Contracepção hormonal e alterações do padrão menstrual: em que ponto estamos?
Hormonal contraception and bleeding pattern changes: where do we stand?
Filipa de Castro Coelho*, Francisco Macedo**, Cláudia Freitas***
Serviço de Saúde da Região Autónoma da Madeira (SESARAM), E.P.E., Funchal, Portugal
*Interna do Internato Complementar de Obstetrícia e Ginecologia, Hospital Dr. Nélio Mendonça - Serviço de Saúde da Região Autónoma da Madeira (SESARAM), E.P.E., Funchal, Portugal
**Interno do Internato Complementar de Medicina Geral e Familiar, Centro de Saúde Dr. Rui Adriano de Freitas - SESARAM, E.P.E., Funchal, Portugal
***Assistente Hospitalar de Obstetrícia e Ginecologia, Hospital Dr. Nélio Mendonça - SESARAM, E.P.E., Funchal, Portugal
Endereço para correspondência | Dirección para correspondencia | Correspondence
]]> ABSTRACT
Unscheduled bleeding is a major current problem in women’s health. The management of bleeding pattern changes in women using hormonal contraception is challenging. Recommendations for the best practices are provided where evidence exists. This paper provides an updated review regarding the approach of unscheduled bleeding in women who use hormonal contraception.
Keywords: Uterine hemorrhage; Metrorrhagia; Contraceptive agents; Contraceptive devices; Treatment outcome.
Introdução
A hemorragia uterina anómala (HUA) representa um importante problema actual na saúde da mulher que motiva o recurso a cerca de um terço de todas as consultas das especialidades de Medicina Geral e Familiar e de Ginecologia1. Este facto acarreta um grande ónus social e económico, resultado não só da possível associação com patologia do trato genital, mas também pela ansiedade gerada, com consequentes investigações invasivas, de elevados custos e que raramente traduzem patologia na mulher jovem.
A administração exógena de esteróides sexuais (estrogénios, progestativos e androgénios) para controlo da fecundidade, tratamento de HUA ou como terapêutica hormonal, está frequentemente associada a hemorragia uterina intercíclica2. A actuação clínica perante mulheres que se apresentam com alterações do padrão menstrual durante o uso de contraceptivos hormonais constitui um desafio.
Neste artigo, é revisto o conhecimento actual sobre a avaliação e o tratamento das alterações do padrão menstrual associadas ao uso de contracepção hormonal. Todas as indicações sugeridas destacam, da melhor forma possível, os estudos bibliográficos, os pareceres e recomendações de peritos.
Importância da alteração do padrão menstrual com a contracepção hormonal
Em Portugal, de acordo com o último Inquérito Nacional de Saúde3, estima-se que 85,1% da população residente feminina com idade compreendida entre os 15-55 anos e em condição passível de gravidez utiliza algum método contraceptivo, sendo a contracepção hormonal combinada oral, o preservativo e o dispositivo intrauterino, os métodos mais frequentes. Contudo, o uso de um método contraceptivo não garante a sua eficácia (conceito implícito no Índice de Pearl). Em países como França, onde 82% das mulheres utilizam um método contraceptivo hormonal, a frequência de gravidezes não planeadas representa paradoxalmente 33% de todas as gravidezes, e 50% destas terminam em interrupção voluntária da gravidez (IVG)4,5.
]]> A análise de factores que determinam uma gravidez não planeada sugere que a interrupção do método de contracepção tem um contributo significativo5-7. A segunda causa mais citada para interrupção do método de contracepção é a presença concomitante de alterações do ciclo menstrual associadas ao seu uso7.Em suma, a intervenção médica preventiva e terapêutica aquando da alteração do padrão menstrual associado ao uso de contraceptivos hormonais, deve passar pelo encorajamento ao uso continuado do método, de modo a que melhore a sua aceitabilidade e a compliance, contribuindo assim para um menor número de gravidezes não planeadas e IVG consequentes8-11.
Terminologia
O uso de múltiplas nomenclaturas e terminologias para a definição e classificação das hemorragias uterinas, inclusive as de causa iatrogénica, tem dificultado a interpretação de resultados aquando da investigação deste tema. Esta situação promoveu a formação de um grupo internacional de trabalho sob a égide da International Federation of Gynecology and Obstetrics (FIGO) - FIGO Menstrual Disorders Working Group - que recomenda definir menstruação e ciclo menstrual normal de acordo com os seguintes parâmetros do fluxo: (1) regularidade, (2) frequência, (3) volume e (4) duração. Qualquer hemorragia genital com origem intrauterina, que se apresente com pelo menos um destes parâmetros anómalo, deve ser considerada como HUA12,13. A hemorragia intercíclica define-se pela presença de hemorragia genital que ocorre entre os períodos menstruais normais. Tal hemorragia pode surgir de um modo cíclico e previsível ou pode não apresentar nenhum padrão em particular13,14. As alterações do padrão menstrual são quase inevitáveis com o uso de terapêutica progestativa e/ou estrogénica, muitas vezes associada a um padrão de difícil previsão, dado a particularidade e variabilidade que cada método apresenta em cada mulher. Segundo o sistema de classificação das causas de HUA (anomalous uterine bleeding - AUB) da FIGO, classifica-se, segundo o acrónimo «PALM-COEIN», as causas como «estruturais» e identificáveis por imagiologia e histologia - Polyp, Adenomyosis, Leiomyoma, Malignancy and Hyperplasia - e «não-estruturais» - Coagulopathy, Ovulatory Disorders, Endometrium, Iatrogenic and Not Yet Classified. Quando a hemorragia ocorre durante a suplementação com hormonas esteróides sexuais, é classificada como de causa iatrogénica: AUB - I (I - Iatrogenic)13.
Patogénese
O ciclo menstrual, na maioria das mulheres, é caracterizado por uma ovulação regular e uma sequência ordenada de sinais endócrinos que se traduzem na previsibilidade, regularidade e consistência das menstruações15,16. A patogénese da hemorragia intercíclica nas mulheres que utilizam métodos contraceptivos hormonais permanece pouco esclarecida. Após o início do método, a hemorragia intermenstrual parece relacionar-se com a transição de um endométrio que inicialmente está relativamente espessado para depois se tornar atrófico, em resultado da dominância do componente progestativo presente em todos os contraceptivos hormonais. Nestas condições, surge no endométrio vasos sanguíneos superficiais que se tornam dilatados, espiralados e desagregados, propensos a hemorragias focais pela sua fragilidade. Juntamente com esta perda de suporte estrutural do estroma endometrial, alterações da hemostase local e de processos pró e anti-oxidantes, poderão também contribuir para a patogénese da hemorragia intercíclica17,18.
Avaliação
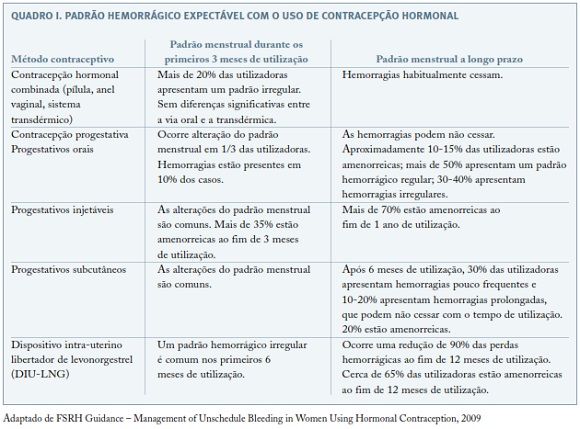
São muitos os factores que determinam a escolha de um contraceptivo. Segundo o Consenso sobre Contracepção da Sociedade Portuguesa de Ginecologia (SPG)19, aquando da prescrição de um método contraceptivo hormonal, torna-se fundamental para uma boa adesão, o aconselhamento: as mulheres devem ser informadas correctamente e de forma clara sobre os métodos de contracepção disponíveis, e devem escolhê-lo livremente de acordo com a sua condição médica, as suas necessidades e expectativas. A Faculty of Sexual and Reproductive Health Care Clinical Effectiveness Unit (FSRH) em colaboração com o Royal College of Obstetricians and Gynaecologists (RCOG), recomendam que durante o aconselhamento devem ser incluídos dois aspectos: 1) informação sobre os padrões hemorrágicos expectáveis associados ao método adoptado, inicialmente e a longo prazo (Quadro I); 2) na presença de hemorragia intercíclica transmitir a ausência de evidência que demonstre que esse sintoma diminui a eficácia contraceptiva20. Deste modo, a oferta atempada de uma informação clara, completa e baseada na evidência, acerca da frequência, evolução e significado clínico da hemorragia intermenstrual, torna a mulher mais apta a continuar um determinado método contraceptivo na presença de hemorragia intercíclica, reduzindo-se o risco de gravidez não planeada.
]]>
A abordagem da hemorragia intercíclica que surge durante os primeiros meses (i.e. 3 - 6 meses) de utilização de um método contraceptivo pode ser diferente da actuação clínica perante uma hemorragia com meses de evolução e/ou que apresenta uma alteração de novo do padrão menstrual. Se durante o uso de contracepção hormonal o padrão menstrual difere do normal e expectável para o período de utilização, então poderá existir indicação para a realização do exame ginecológico, investigação e posterior tratamento20,21.
Embora a contracepção hormonal seja uma causa comum de HUA, outras causas devem ser consideradas (Quadro II). No estudo da HUA, uma história clínica pormenorizada é o passo inicial fundamental que contribui de forma determinante para o diagnóstico diferencial1,20-22 .
A avaliação da HUA associada à utilização de contracepção hormonal é apresentada no Quadro III. Neste contexto, é fundamental verificar qual a compliance com o método contraceptivo; solicitar um calendário menstrual, de modo a determinar um padrão menstrual; excluir uma possível gravidez; excluir infecções sexualmente transmissíveis; e verificar a data e resultado do último exame colpocitológico.
]]> Em todas as mulheres, deve ser avaliada a existência de factores de risco para uma infeção sexualmente transmissível (IST). A infeção por Chlamydia trachomatis é a IST bacteriana mais comum; cerca de 80% das mulheres infectadas por este patogéneo são assintomáticas, no entanto, a HUA pode manifestar-se como sintoma inicial20.
Tratamento
Apesar dos numerosos estudos relacionados com a prevenção e terapêutica da hemorragia intercíclica associada aos métodos de contracepção hormonal, são insuficientes os que apresentam evidência comprovada na abordagem desta situação na prática clínica.
Hemorragia intercíclica associada a contracepção hormonal combinada
A contracepção hormonal combinada (CHC) pode ser administrada por via oral, transdérmica (sistemas ou adesivos transdérmicos) ou vaginal (anel vaginal). A hemorragia intercíclica é menos frequente com o uso de CHC comparativamente ao uso de progestativo isolado.
Nos primeiros 3 meses de utilização, a hemorragia intercíclica está presente em mais de 20% das utilizadoras de CHC, com maior relação nas formulações orais de baixa dosagem (20 ìg) de etinilestradiol (EE) comparativamente com as doses standard (30 a 35 ìg de EE)23. Embora a contracepção oral combinada (COC) com 20 ìg de EE seja teoricamente mais segura, a sua associação a distúrbios hemorrágicos relaciona-se com uma maior frequência de interrupção do método24.
Segundo as recomendações da FSRH em colaboração com o RCOG, uma vez que a hemorragia intercíclica habitualmente cessa ao longo do tempo de utilização, a mudança de método não está indicada nos primeiros 3 meses de utilização20. Tanto a SPG19 como a SOGC (Society of Obstetricians and Gynecologists of Canada)25, recomendam às novas utilizadoras de CHC o uso consistente, com administração diária à mesma hora, associado à informação da utente sobre a natureza transitória da irregularidade do padrão hemorrágico, com melhoria ao longo do tempo. Se após o terceiro ciclo de utilização, a hemorragia persiste ou surge uma hemorragia de novo, outras causas de HUA devem ser excluídas: administração irregular da pilula, tabagismo, patologia cervical ou uterina, gravidez, causas infeciosas, uso de medicação concomitante e/ou síndromes de má absorção20,25. Os processos de absorção e metabolização da CHC podem interagir com o de outros medicamentos (Quadro IV). No entanto, o significado clínico destas interações é questionável e alvo de idiossincrasias nos processos de absorção, ligação às proteínas de transporte ou ao receptor, e na metabolização hepática.
Uma vez eliminadas outras causas de HUA, a abordagem da hemorragia intercíclica associada a CHC, pode implicar o recurso a regimes empíricos sem fortes evidências científicas.
]]>
Contracepção oral combinada - regime cíclico
Deve ser utilizado preferencialmente um método de COC com a menor dose de EE, contudo, de forma a garantir um melhor controlo do ciclo menstrual, pode ser necessário em algumas mulheres, recorrer a formulações contendo 30-35 ìg de EE20. Perante uma hemorragia intermenstrual persistente, um ciclo de estrogenioterapia oral poderá ser útil: 1,25 mg de estrogénio conjugado ou 2 mg de 17â - estradiol, diariamente durante 7 dias.25
Segundo um ensaio clínico randomizado, a adopção de um regime de administração de COC durante 21 dias consecutivos está associado a menos dias com hemorragia intercíclica comparativamente a um regime de 24 dias consecutivos (4,6 dias versus 6,1 dias), apesar da totalidade do número de dias com hemorragia (intercíclica e de privação) ser superior no regime de 21 dias26. Se nenhuma melhoria ou resolução do quadro for observada, poderá estar indicada uma prova terapêutica com outro COC.25.
Outra abordagem possível é a alteração da via de administração substituindo a oral pela via vaginal. Teoricamente, esta abordagem apresenta dois po- tenciais benefícios: (1) diminui a probabilidade de esquecimento, associado a melhor compliance do método e (2) o anel vaginal está associado a níveis hormonais sanguíneos mais estáveis ao longo de todo o ciclo27.
Quanto à componente progestativa do CHC, em regime cíclico, não há evidência científica suficiente que sugira a associação de um determinado tipo de progestativo com menor incidência de hemorragia intercíclica28.
Estudos de revisão sistemática demonstraram ausência de evidência suficiente para recomendar o uso de CHC orais bifásicos ou trifásicos com o intuito de melhorar o padrão hemorrágico29-31.
Contracepção oral combinada - regime contínuo
A opção por um regime contínuo de um método de COC apresenta uma série de vantagens, incluindo a diminuição da incidência de dor pélvica, cefaleias e tensão mamária que surgem durante o período «livre de hormona»; melhoria na sintomatologia de endometriose e do síndrome do ovário policístico; e poderá ser ainda uma escolha conveniente, quando é opção da mulher reduzir o número de dias com hemorragia por ano32,33.
As desvantagens no regime contínuo relacionam-se com a insuficiente evidência na segurança a longo prazo e com os custos associados à medicação despendida nos períodos que seriam de hemorragia de privação. Em comparação com o regime tradicional cíclico, o regime contínuo diminui o número total de dias com hemorragia num ciclo. No entanto, durante os primeiros 3 meses de utilização de um regime contínuo, ocorre com elevada frequência hemorragia intercíclica, que diminui progressivamente com a sua utilização33-35.
]]> Relativamente ao componente progestativo, ao contrário do observado com o regime cíclico, deve ser considerado o tipo de progestativo. A duração da hemorragia intercíclica aparenta ser superior nas mulheres que utilizam em regime contínuo um COC que contenha levonorgestrel (LNG) ao invés do verificado com os que contêm acetato de noretindrona36.De acordo com as mais recentes recomendações do Centers for Disease Control and Prevention (CDC), se após 21 dias de um regime continuado de COC, surge uma hemorragia intercíclica, pode-se interromper o método durante 3 ou 4 dias, para indução de hemorragia de privação e retomá-lo por, pelo menos, 21 dias contínuos. Esta abordagem pode ser repetida sempre que surja uma hemorragia intercíclica, desde que tenham sido administrados comprimidos hormonalmente activos durante 21 dias consecutivos, de modo a se manter a eficácia contraceptiva do método37. Com o tempo, os episódios de hemorragia intercíclica tornam-se mais espaçados e eventualmente cessam.
Hemorragia intercíclica associada a contracepção hormonal progestativa
Contracepção progestativa oral
A hemorragia intercíclica é uma das principais desvantagens do uso de contracepção exclusiva com progestativos. Apesar desta HUA cessar com a continuação do método, ainda não se encontra definido o período de tempo decorrido até ao termo ou melhoria deste sintoma20.
A apresentação oral da contracepção só com progestagénio, existente em Portugal, é o desogestrel. A evidência sobre a abordagem terapêutica na presença concomitante de hemorragia intercíclica é escassa. Algumas recomendações de peritos referem o uso de anti-inflamatórios não esteroides (AINEs)21,25 durante 10 dias ou a estrogenioterapia suplementar25. Na maioria das mulheres, a opção por um método progestativo deve-se à presença de contra-indicações aos estrogénios. No entanto, na ausência de contra-indicação, a estrogenioterapia pode ser uma opção: 1-2 mg de EE via oral durante 28 dias, ou 50-100 ìg via transdérmica durante 25 dias25. Outra opção válida é a troca por uma formulação com baixa dose de COC.
Contracepção progestativa injectável
A contracepção injectável por via intramuscular (IM), em Portugal, é constituída pelo progestativo acetato de medroxiprogesterona (DMPA). Face ao decréscimo previsível dos episódios de hemorragia intercíclica com a continuidade do método, muitas mulheres aceitam aguardar a resolução espontânea do sintoma. Quanto às opções farmacológicas, não existe uma abordagem da hemorragia sustentada em evidência científica sólida.
Na ausência de contra-indicações à estrogenioterapia, a FSRH recomenda como primeira linha terapêutica, a associação de COC (30 - 35 ìg EE com levonorgestrel ou noretisterona) durante 1 a 3 meses, em regime cíclico ou contínuo20. Na presença de contra-indicações a CHC pode-se optar por associar AINEs (ácido mefenâmico 500mg 2id ou 3id, durante 5 dias), apesar da ausência de evidência do seu benefício a longo prazo20,38.
Outras orientações incluem como opções: a suplementação com estrogénio (1-2 mg de 17â-estradiol oral durante 28 dias; 50-100 ìg 17â-estradiol via transdérmica durante 25 dias); AINEs (ibuprofeno 400-600 mg 2id, 10 dias); o aumento da dose de DMPA (entre 225 e 300 mg IM em 2 ou 3 administrações); e a diminuição do intervalo entre as injeções de DMPA25. Quanto a esta última opção terapêutica, apesar de extensamente utilizada, não existem estudos que suportem a sua eficácia. Assim, não está recomendado o encurtamento do intervalo das injeções de DMPA para tratamento da hemorragia intermenstrual. Segundo as recomendações da CDC apenas está indicado o uso de AINEs durante 5 a 7 dias. Se persistirem os sintomas deve ser ponderado um método contraceptivo alternativo37.
]]> Contracepção progestativa subcutâneaOs implantes subcutâneos progestativos estão associados a vários padrões de HUA. Calculou-se que 14 a 18% das utilizadoras do implante com etonogestrel (ETN) interromperam a sua utilização devido ao padrão de hemorragia relacionado com o método39,40.
São escassos os ensaios clínicos que abordam a hemorragia intercíclica associada ao implante com ETN e, até à data, não há evidência a comprovar que os estudos com os implantes que contêm LNG (não comercializado em Portugal) são equiparáveis ao que contém ETN. A investigação da abordagem da hemorragia intercíclica associada aos implantes subcutâneos é inconclusiva, portanto, uma conduta pragmática deve ser aplicada na presença deste efeito adverso41.
A estrogenioterapia (COC contendo 30-35 ìg de EE) assume-se como a associação de primeira linha, com o uso off-label em regime cíclico ou contínuo, durante 3 meses. Esta terapêutica pode ser repetida sempre que necessária, sendo o seu uso restrito a mulheres sem contra-indicações a formulações contendo estrogénios21,41,42. Uma vez mais, os AINEs constituem uma alternativa válida na HUA associada à contracepção exclusiva com progestativos21,37,42. Ensaios clínicos demonstraram diminuição da hemorragia associada ao implante subcutâneo com LNG com a administração oral de celecoxib (200 mg/dia durante 5 dias)43, e da hemorragia associada ao implante com ETN com o ácido mefenâmico (500 mg 3 vezes/dia durante 5 dias)44.
Alguns peritos sugerem que a doxiciclina pode ser benéfica42, contudo a evidência é insuficiente para suportar o seu uso por rotina na prática clínica45,46. Os mesmos realçam ainda a utilidade de progestativos de alta dosagem quando é requerida a interrupção da hemorragia a curto-prazo (e.g. noretisterona 5mg)42.
Contracepção progestativa intrauterina
Presentemente, os dispositivos intrauterinos (DIU) têm na sua composição cobre (CuDIU), mistura de cobre e prata, mistura de prata e de sais de ouro ou progestagénio (LNG). A contracepção intrauterina progestativa designa-se dispositivo de libertação intrauterina (DLIU)47.
Durante os primeiros 6 meses de utilização do dispositivo de libertação intrauterina de levonogestrel (DLIU-LNG) é frequente a presença de um padrão hemorrágico irregular. No entanto, após um ano de utilização do método, pode verificar-se uma redução até cerca de 90% do fluxo menstrual; 65% das utilizadoras estão amenorreicas ou apresentam diminuição da hemorragia uterina findo o primeiro ano de utilização20.
Segundo as recentes orientações do Departamento de Saúde dos EUA, quando a hemorragia intercíclica persiste após os primeiros 3 a 6 meses de utilização e excluída a possibilidade de patologia cervical ou endometrial, devem ser aconselhados métodos contraceptivos alternativos37. De facto, na presente revisão, os autores não encontraram evidência robusta a comprovar os benefícios da farmacoterapia na abordagem das alterações do padrão menstrual associadas ao uso do DLIU-LNG. Um estudo randomizado demonstrou nas utilizadoras deste DIU, uma redução do número de dias com hemorragia com o uso de naproxeno, enquanto o oposto foi observado com o uso de estrogénios suplementares48. Nos primeiros meses de utilização do DIU-LNG ocorre um aumento das prostaglandinas e citocinas no endométrio, o que poderá contribuir para o padrão hemorrágico irregular. Deste modo, o efeito anti-prostaglandinas dos AINEs pode ser benéfico. Assim, justifica-se também o resultado da estrogenioterapia não surtir efeito, uma vez que a hemorragia não se deve a uma verdadeira atrofia endometrial48. No entanto não se demonstrou melhoria do padrão hemorrágico com a utilização de ácido tranexâmico nem com a utilização de ácido mefenâmico49.
Hemorragia intercíclica associada a contracepção não hormonal
]]> A contracepção hormonal subcutânea, os injectáveis, os Cu-DIU e os DLIU-LNG são actualmente classificados como CRLD (contracepção reversível de longa duração), designados como LARC (long-acting reversible contraception) na terminologia anglo-saxónica47. Embora o Cu-DIU não se enquadre nos métodos contraceptivos hormonais, ambos os dispositivos de contracepção intrauterina estão associados a alterações do padrão menstrual. Por este motivo, é feita uma breve referência à abordagem da HUA associada ao Cu-DIU.Similarmente ao DLIU-LNG, a evidência é limitada quanto aos fármacos, doses e duração do tratamento da hemorragia intercíclica associada ao uso do Cu-DIU. Decorridos os primeiros três a seis meses de utilização e, uma vez mais, após exclusão de patologia cervical ou endometrial, pode-se considerar o uso de AINEs durante 5-7 dias37. Contudo, esta opção apesar de reduzir a hemorragia menstrual, não reduz a hemorragia intercíclica50.
Se a hemorragia persiste e é inaceitável para a mulher, outro método contraceptivo deve ser aconselhado37.
Conclusão
A hemorragia intercíclica é um efeito secundário comum da contracepção hormonal, particularmente com a utilização de métodos progestativos. Antes de iniciar um método contraceptivo hormonal, as mulheres devem ser informadas quanto ao padrão hemorrágico expectável. O aconselhamento pré-terapêutico pode determinar substancialmente a compliance.
A actuação clínica perante hemorragias intercíclicas varia com o método e regime utilizados e deve ser dirigida especificamente ao contraceptivo hormonal adoptado. Recomenda-se tranquilizar e orientar a mulher para a não interrupção do método. São opções válidas o recurso a suplementação hormonal estrogénica, progestativa e/ou anti-inflamatória. Por último, um método alternativo pode ser considerado. Uma hemorragia persistente ou ocorrência de uma hemorragia de novo incaracterística, exige que outras causas de HUA sejam excluídas.
É fundamental um maior grau de evidência na abordagem desta situação na prática clínica. O investimento futuro no conhecimento da patogénese das alterações do padrão menstrual associadas à contracepção hormonal, poderá atenuar o elevado ónus sócio-económico que a HUA representa na saúde da mulher.
REFERÊNCIAS BIBLIOGRÁFICAS
1. Martinho M, Melo A. Hemorragias uterinas anormais. In: Manual de Ginecologia. Portugal: Permanyer; 2011. p. 135-146. [ Links ]
2. Hickey M, Fraser IS. Iatrogenic unscheduled (breakthrough) endometrial bleeding. Rev Endocr Metab Disord. 2012 Dec;13(4): 301-308. [ Links ]
3. Instituto Nacional de Saúde/Instituto Nacional de Estatística. 4º Inquérito Nacional de Saúde 2005/2006. Lisboa: INSA/INS; 2009. p.56 [ Links ]
4. Bajos N, Leridon H, Goulard H, Oustry P, Job-Spira N; COCON Group. Contraception: from accessibility to efficiency. Hum Reprod. 2003 May;18(5):994-999. [ Links ]
5. Bajos N, Lamarche-Vadel A, Gilbert F, Ferrand M; COCON Group, Moreau C et al. Contraception at the time of abortion: high-risk time or high-risk women? Hum Reprod. 2006 Nov;21(11):2862-2867. [ Links ]
6. Jones RK, Darroch JE, Henshaw SK. Contraceptive use among U.S. women having abortions in 2000-2001. Perspect Sex Reprod Health. 2002 Nov-Dec;34(6):294-303.
7. Moreau C, Cleland K, Trussell J. Contraceptive discontinuation attributed to method dissatisfaction in the United States. Contraception. 2007 Oct;76(4):267-272. [ Links ]
8. Frost JJ, Darroch JE. Factors associated with contraceptive choice and inconsistent method use, United States, 2004. Perspect Sex Reprod Health. 2008 Jun;40(2):94-104. [ Links ]
9. Frost JJ, Singh S, Finer LB. Factors associated with contraceptive use and nonuse, United States, 2004. Perspect Sex Reprod Health. 2007 Jun;39(2):90-99.
10. Huber LR, Hogue CJ, Stein AD, Drews C, Zieman M, King J et al. Contraceptive use and discontinuation: findings from the contraceptive history, initiation, and choice study. Am J Obstet Gynecol. 2006 May;194(5):1290-1295.
11. Trussell J. Contraceptive failure in the United States. Contraception. 2011 May;83(5):397-404.
12. Fraser IS, Critchley HO, Broder M, Munro MG. The FIGO recommendations on terminologies and definitions for normal and abnormal uterine bleeding. Semin Reprod Med. 2011 Sep; 29(5):383-390.
13. Munro MG, Critchley HO, Broder MS, Fraser IS; FIGO Working Group on Menstrual Disorders. FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age. Int J Gynaecol Obstet. 2011 Apr;113(1):3-13.
14. Munro MG, Critchley HO, Fraser IS. The FIGO systems for nomenclature and classification of causes of abnormal uterine bleeding in the reproductive years: who needs them? Am J Obstet Gynecol. 2012 Oct; 207(4):259-265.
15. Jabbour HN, Kelly RW, Fraser HM, Critchley HO. Endocrine regulation of menstruation. Endocr Rev. 2006 Feb;27(1):17-46.
]]> 16. Henriet P, Gaide Chevronnay HP, Marbaix E. The endocrine and paracrine control of menstruation. Mol Cell Endocrinol. 2012 Jul 25;358(2):197-207.17. ESHRE Capri Workshop Group. Ovarian and endometrial function during hormonal contraception. Hum Reprod. 2001 Jul; 16(7):1527-1535.
18. Hickey M, Fraser IS. Clinical implications of disturbances of uterine vascular morphology and function. Baillieres Best Pract Res Clin Obstet Gynaecol 2000;14:937-951. [ Links ]
19. Consenso sobre contracepção 2011. Reunião de Consenso Nacional sobre Contracepção. Estoril, 2011. [ Links ]
20. Faculty of Sexual and Reproductive Health Care Clinical Effectiveness Unit FSRH Guidance in collaboration with the Royal College of Obstetricians and Gynaecologists. Management of Unschedule Bleeding in Women Using Hormonal Contraception; 2009. [ Links ]
21. Lumsden MA, Gebbie A, Holland C. Managing unscheduled bleeding in non-pregnant premenopausal women. BMJ. 2013 Jun 4;346:f3251. [ Links ]
]]>22. Schrager S. Abnormal uterine bleeding associated with hormonal contraception. Am Fam Physician. 2002 May 15;65(10): 2073-2080. [ Links ]
23. Gallo MF, Nanda K, Grimes D, Schulz KF. Twenty micrograms vs. >20 µg estrogen oral contraceptives for contraception: systematic review of randomized controlled trials. Contraception. 2005 Mar; 71(3): 162-169. [ Links ]
24. Gallo MF, Nanda K, Grimes DA, Lopez LM, Schulz KF. 20 µg versus >30 µg estrogen combined oral contraceptives for contraception. Cochrane Database Syst Rev. 2011 Jan 19;1:CD003989. [ Links ]
25. Black A, Francoeur D, Rowe T, et al. Canadian Contra¬ception Consensus. SOGC, Clinical Guidelines. Nº 143, part 2 of 3; 2004.
26. Kaunitz AM, Burkman RT, Fisher AC, Laguardia KD. Cycle control with a 21-day compared with a 24-day oral contraceptive pill: a randomized controlled trial. Obstet Gynecol. 2009 Dec;114(6):1205-1212. [ Links ]
27. Oddsson K, Liefels-Fischer B, Wiel-Masson D, de Melo NR, Benedetto C, Verhoeven CH et al. Superior cycle control with a contraceptive vaginal ring compared with an oral contraceptive containing 30 mcg ethinylestradiol and 150 mcg levonorgestrel: a randomized trial. Hum Reprod 2005;20(2):557-562. [ Links ]
28. Lawrie TA, Helmerhorst FM, Maitra NK, Kulier R, Bloemenkamp K, Gülmezoglu AM. Types of progestogens in combined oral contraception: effectiveness and side-effects. Cochrane Database Syst Rev. 2011 May 11;(5):CD004861. [ Links ]
29. Van Vliet HA, Grimes DA, Helmerhorst FM, Schulz KF. Biphasic versus monophasic oral contraceptives for contraception. Cochrane Database Syst Rev. 2006 Jul 19;(3):CD002032. [ Links ]
30. Van Vliet HA, Grimes DA, Helmerhorst FM, Schulz KF. Biphasic versus triphasic oral contraceptives for contraception. Cochrane Database Syst Rev. 2006 Jul 19;(3):CD003283. [ Links ]
31. Van Vliet HA, Grimes DA, Lopez LM, Schulz KF, Helmerhorst FM. Triphasic versus monophasic oral contraceptives for contraception. Cochrane Database Syst Rev. 2011 Nov 9;(11): CD003553. [ Links ]
32. Sulak PJ, Kuehl TJ, Coffee A, Willis S. Prospective analysis of occurrence and management of breakthrough bleeding during an extended oral contraceptive regimen. Am J Obstet Gynecol. 2006 Oct;195(4):935-941. [ Links ]
33. Miller L, Hughes JP. Continuous combination oral contraceptive pills to eliminate withdrawal bleeding: a randomized trial. Obstet Gynecol. 2003 Apr;101(4):653-661. [ Links ]
34. Anderson FD, Hait H. A multicenter, randomized study of an extended cycle oral contraceptive. Contraception. 2003 Aug;68(2):89-96.
35. Archer DF, Jensen JT, Johnson JV, Borisute H, Grubb GS, Constantine GD. Evaluation of a continuous regimen of levonorgestrel/ethinyl estradiol: phase 3 study results. Contraception. 2006 Dec;74(6):439-445.
36. Edelman AB, Koontz SL, Nichols MD, Jensen JT. Continuous oral contraceptives: are bleeding patterns dependent on the hormones given? Obstet Gynecol. 2006 Mar;107(3):657-665.
37. Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion. U.S. Selected Practice Recommendations for Contraceptive Use, 2013: adapted from the World Health Organization Selected Practice Recommendations for Contraceptive Use, second edition. MMWR Recomm Rep 2013; 62(RR-05):1-60. [ Links ]
38. Tantiwattanakul P, Taneepanichskul S. Effect of mefenamic acid on controlling irregular uterine bleeding in DMPA users. Contraception. 2004 Oct;70(4):277-279.
39. Mansour D, Korver T, Marintcheva-Petrova M, Fraser IS. The effects of Implanon on menstrual bleeding patterns. Eur J Contracept Reprod Health Care. 2008 Jun;13 Suppl 1:13-28. [ Links ]
40. Casey PM, Long ME, Marnach ML, Bury JE. Bleeding related to etonogestrel subdermal implant in a US population. Contraception. 2011 May;83(5):426-430.
41. Mansour D, Bahamondes L, Critchley H, Darney P, Fraser IS. The management of unacceptable bleeding patterns in etonogestrel-releasing contraceptive implant users. Contraception 2011;83(3):202-210. [ Links ]
42. Dickson J, Hoggart L, Newton VL. Unanticipated bleeding with the etonogestrel implant: advice and therapeutic interventions. J Fam Plann Reprod Health Care. 2014 Jul;40(3):158-160.
43. Buasang K, Taneepanichskul S. Efficacy of celecoxib on controlling irregular uterine bleeding secondary to Jadelle use. J Med Assoc Thai. 2009 Mar;92(3):301-307.
44. Phaliwong P, Taneepanichskul S. The effect of mefenamic acid on controlling irregular uterine bleeding second to Implanon use. J Med Assoc Thai. 2004 Oct;87 Suppl 3:S64-68.
45. Weisberg E, Hickey M, Palmer D, O’Connor V, Salamonsen LA, Findlay JK, Fraser IS. A pilot study to assess the effect of three short-term treatments on frequent and/or prolonged bleeding compared to placebo in women using Implanon. Hum Reprod. 2006 Jan;21(1):295-302.
46. Weisberg E, Hickey M, Palmer D, O’Connor V, Salamonsen LA, Findlay JK et al. A randomized controlled trial of treatment options for troublesome uterine bleeding in Implanon users. Hum Reprod 2009;24(8):1852-1861. [ Links ]
]]>47. Ramalho J, Neves J. Contraceção intrauterina. In: Contraceção. Lidel, Lisboa, 2013:252-265. [ Links ]
48. Madden T, Proehl S, Allsworth JE, Secura GM, Peipert JF. Naproxen or estradiol for bleeding and spotting with the levonorgestrel intrauterine system: a randomized controlled trial. Am J Obstet Gynecol. 2012 Feb;206(2):129.e1-8.
49. Sordal T, Inki P, Draeby J, O’Flynn M, Schmelter T. Management of initial bleeding or spotting after levonorgestrel-releasing intrauterine system placement: a randomized controlled trial. Obstet Gynecol. 2013 May;121(5):934-941.
50. Godfrey EM, Folger SG, Jeng G, Jamieson DJ, Curtis KM. Treatment of bleeding irregularities in women with copper-containing IUDs: a systematic review. Contraception. 2013 May;87(5): 549-566.
Contribuições dos autores
O primeiro e segundo autor contribuíram de forma semelhante neste artigo.
nota
Por opção dos autores, o artigo foi escrito sem aplicação do novo acordo ortográfico.
]]>Endereço para correspondência | Dirección para correspondencia | Correspondence
Filipa de Castro Coelho
E-mail: filipacastrocoelho@gmail.com
Recebido em: 20-10-2014
Aceite para publicação: 15-12-2014
]]>