD vary widely. In a recent study, where BPD was defined as oxygen need at 36 weeks postconceptional age, the incidence was 52% in infants with birth weight of 501-750g, 34% in infants with birth weight of 751-1000g, 15% in infants with birth weight of 1001-1200g, 7% in infants with birth weight of 1201-1500g3.
In these very preterm babies, BPD (New BPD) is quite different from the BPD described in the most mature babies (Old or Classic BPD), because the delivery occurred in the very immature stage of the normal lung development4-6. This can explain the histological characteristic features showing rarefaction of the pulmonary vascular bed, reduced alveolarization, less fibrosis and less bronchial metaplasia. In this New PBD the immaturity seems to be much more important than external factors. However oxygen toxicity, volu and barotrauma of mechanical ventilation, inflammation and/or infection (biotrauma) and increased pulmonary flow and lung oedema are also very important risk factors to be taken into account in these immature babies1.
Our aim was to evaluate the prevalence of BPD and to identify risk factors for BPD in preterm babies of five Portuguese NICUs in order to develop better practices in the management of these newborns.
Patients and methods
Very low birth weight (VLBW) infants with gestational age (GA) less than 30 weeks and/ or birth weight (BW) less than 1250 grams admitted in five Portuguese NICUs, between 1st January 2004 and 31st December 2006 and alive at 36 weeks of PCA were included. VLBW infants with major malformations, grade IV intraventricular haemorrhage in the first week of life, metabolic or neuromuscular disease were excluded. A protocol was filled in based on clinical information registered in the hospital charts: maternal history, newborn demographical and clinical data, mechanical ventilation (MV), oxygen supplementation and fluid administration until 36 weeks of postconceptional age (PCA). Neonatal sepsis, patent ductus arteriosus (PDA), necrotizing enterocolitis (NEC), retinopathy of prematurity (ROP), intraventricular haemorrhage (IVH), periventricular leukomalacia (PVL) were also registered. BPD was defined as oxygen dependency at 36 weeks of PCA and had characteristic chest radiographs7. Gestational age (in this study we considered the completed weeks) was assessed by menstrual age (women with regular menstrual cycles), ultrasound examination (when a discrepancy of two or more weeks existed between the age derived by menstrual dating and the age derived sonographically, or in the absence of a menstrual date)8 or the New Ballard Score (in the absence of obstetrical indexes)9. Respiratory distress syndrome (hyaline membrane disease) was defined according to Rudolf AJ et al criteria10. Proven neonatal sepsis was defined as any systemic bacterial or fungal infection documented by a positive blood culture. Hemodynamically significant patent ductus arteriosus was diagnosed on the basis of the echocardiographic findings. The criteria of Bell were used for the diagnosis and staging of necrotizing enterocolitis11. Staging of retinopathy of prematurity was done according to the International Classification12,13. Intraventricular haemorrhage was classified according to Papile LA14. Periventicular leukomalacia was classified according to de Vries L and Rennie JM15.
Odds ratios were used to measure the magnitude of the association between BPD and BPD risk factors. Crude and adjusted Odds ratios were calculated using unconditional logistic regression.
The Breslow-Day and Tarons statistics were computed for the test of homogeneity of the odds ratios among the five hospitals.
]]> A p value <0.05 was considered significant. Statistical analysis was performed using the statistical package SPSS 17.0.
Results
A sample of 256 newborns met inclusion criteria. We observed a decrease in BPD risk of 46% per week of GA and of 39% per 100g BW. The prevalence of BPD was 12.9% (33/256).
Out of 256 infants, 143 (56%) had HMD. Two (6.7%) infants without HMD and in 81 (41.3%) with HMD developed BPD.
The risk of BPD was higher among preterm infants with low GA (crude OR=054, 95% CI=0.43-0.70); low birth weight (crude OR=0.61, 95%CI=0.50-0.74); intrauterine growth restriction (crude OR=1.23, 95%CI=0.50-3.04), those with HMD (crude OR=7.15, 95%CI=1.48-34.41, for mild HMD; crude OR=9.72, 95%CI 2.09-45.24, for moderate HMD and crude OR=20.25, C95%CI=3.81-107.65, for severe HMD), ventilated newborns (crude OR=5.97, 95%CI=1.39-25.7), those with FiO2>0.4 (3.92, 95%CI=1.60-9.58), with higher daily mean fluid administration (crude OR=1.01, 95%CI=1.01-1.10) and those with nosocomial sepsis crude (OR=6.41, 95%CI=2.54-16.14) and PDA (crude OR=4.48, 95%CI=2.10-9.58). After adjustment for all variables in the model, only OR for birth weight, severe HMD and sepsis (early and late or nosocomial) were significantly associated with BPD (Table I).
Table I – Bronchopulmonary risk factors

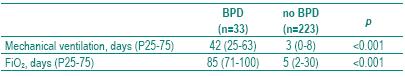
]]> The median of the duration of mechanical ventilation was 42 days in BPD patients and 3 in non BPD patients, p<0.001. The median of the duration of oxygen therapy was 85 days in PBD patients and 5 in non BPD patients, p<0.001 (Table II).
Table II – Duration of mechanical ventilation and FiO2 in patients with and without BPD
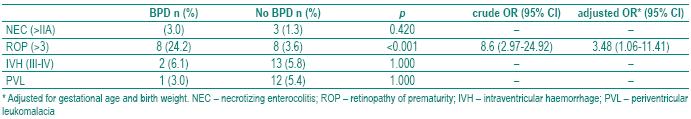
From the associated pathology we looked for (NEC, ROP; IVH and PVL) only ROP was significantly associated with BPD, p<0.001 and OR=3.48 (Table III).
Table III – Associated pathology with bronchopulmonary dysplasia
]]> Discussion
Despite increased knowledge and improving technology, BPD rates have remained high. Its incidence varies among institutions, ranging between 15 and 50% of all VLBW infants16. These differences can be due in part to the definition of BPD and to the decision to administer oxygen that is not uniform, because there is no consensus in the literature and neonatologists have widely divergent practices regarding oxygen saturations targets. In this study we used the BPD definition of oxygen dependency at 36 weeks of PCA.
To decrease the big differences, efforts must be made to identify infants treated with oxygen who are able to maintain saturations exceeding 90% in room air17,18. After 4 decades since the original description by Northway, its clinical presentation, evidence about its pathogenesis and epidemiology have changed and the understanding of this process has provided new possibilities for BPD prevention.
The prevalence of BPD in our study was 12.9%; a similar frequency of 15% has been published in infants with birth weight of 1001-1200g3. In VLBW infants Portugal the frequency of BPD is of 20 %19. However these frequencies must be compared taking into account the mortality rate observed in these NICUs. In this study the global rate of mortality in this group of preterm infants was 17.8%.
The incidence of BPD in premature infants is inversely proportional to gestational age and birth weight16,20. We observed a decrease in BPD risk of 46% per GA week and of 39% per 100g BW; we found no significant difference between sexes.
Chorioamnionitis Inflammation (and infection), either antenatal or postnatal, is likely to be a major trigger for the lung inflammation that plays a role in the pathogenesis of BPD1,21,22. Airways remodelling can occur as a consequence of lung injury23,24.Although there is a recent evidence that premature infants born to mothers with chorioamnionitis are at increased risk of developing BPD21,23, other studies couldnt confirm this association25,26. In this study chorioamnionitis was not analysed because placental histological data, essential for the chorioamnionitis diagnosis, were missing in many patients charts.
Intrauterine growth restriction Preterm infants with intrauterine growth retardation (IUGR) reveal an increased risk of perinatal mortality and neonatal morbidity, namely for the development of acute and chronic pulmonary disorders, i.e. BPD20,27-28. Another recent study showed that AGA infants of 26-28 weeks gestation with birth weights below the median had an increased risk of developing BPD29. In this study, IUGR was not identified as a risk factor of BPD, registered in 21.2% and 17.9% of the babies, respectively with and without the disease. This may be due in part to the fact of the small size of the sample to identify risk factors of this multifactorial disease. However, a recent study shows that the most significant va riable that can be correlated to the long-term outcome is the gestational age20.
Hyaline membrane disease HMD or respiratory distress syndrome is a common cause of morbidity and mortality associated with premature delivery. In uncomplicated cases, typically seen in more mature infants, recovery is rapid and infants generally no longer require oxygen or ventilatory support after the first week of life. The most premature infants are at greatest risk of severe RDS and frequently develop complications, including central nervous system haemorrhage, PDA, air leak, and infection, which contribute to prolonged requirements of oxygen and ventilatory support and consequently preterm infants develop BPD30. BPD rarely develops, nowadays, in infants greater than 32 weeks of gestation, being inversely proportional to GA and BW16. The present study shows, as we expected, that severe HMD was significantly associated with BPD. The increased vulnerability of extremely preterm infants relates to the immature state of the lung development that can be easily damaged by mechanical ventilation and oxygen, required to ensure survival. The premature birth plus therapeutic interventions can disrupt the normal progression of lung architecture, related to the development of alveoli and lung vasculature. This produces significant sequelae, inhibition of acinar development and reduction in number of alveoli and capillaries, seen in the New BPD. In both, Classic and New BPD the lung immaturity is a major BPD risk factor1.
Mechanical ventilation Invasive ventilation via the endotracheal tube is one of the most common therapeutic interventions performed in preterm infants with respiratory failure. Mechanical ventilation using conventional or high-frequency ventilation and surfactant therapy have become the standard of care in management of preterm infants with RDS. However, BPD remains as a major morbidity with adverse pulmonary and non pulmonary outcomes in preterm infants despite these interventions.Ventilator-associated lung injury appears to be related to the duration of invasive ventilation via the endotracheal tube rather than the mode of ventilation. Randomized controlled trials comparing conventional mechanical ventilation and high-frequency ventilation, using optimal ventilatory strategies, have shown no significant difference in rates of BPD. Use of noninvasive ventilation, such as N-CPAP has shown a significant decrease in post extubation failure as well as reduced incidence of BPD31-33.
In the present study the risk of BPD was significantly higher among ventilated newborns, but after adjustment for all variables in the model in MV the difference was not statistically significant (Table I). However the median of the duration of mechanical ventilation was 42 days in BPD patients and 3 in non BPD patients, p<0.001 (Table II). This aspect confirms the importance of MV as a risk factor of BPD and should be taken into account in the management of these preterm infants.
]]> N-CPAP or early surfactant therapy with early extubation onto N-CPAP rather than continued mechanical ventilation has been adopted by many centres, particularly in Scandinavia, as part of the treatment of newborns with respiratory distress syndrome. It has been suggested that BPD is less of a problem in centres adopting such a policy. Results from randomized trials suggest prophylactic or early N-CPAP may reduce BPD, but further studies are required to determine the relative contributions of an early lung recruitment policy, early surfactant administration and N-CPAP in reducing BPD. In addition, the optimum method of generating and delivering NCPAP needs to be determined. The efficacy of N-CPAP in improving long-term respiratory outcomes needs to be compared with the newer ventilator techniques with the optimum and timing of delivery of surfactant administration34,35.Oxygen Oxygen is the most commonly used therapy in NICUs as an integral part of respiratory support. The objective of oxygen therapy is to achieve adequate delivery of oxygen to the tissue without creating oxygen toxicity. However current evidence for optimal oxygen saturation for extremely premature infants is scarce. We still know very little about how much oxygen these babies actually need, or how much oxygen is safe to give, especially in the first few weeks of life35-38.
In the STOP-ROP trial Supplemental Therapeutic Oxygen for Prethreshold Retinopathy), babies in supplemental oxygen arm (target saturations of 96-99%) had evidence of adverse pulmonary outcome comparing with the conventional oxygen arm (target saturations of 89-94%)39.
Recent studies of Saugstad and co-workers showed that in ELBW infants, oxygen saturations levels should be kept between 85 and 93% or possibly between 88 and 95%, but should definitely not exceed 95% and fluctuations should be avoided35.
In this study the risk of BPD was significantly higher among newborns with FiO2>0.4, but after adjustment for all variables in the model, the difference was no longer statistically significant (Table I). However the median of the duration of oxygen therapy was 85 days in PBD patients and 5 in no BPD patients, p<0.001 (Table II). This aspect confirms the importance of the high FiO2 as risk factor of BPD and should be taken into account in the management of these preterm infants. To avoid hyperoxemia is an important goal during respiratory support and neonatal exposure to 100% oxygen is almost never necessary. Much lower FiO2 during the neonatal period can also lead to oxygen toxicity if oxygen is used when it is not necessary. Even brief neonatal exposures to pure oxygen must be avoided40.
Recent data show that a lower FIO2, less than 0.45, confers greater advantage in reducing the incidences of air leak syndromes and BPD comparing with a higher FIO2 (more than 0.45), in the treatment of RDS21.
Fluids We couldnt identify fluids administration as a risk factor of BPD in our patients. The risk of BPD was significantly higher among newborns with higher daily mean fluid administration, but after adjustment for all variables in the model, the difference was not statistically significant (Table I). This may be due in part to the fact of the small size of the sample to identify risk factors of this multifactorial disease. However the excessive fluid intake and/or decreased early weight loss and prolonged PDA are major pathogenic mechanisms for BPD well known. Infants with BPD have increased lung water and are susceptible to gravity-induced collapse and alveolar flooding in the dependent lung with focal tissue damage being distributed inhomogenously. High fluid volumes in the first days of life may increase neonatal morbidity, being associated to increased risk of PDA. Therefore fluid restriction is a standard treatment in the care of the premature infant, with the goal of reducing BPD risk31,41-44.
Neonatal sepsis The presence of nosocomial infections during the first month of life increases the risk of BPD in preterm infants requiring prolonged mechanical ventilation, another risk factor to the disease45,46. Neonatal infection increased also the risk of late death, neurosensory impairment and in extremely low birth weight infants47.
In our study, nosocomial sepsis was observed in 81.8% of preterm infants with BPD and in 41.3% of preterm infants without the disease, OR=6.41, 95% CI=2.54-16.14, (Table I). The national average of sepsis in VLBW infants is 35 %19. Rates of early and late-onset septicaemia of 5% and 29.4%, respectively, were recently published in VLBW infants48.
It is crucial to reduce neonatal sepsis in our preterm infants in all NICUs. As neonatal sepsis is significantly associated with BPD in our patients, in decreasing sepsis we can decrease BPD rate in Portuguese preterm infants. (Table I). With the increasing survival of extremely premature infants there are a large number of them who are developing chronic lung disease, but the severity of the lung damage is considerably less than that observed in the classic form of BPD. Many of these infants have only a mild initial respiratory distress and therefore do not receive aggressive ventilation. So it seems that factors other than oxygen toxicity and mechanical ventilation are involved in the pathogenesis of this new milder type of BPD1,45.
]]> Patent ductus arteriosus In this study the risk of BPD was significantly higher among newborns with PDA (crude OR=4.48, 95%CI=2.10-9.58), but after adjustment for all variables in the model, the difference was no longer statistically significant (Table I). This may be due in part to the fact of the small size of the sample to identify risk factors of this multifactorial disease.Clinical and epidemiological data strongly suggest that the presence of a PDA plays a major role in the development of BPD in these infants and accounts for significant morbidity in preterm newborns49. For this reason, efforts to prevent BPD in extremely low birth weight infants should include an aggressive approach to an early closure of the PDA hemodynamically significant50-53. However it has also been assessed that in randomized control trials, neither a significant reduction, nor even a trend towards a reduction on BPD was observed54.
Major pathology In major pathology we included NEC (grade > IIA), ROP (grades >3), IVH (grades 3-4) and PVL, because of the few cases in the sample. The major pathology observed in our patients was significantly different among the centers.
In our study, from the BPD associated pathology (NEC, ROP; IVH and PVL) we looked for, only ROP was significantly associated with BPD, p<0.001, adjusted OR=3.48, 95%CI=1.06-11.41 (Table III). In VLBW infants in Portugal, we found IVH in 27 %, NEC in 10 %, ROP in 9 %, PVL in 6 %, of preterm infants less than 1500 grams19.
In a recent Spanish study, intraventricular haemorrhage grades 3 to 4 (8.1%) and cystic leukomalacia (2.6%) were the most relevant brain ultrasound findings and NEC was observed in 6.9% of VLBW infants48. In a 10 years period, to investigate trends in mortality and morbidity in very preterm infants there were no changes in the rates of IVH (grades 3-4), ROP (grades > 3), seizures or NEC (grade > IIA. The increasing rate of sepsis was present in infants <28 gestational weeks, whereas the increase in BPD was demonstrated in the whole study population <32 gestational weeks49.
Comments
Bronchopulmonary dysplasia has been increasing over the past two decades in parallel with an improvement in the survival ELBW infants. It stems from the interaction of multiple factors that can damage the immature lung. For this reason prevention must be based on the elimination of all the factors implicated in its pathogenesis.
Our data show that in the five centers of the study the prevalence of BPD was 12.9% (33/256) and the most relevant risk factors identified were low birth weight, severe HMD, duration of MV, duration of oxygen therapy and neonatal sepsis.
The implementation of potentially better practices to reduce lung injury in neonates in Portuguese NICUs must be addressed to decrease HMD, mechanical ventilation, oxy gen therapy and the prevalence of sepsis. However all NICUs must keep making efforts to assure known better practices, decreasing risk factors and contributing to BPD prevention55-57.
]]>Bibliography
1. Sosenko IRS, Bancalari E. New developments in the presentation, pathogenesis, epidemiology and prevention of bronchopulmonary dysplasia. In: Polin RA (Ed.). The Newborn Lung Eduardo Bancalari. Saunders Elsevier 2008; 187-207.
2. Halliday HL, O´Neil CP. What is the evidence for drug therapy in the prevention and management of bronchopulmonary dysplasia? In: Bancalari E, Polin RA (Ed.). The newborn lung. Neonatal questions and controversies. Saunders Elsevier 2008; 208-232.
3. Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA, Wrage LA, Poole K. National Institutes of Child Health and Human Development Neonatal Research Network Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics 2005; 116(6):1353-1360.
4. Northway WH Jr, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease.Bronchopulmonary dysplasia. N Engl J Med 1967; 16;276(7):357-368.
5. Bancalari E, Abdenour GE, Feller R, Gannon J. Bronchopulmonary dysplasia: clinical presentation. J Pediatr 1979; 95(5 Pt 2):819-823.
6. Shennan AT, Dunn MS, Ohlsson A, Lennox K, Hoskins EM. Abnormal pulmonary outcomes in premature infants: prediction from oxygen requirement in the neonatal period. Pediatrics 1988; 82(4):527-532.
7. Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respi Crit Care Med 2001; 163:1723-1729.
8. MacDonald H. American Academy of Pediatrics. Committee on fetus and newborn. Perinatal care at the threshold of viability. Pediatrics 2002; 110: 1024-1027.
]]> 9. Ballard JL, Khoury JC, Wedig K, Wang L, Eilers- Walsman BL, Lipp R. New ballard score, expanded to include extremely premature infants. J Pediatr 1991; 119: 417-423.10. Rudolph AJ, Smith CA. Idiopathic respiratory distress syndrome of the newborn. J Pediatr 1960;57:905- 921.
11. Walsh MC, Kliegman RM. Necrotizing enterocolitis: treatment based on staging criteria. Ped Clin N Am, 1986; 33:179-201.
12. An International Classification of Retinopathy of Prematurity. Pediatrics 1984; 74:127-133.
13. The International Classification of Retinopathy of Prematurity revisited. International Committee for the Classification of Retinopathy of Prematurity. Arch Ophthalmol 2005; 123:991-999.
14. Papile LA, Burstein J, Burstein R. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birthweights less than 1500g. J Pediatr 1978; 92:529-534.
15. de Vries L, Rennie JM. Preterm brain injury. In: Rennie JM, Roberton NRC. Textbok of neonatology, 3th edition. Churchill Livingstone. London 1999: 1252-1270.
16. Bancalari E, Claure N, Sosenko IR. Bronchopulmonary dysplasia: changes in pathogenesis, epidemiology and definition. Semin Neonatol 2003; 8(1):63-71.
17. Walsh MC, Yao Q, Gettner P, Hale E, Collins M, Hensman A, Everette R, Peters N, Miller N, Muran G, Auten K, Newman N, Rowan G, Grisby C, Arnell K, Miller L, Ball B, McDavid G. National Institute of Child Health and Human Development Neonatal Research Network. Impact of a physiologic definition on bronchopulmonary dysplasia rates. Pediatrics 2004; 114(5):1305-1311.
18. Walsh M, Engle W, Laptook A, Kazzi SN, Buchter S, Rasmussen M, Yao Q. National Institute of Child Health and Human Development Neonatal Research Network. Oxygen delivery through nasal cannulae to preterm infants: can practice be improved? Pediatrics 2005; 116(4):857-861.
]]> 19. VLBW infants in Portugal. National Multicenter Study 1996-2000. Portuguese Neonatal Network. Bial Award of Clinical Medicine 2002. ISBN 972-99224-0-3.20. Valcamonico A, Accorsi P, Sanzeni C, Martelli P, La Boria P, Cavazza A, Frusca T. Mid- and long-term outcome of extremely low birth weight (ELBW) infants: na analysis of prognostic factors. Matern Fetal Neonatal Med 2007; 20(6):465-471.
21. Speer CP. Inflammation and bronchopulmonary dysplasia: a continuing story. Semin Fetal Neonatal Med 2006; 11(5):354-362.
22. Bose CL, Dammann CE, Laughon MM. Bronchopulmonary dysplasia and inflammatory biomarkers in the premature neonate. Arch Dis Child Fetal Neonatal Ed 2008; 93(6):F455-F461.
23. Groneck P, Schmale J, Soditt V, Stützer H, Götze-Speer B, Speer CP. Bronchoalveolar inflammation following airway infection in preterm infants with chronic lung disease. Pediatr Pulmonol 2001; 31(5):331-338.
24. Sweet DG, Halliday HL. Modeling and remodeling of the lung in neonatal chronic lung disease: implications for therapy. Treat Respir Med 2005; 4(5):347- 359.
25. Rocha G, Proença E, Quintas C, Rodrigues T, Guimarães H. Chorioamnionitis and lung damage in the extremely low birth weight infant. Rev Port Pneumol 2007; 3(5):745-754. [ Links ]
26. Been JV, Zimmermann LJ. Histologic chorioamnionitis and respiratory outcome in preterm infants. Arch Dis Child Fetal Neonatal; Ed. 2009 Jan 8. [Epub ahead of print.]
26. Gortner L, Reiss I, Hilgendorff A. Bronchopulmonary dysplasia and intrauterine growth restriction. Lancet 2006; 368(9529):28.
27. Marks KA, Reichman B, Lusky A, Zmora E; Israel Neonatal Network. Fetal growth and postnatal growth failure in very-low-birthweight infants. Acta Paediatr 2006; 95(2):236-242. [ Links ]
28. Gortner L, Hilgendorff A, Bähner T, Ebsen M, Reiss I, Rudloff S. Hypoxia-induced intrauterine growth retardation: effects on pulmonary development and surfactant protein transcription. Biol Neonate 2005; 88(2):129-135.
29. Kewitz G, Wudel S, Hopp H, Hopfenmüller W, Vogel M, Roots I. Below median birth weight in appropriate- for-gestational-age preterm infants as a risk factor for bronchopulmonary dysplasia. J Perinat Med 2008; 36(4):359-364.
30. Sweet D, Bevilacqua G, Carnielli V, Greisen G, Plavka R, Didrik Saugstad O,Simeoni U, Speer CP, Valls-I-Soler A, Halliday H. Working Group on Prematurity of the World Association of Perinatal Medicine, European Association of Perinatal Medicine. European consensus guidelines on the management of neonatal respiratory distress syndrome. J Perinat Med 2007; 35(3):175-186.
31. Burch K, Rhine W, Baker R, Litman F, Kaempf JW, Schwarz E, Sun S, Payne NR, Sharek PJ. Implementing potentially better practices to reduce lung injury in neonates. Pediatrics 2003; 111(4 Pt 2):e432-e436.
32. Ramanathan R. Optimal ventilatory strategies and surfactant to protect the preterm lungs. Neonatology 2008; 93(4):302-308.
33. Ramanathan R, Sardesai S. Lung protective ventilatory strategies in very low birth weight infants. J Perinatol. 2008;28(Suppl 1):S41-S46.
34. Bohlin K, Jonsson B, Gustafsson AS, Blennow M Continuous positive airway pressure and surfactant. Neonatology 2008; 93(4):309-315.
35. Patel D, Greenough A. Does nasal CPAP reduce bronchopulmonary dysplasia (BPD)? Acta Paediatr 2008; 97(10):1314-1317. [ Links ]
36. Saugstad OD. Take a breath-but do not add oxygen (if not needed). Acta Paediatr 2007; 96(6):798-800. [ Links ]
37. Saugstad OD, Ramji S, Soll RF, Vento M. Resuscitation of newborn infants with 21% or 100% oxygen: na updated systematic review and meta-analysis. Neonatology 2008; 94(3):176-182.
]]> 38. Tin W, Gupta S. Optimum oxygen therapy in preterm babies. Arch Dis Child Fetal Neonatal; Ed. 2007; 92(2):F143-F147.39. STOP-ROP Multicenter Study Group. Supplemental therapeutic oxygen for pretreshold retinopathy of prematurity (STOP-ROP), a randomized controlled trial. I: Primary outcomes. Pediatrics 2000; 105:295-310.
40. Sola A. Oxygen in neonatal anesthesia: friend or foe? Curr Opin Anaesthesiol 2008; 21(3):332-339.
41. Stevens TP, Harrington EW, Blennow M, Soll RF. Early surfactant administration with brief ventilation vs. selective surfactant and continued mechanical ventilation for preterm infants with or at risk for respiratory distress syndrome. Cochrane Database Syst Rev 2007; 17;(4):CD003063.
42. Oh W, Poindexter BB, Perritt R, Lemons JA, Bauer CR, Ehrenkranz RA, Stoll BJ, Poole K, Wright LL. Neonatal Research Network. Association between fluid intake and weight loss during the first ten days of life and risk of bronchopulmonary dysplasia in extremely low birth weight infants. J Pediatr 2005; 147(6):786-790.
43. Stephens BE, Gargus RA, Walden RV, Mance M, Nye J, McKinley L, Tucker R, Vohr BR. Fluid regimens in the first week of life may increase risk of patent ductus arteriosus in extremely low birth weight infants. J Perinatol 2008; 28(2):123-128.
44. Bell EF, Acarregui MJ. Restricted versus liberal water intake for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 2008; (1):CD000503.
45. Gonzalez A, Sosenko IR, Chandar J, Hummler H, Claure N, Bancalari E. Influence of infection on patent ductus arteriosus and chronic lung disease in premature infants weighing 1000 grams or less. J Pediatr 1996; 128(4):470-478.
46. Liljedahl M, Bodin L, Schollin J. Coagulase-negative staphylococcal sepsis as a predictor of bronchopulmonary dysplasia. Acta Paediatr 2004; 93(2):211-215. [ Links ]
47. Bassler D, Stoll BJ, Schmidt B, Asztalos EV, Roberts RS, Robertson CM, Sauve RS. Using a count of neonatal morbidities to predict poor outcome in extremely low birth weight infants: added role of neonatal infection. Trial of Indomethacin Prophylaxis in Preterms Investigators. Pediatrics 2009; 123(1):313-318.
]]> 48. Moro M, Pérez-Rodriguez J, Figueras-Aloy J, Fernández C, Doménech E, Jiménez R, Pérez-Sheriff V, Quero J, Roques V. Predischarge morbidities in extremely and very low-birth-weight infants in spanish neonatal units. Am J Perinatol 2008; 17.49. Lundqvist P, Källén K, Hallström I, Westas LH. Trends in outcomes for very preterm infants in the southern region of Sweden over a 10-year period. Acta Paediatr 2009; 98(4):648-653. [ Links ]
50. First SIBEN clinical consensus: diagnostic and therapeutic approach to patent ductus arteriosus in premature newborns. Golombek SG, Sola A, Baquero H, Borbonet D, Cabañas F, Fajardo C, Goldsmit G, Lemus L, Miura E, Pellicer A, Pérez JM, Rogido M, Zambosco G, van Overmeire B, Primer Grupo de Consenso Clínico SIBEN (Golombek SG, Sola A, Clyman R, van Overmeire B, Goldsmit G, Natta D, Zambosco G, Miura E, Péerez JM, Weissheimer C, Baquero H, García Harker J,Oviedo Barrantes AN, Morgues M, Tapia JL, Domínguez F, Majano M, Cabañas F, Pellicer A, Cruz H, Fajardo C, Rogido M, Lemus L, Origel AV, Lacarruba JM, Lee M, Tresierra J, Guimarães H, Bustos R, Borbonet D, Perales JL. An Pediatr 2008; 69(5):454-481.
51. Ohlsson A, Walia R, Shah S. Ibuprofen for the treatment of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database Syst Rev 2008; 23;(1):CD003481.
52. Mosalli R, Alfaleh K. Prophylactic surgical ligation of patent ductus arteriosus for prevention of mortality and morbidity in extremely low birth weight infants. Cochrane Database Syst Rev 2008; 23;(1):CD006181.
53. McCurnin D, Seidner S, Chang LY, Waleh N, Ikegami M, Petershack J, Yoder B, Giavedoni L, Albertine KH, Dahl MJ, Wang ZM, Clyman RI. Ibuprofen-induced patent ductus arteriosus closure: physiologic, histologic, and biochemical effects on the premature lung. Pediatrics 2008; 121(5):945-956.
54. Tin W, Wiswell TE. Adjunctive therapies in chronic lung disease: examining the evidence. Semin Fetal Neonatal Med 2008; 13(1):44-52.
55. Payne NR, LaCorte M, Karna P, Chen S, Finkelstein M, Goldsmith JP, Carpenter JH. Breathsavers Group, Vermont Oxford Network Neonatal Intensive Care Quality Improvement Collaborative. Reduction of bronchopulmonary dysplasia after participation in the Breathsavers Group of the Vermont Oxford Network Neonatal Intensive Care Quality Improvement Collaborative. Pediatrics 2006; 118(Suppl 2):S73-S77.
56. Payne NR, LaCorte M, Sun S, Karna P, Lewis-Huns tiger M, Goldsmith JP. Breathsavers Group. Evaluation and development of potentially better practices to reduce bronchopulmonary dysplasia in very low birth weight infants. Pediatrics 2006; 118(Suppl 2):S65-S72.
57. Sharek PJ, Baker R, Litman F, Kaempf J, Burch K, Schwarz E, Sun S, Payne NR. Evaluation and development of potentially better practices to prevent chronic lung disease and reduce lung injury in neonates. Pediatrics 2003; 111(4 Pt 2):e426-e431.
]]>Recebido para publicação/received for publication: 09.04.23
Aceite para publicação/accepted for publication: 09.11.05
]]>