Serviços Personalizados
Journal
Artigo
Indicadores
Links relacionados
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.25 no.6 Lisboa dez. 2018
https://doi.org/10.1159/000486448
CLINICAL CASE STUDY
Common Variable Immunodeficiency-Associated Inflammatory Enteropathy: The New Era of Biological Therapy
Enteropatia Inflamatória Associada à Imunodeficiência Variável Comum: A Nova Era da Terapêutica Biológica
Cátia Arieira, Francisca Dias de Castro, Maria João Moreira, José Cotter
Gastroenterology Department, Hospital da Senhora da Oliveira, Guimarães, Portugal; Life and Health Sciences Research Institute, School of Medicine, University of Minho, Braga/Guimarães, Portugal; ICVS/3Bs, PT Government Associate Laboratory, Braga/Guimarães, Portugal
*Corresponding author.
ABSTRACT
Common variable immunodeficiency (CVID) is the most frequent form of immunodeficiency in adults. Clinical manifestations are heterogeneous with an increased susceptibility to infections and inflammatory conditions, namely autoimmune diseases, such as inflammatory bowel disease. We present the clinical case of a Caucasian female patient, 21 years old, with a past medical history of CVID, with multiple visits to the emergency department due to abdominal pain in the lower quadrants and diarrhea. Her biochemical analysis showed elevated inflammatory parameters. Stool cultures and parasitological examination of feces were negative. Ileocolonoscopy revealed lymphoid nodular hyperplasia of the terminal ileum, and the small bowel capsule endoscopy demonstrated edema and multiple pleomorphic ulcers (Lewis score = 1,104). CVID-associated inflammatory enteropathy was suspected. Budesonide 9 mg/day was started, but no positive clinical response was observed. It was decided to initiate biological therapy with infliximab. The patients condition is currently clinically improved with no complications due to biological therapy.
Keywords: Common variable immunodeficiency, Crohn disease, Inflammatory bowel disease, Small bowel capsule endoscopy, Biological therapy, Infliximab
RESUMO
A imunodeficiência variável comum (IDVC) é a forma mais frequente de imunodeficiência no adulto. O conjunto de manifestações clínicas é heterogéneo com uma suscetibilidade aumentada a infeções e condições inflamatórias, nomeadamente autoimunes como a doença inflamatória intestinal. Apresenta-se um caso clínico de doente do sexo feminino, 21 anos de idade com antecedentes de imunodeficiência variável comum, com múltiplas admissões ao serviço de urgência por dor abdominal nosquadrantes inferiores e diarreia. Analiticamente apresentava parâmetros inflamatórios elevados. As coproculturas e exame parasitológico de fezes eram negativos. A ileocolonoscopia revelou hiperplasia nodular linfóide do íleon terminal e a enteroscopia por cápsula edema marcado e múltiplas úlceras pleomórficas (Score de Lewis 1104). Suspeitou-se de enteropatia inflamatória associada a IDVC. Iniciou Budesonido 9 mg/dia não tendo sido verificada resposta clínica positiva. Foi decidido iniciar terapêutica biológica com Infliximab. A doente encontra-se, atualmente, clinicamente melhorada sem evidência de atividade inflamatória.
Palavras-Chave: Imunodeficiência variável comum, Doença de Crohn, Doença inflamatória intestinal, Enteroscopia por cápsula, Terapêutica biológica, Infliximab
Introduction
Since the first report over 60 years ago, common variable immunodeficiency (CVID) appears to be the most frequent form of primary immunodeficiency in adults and occurs equally in males and females [1, 2]. It generally presents in the second or third decades, although it may occur at any age [2]. It is a rare condition with an estimated prevalence that ranges from 1/100,000 to 1/10,000 of the population [2]. Although the etiology is unknown [2], genetic factors are involved and seem to be responsible for the presentation and progression of the disease [1]. The diagnosis results from the combination of primary antibody IgG deficiency, along with IgA and/or IgM deficiency, associated with reduced or absent antibody response against vaccination [1, 3]. The clinical spectrum is heterogeneous, and patients usually present a higher susceptibility to recurrent bacterial infection, namely sinopulmonary infections, autoimmune manifestations, and malignancy [4].
There is a higher incidence of autoimmune pathologies in these patients, and, recently, emerging cases of inflammatory bowel disease have been described in the literature with a reported incidence of 2–4% [5]. We report a case of CVID-associated inflammatory enteropathy treated with infliximab with successful outcome.
Case Report
A 21-year-old female was referred to our Gastroenterology Department after multiple admissions to the emergency room due to intense lower abdominal pain and chronic diarrhea with more than 6 months of evolution. She had a past medical history of CVID with multiple rhinopulmonary infections. She also had a history of pulmonary tuberculosis in 2005, which was successfully treated. She was on no regular medication and denied taking nonsteroidal anti-inflammatory drugs. She had no known allergies and had no family history of gastrointestinal pathology.
At the first admission to the emergency room, she had leukocytosis and high C-reactive protein (CRP). At the same admission, an abdominal computed tomography was performed which revealed ileal thickening (Fig. 1). To exclude small intestinal bacterial overgrowth, she underwent a cycle of rifaximin 1,600 mg/day during 7 days, without positive clinical response. At our observation, she presented with lower abdominal tenderness on physical exam. Extended laboratory data were accessed: hemoglobin 13.1 g/dL (normal: 12–16 g/dL); white blood cell count 10,100/μL (normal: 4.8–10.8 . 103/μL); platelet count 234 . 103/μL (normal: 150–350 . 103/μL); erythrocyte sedimentation rate 18 mm (normal: 0–12 mm); CRP 71.6 mg/L (normal: < 3 mg/L); albumin 3.5 g/dL (normal: 3.4–5 g/dL); folate 11 ng/mL (normal: 3.1–17.5 ng/mL); vitamin B12 282 pg/mL (normal: 211–911 pg/mL); fecal calprotectin > 1,800 g/g (normal: < 50 g/g); IgM 46.7 mg/dL (normal: 29–226 mg/dL); IgA < 7.8 mg/dL (normal: 114–457 mg/dL); and IgG 255 mg/dL (normal: 793–1,590 mg/dL). Stool cultures and parasitological examination (3 specimens from 3 different days), namely Giardia lamblia, were negative.
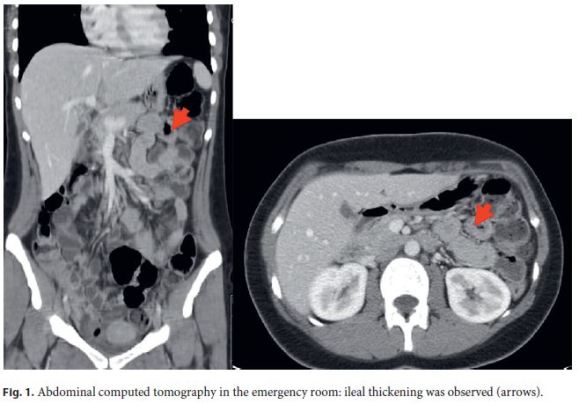
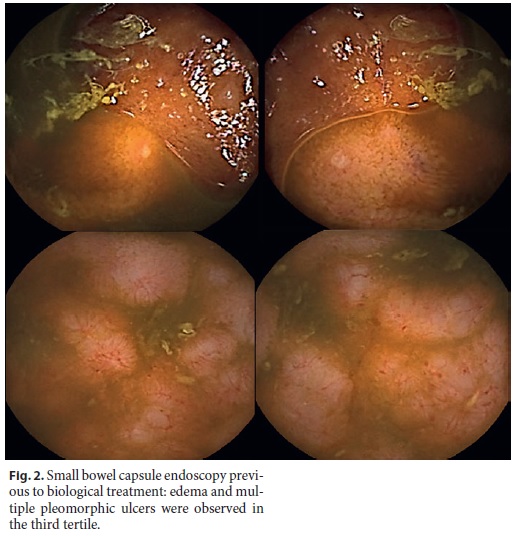
IgG antitransglutaminase antibody was negative. Upper endoscopy was normal, and biopsies of the bulb and second portion of the duodenum were performed, ruling out celiac disease. A colonoscopy with ileoscopy was executed. No lesions were observed in the colon, but the terminal ileum presented with edema and a nodular appearance compatible with nodular lymphoid hyperplasia. Histopathological examination of the terminal ileum confirmed the diagnosis of nodular lymphoid hyperplasia. A small bowel capsule endoscopy (SBCE) was also performed and showed edema and multiple pleomorphic ulcers on the third tertile with a Lewis score of 1,104 (severe inflammatory activity) (Fig. 2) [6, 7]. We concluded that all these findings taken together were consistent with CVID-associated inflammatory enteropathy.
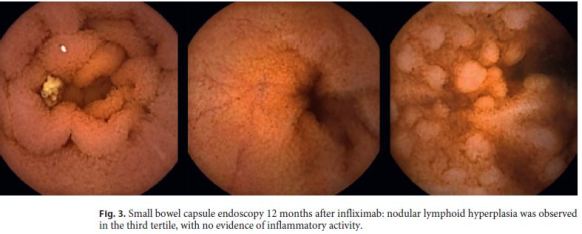
She was started on budesonide 9 mg/day, but abdominal pain and diarrhea persisted. After exclusion of active or latent tuberculosis, it was decided to initiate biological therapy with infliximab 5 mg/kg at 0, 2, and 6 weeks (induction) and, after that, every 8 weeks. After 14 weeks of infliximab, the patient showed clinical improvement and normalization of the acute phase reactants (erythrocyte sedimentation rate 3 mm and CRP < 2.9 mg/L) and fecal calprotectin (< 50 μg/g). A SBCE was performed 12 months after the onset of biological therapy to assess mucosal healing, which revealed nodular lymphoid hyperplasia with no ulcers or evidence of inflammatory activity (Fig. 3). No adverse effects, such as infections, were observed during the follow-up.
Discussion
Gastrointestinal manifestations in CVID are common, ranging from 20 to 60%. Up to 50% of patients have intermittent or chronic diarrhea, and 10% suffer from weight loss and malabsorption [8]. In the last years, the incidence of CVID-associated inflammatory enteropathy has ranged from 2 to 4% [5]. This clinical variability seems to be related to a complex pathogenesis with multiple mechanisms, suggesting a T-cell receptor-mediated pathway dysregulation with an imbalance of proinflammatory cytokines [9]. In a subgroup of patients, a persistent activation of TNF-α may contribute to the pathogenesis of CVID-associated inflammatory enteropathy [9]. Besides, recent data suggest that deficiencies of JAK1 and JAK3 are linked to combined immunodeficiency [10, 11]. In these patients, endoscopic appearance is not specific and is indistinguishable from inflammatory bowel disease patients without CVID [12]. Histological lesions of CVIDassociated inflammatory enteropathy show an absence of plasma cells in the lamina propria, lymphocytosis, apoptosis, granulomas, and crypt distortion [13].
SBCE played an important role in the diagnosis of this patient, proving the need, in gastrointestinal symptomatic patients with a history of CVID and normal endoscopy, to follow investigation, even though 50% of patients have chronic intermittent diarrhea. SBCE may also rule out complications of the disease and may establish small bowel extent.
There is no gold standard for therapeutic management of CVID-associated inflammatory enteropathy. Immunoglobulin replacement therapy, steroids, and immunosuppressive agents have been used, but their efficacy and tolerability are not well established [2, 13].
The first report in the literature was published in 2006 by Nos et al. [14], reporting on 2 young patients who were diagnosed with CVID-associated inflammatory enteropathy and were treated successfully with infliximab with no complications. Since then, some reports have been emerging, and anti-TNF-α drugs (infliximab and adalimumab) seem to be proper and powerful therapeutics in this type of patients with rapid control of severe symptoms [5, 15, 16].
In the current case, infliximab allowed clinical and laboratory remission and mucosal healing without increased bacterial infections. Despite limited experience with biological therapy in CVID-associated inflammatory enteropathy, emerging case reports have shown remarkable clinical and analytical improvement in these patients, supporting biological drugs as a first-line therapy [5, 15–17].
We would like to emphasize that SBCE has revolutionized the diagnosis and monitoring of Crohn disease, being a patient-friendly and noninvasive tool with proved advantages in diagnostic yield when compared to crosssectional imaging [18–20]. It allows a direct and detailed evaluation of small bowel mucosa with a high sensitivity for the earliest lesions, i.e., superficial lesions [18–20]. Regarding this case report, SBCE played a fundamental role in the diagnosis and in the monitoring of therapeutic response in this rare entity.
References
1 Abbott JK, Gelfand EW: Common variable immunodeficiency: diagnosis, management, and treatment. Immunol Allergy Clin North Am 2015;35:637–658. [ Links ]
2 Bonilla FA, Barlan I, Chapel H, et al: International Consensus Document (ICON): common variable immunodeficiency disorders. J Allergy Clin Immunol Pract 2016;4:38–59. [ Links ]
3 Ameratunga R, Brewerton M, Slade C, et al: Comparison of diagnostic criteria for common variable immunodeficiency disorder. Front Immunol 2014;5:415. [ Links ]
4 Saikia B, Gupta S: Common variable immunodeficiency. Indian J Pediatr 2016;83:338–344. [ Links ]
5 Vazquez-Moron JM, Pallares-Manrique H, Martin-Suarez IJ, Benitez-Rodriguez B, Ramos-Lora M: Crohns-like disease in a patient with common variable immunodeficiency treated with azathioprine and adalimumab. Rev Esp Enferm Dig 2013;105:299–302. [ Links ]
6 Cotter J, Dias de Castro F, Magalhaes J, Moreira MJ, Rosa B: Validation of the Lewis score for the evaluation of small-bowel Crohns disease activity. Endoscopy 2015;47:330–335. [ Links ]
7 Gralnek IM, Defranchis R, Seidman E, Leighton JA, Legnani P, Lewis BS: Development of a capsule endoscopy scoring index for small bowel mucosal inflammatory change. Aliment Pharmacol Ther 2008;27:146–154. [ Links ]
8 Cunningham-Rundles C: The many faces of common variable immunodeficiency. Hematology Am Soc Hematol Educ Program 2012;2012:301–305. [ Links ]
9 Cunningham-Rundles C: Common variable immunodeficiency. Curr Allergy Asthma Rep 2001;1:421–429. [ Links ]
10 Abolhassani H, Cheraghi T, Rezaei N, Aghamohammadi A, Hammarstrom L: Common variable immunodeficiency or late-onset combined immunodeficiency: a new hypomorphic JAK3 patient and review of the literature. J Investig Allergol Clin Immunol 2015;25:218–220. [ Links ]
11 Van Rompaey L, Galien R, van der Aar EM, et al: Preclinical characterization of GLPG0634, a selective inhibitor of JAK1, for the treatment of inflammatory diseases. J Immunol 2013;191:3568–3577. [ Links ]
12 Malamut G, Verkarre V, Suarez F, et al: The enteropathy associated with common variable immunodeficiency: the delineated frontiers with celiac disease. Am J Gastroenterol 2010;105:2262–2275. [ Links ]
13 Agarwal S, Mayer L: Diagnosis and treatment of gastrointestinal disorders in patients with primary immunodeficiency. Clin Gastroenterol Hepatol 2013;11:1050–1063. [ Links ]
14 Nos P, Bastida G, Beltran B, Aguas M, Ponce J: Crohns disease in common variable immunodeficiency: treatment with antitumor necrosis factor alpha. Am J Gastroenterol 2006;101:2165–2166. [ Links ]
15 Akazawa Y, Takeshima F, Yajima H, et al: Infliximab therapy for Crohns-like disease in common variable immunodeficiency complicated by massive intestinal hemorrhage: a case report. BMC Res Notes 2014;7:382. [ Links ]
16 Chua I, Standish R, Lear S, et al: Anti-tumour necrosis factor-alpha therapy for severe enteropathy in patients with common variable immunodeficiency (CVID). Clin Exp Immunol 2007;150:306–311. [ Links ]
17 Ruiz de Morales JG, Munoz F, Hernando M: Successful treatment of common variable immunodeficiency-associated inflammatory bowel disease with ustekinumab. J Crohns Colitis 2017;11:1154–1155. [ Links ]
18 Cotter J, Dias de Castro F, Moreira MJ, Rosa B: Tailoring Crohns disease treatment: the impact of small bowel capsule endoscopy. J Crohns Colitis 2014;8:1610–1615. [ Links ]
19 Dionisio PM, Gurudu SR, Leighton JA, et al: Capsule endoscopy has a significantly higher diagnostic yield in patients with suspected and established small-bowel Crohns disease: a meta-analysis. Am J Gastroenterol 2010;105:1240–1248;quiz 9. [ Links ]
20 Jensen MD, Nathan T, Rafaelsen SR, Kjeldsen J: Diagnostic accuracy of capsule endoscopy for small bowel Crohns disease is superior to that of MR enterography or CT enterography. Clin Gastroenterol Hepatol 2011;9:124–129. [ Links ]
Statement of Ethics
This study did not require informed consent nor review/approval by the appropriate ethics committee
Disclosure Statement
None of the authors have any conflicts of interest to declare.
Funding Sources
There were no funding sources.
* Corresponding author.
Dr. Cátia Arieira
Hospital da Senhora da Oliveira
Rua dos Cutileiros, Creixomil
PT–4835-044 Guimarães (Portugal)
E-Mail catia_arieira@hotmail.com
Received: September 4, 2017; Accepted after revision: December 21, 2017
Author Contributions
C. Arieira drafted the manuscript; F. Dias de Castro and M.J. Moreira revised the manuscript, and J. Cotter critically revised the manuscript and approved the final version to be submitted.














