Services on Demand
Journal
Article
Indicators
Related links
Share
GE-Portuguese Journal of Gastroenterology
Print version ISSN 2341-4545
GE Port J Gastroenterol vol.27 no.3 Lisboa June 2020
https://doi.org/10.1159/000502981
REVIEW ARTICLE
Nutritional Support of Cancer Patients without Oral Feeding: How to Select the Most Effective Technique?
Suporte nutricional no doente oncológico sem via oral: Como selecionar a técnica mais adequada?
Gonçalo Nunesa, Jorge Fonsecaa,b, Ana Teresa Barataa, Mário Dinis-Ribeiroc,d, Pedro Pimentel-Nunesc,d
aGastroenterology Department, GENE – Artificial Feeding Team, Hospital Garcia de Orta, Almada, Portugal; bCiiEM – Center for Interdisciplinary Research Egas Moniz, Monte da Caparica, Portugal; cGastroenterology Department, Instituto Português de Oncologia Francisco Gentil, Porto, Portugal; dCenter for Research in Health Technologies and Information Systems (CINTESIS), Faculty of Medicine, University of Porto, Porto, Portugal
* Corresponding author.
ABSTRACT
Background: Digestive tumours are among the leading causes of morbidity and mortality. Many cancer patients cannot maintain oral feeding and develop malnutrition. The authors aim to: review the endoscopic, radiologic and surgical techniques for nutritional support in cancer patients; address the strategies for nutritional intervention according to the selected technique; and establish a decision-making algorithm to define the best approach in a specific tumour setting. Summary: This is a narrative non-systematic review based on an electronic search through the medical literature using PubMed and UpToDate. The impossibility of maintaining oral feeding is a major cause of malnutrition in head and neck (H&N) cancer, oesophageal tumours and malignant gastric outlet obstruction. Tube feeding, endoscopic stents and gastrojejunostomy are the three main nutritional options. Nasal tubes are indicated for short-term enteral feeding. Percutaneous endoscopic gastrostomy (PEG) is the gold standard when enteral nutrition is expected for more than 3–4 weeks, especially in H&N tumour and oesophageal cancer patients undergoing definite chemoradiotherapy. A gastropexy push system may be considered to avoid cancer seeding. Radiologic and surgical gastrostomy are alternatives when an endoscopic approach is not feasible. Postpyloric nutrition is indicated for patients intolerant to gastric feeding and may be achieved through nasoenteric tubes, PEG with jejunal extension, percutaneous endoscopic jejunostomy and surgical jejunostomy. Oesophageal and enteric stents are palliative techniques that allow oral feeding and improve quality of life. Surgical or EUS-guided gastrojejunostomy is recommended when enteric stents fail or prolonged survival is expected. Nutritional intervention is dependent on the technique chosen. Institutional protocols and decision algorithms should be developed on a multidisciplinary basis to optimize nutritional care. Conclusions: Gastroenterologists play a central role in the nutritional support of cancer patients performing endoscopic techniques that maintain oral or enteral feeding. The selection of the most effective technique must consider the cancer type, the oncologic therapeutic program, nutritional aims and expected patient survival.
Keywords: Endoscopy, Nutrition, Cancer, Gastrointestinal obstruction
RESUMO
Introdução: O cancro digestivo encontra-se entre as principais causas de morbilidade e mortalidade globais. Uma fração significativa de doentes oncológicos não consegue manter a via oral no decurso da doença e desenvolve desnutrição. Os autores pretendem: rever as técnicas endoscópicas, radiológicas e cirúrgicas de suporte nutricional em doentes com cancro; abordar as estratégias de intervenção nutricional tendo em conta a técnica escolhida; estabelecer algoritmos de decisão para definir a melhor abordagem técnica no contexto de neoplasias específicas. Sumário: Revisão narrativa não sistemática, realizada através da pesquisa eletrónica na literatura médica com recurso à PubMed e UpToDate. A impossibilidade de manter a via oral é uma causa major de desnutrição em doentes com neoplasias cervicofaciais, cancro do esófago e tumores que condicionam obstrução do trato de saída gástrico. A alimentação por tubo (tube feeding), as próteses metálicas autoexpansíveis (PMAE) colocadas por via endoscópica e a gastrojejunostomia são as três principais abordagens para suporte nutricional. As sondas nasais estão indicadas para nutrição entérica de curta duração. A gastrostomia endoscópica percutânea (PEG) é a técnica gold-standard sempre que se antecipa a necessidade de nutrição entérica por um período superior a 3–4 semanas, em particular nos doentes com neoplasias cervicofaciais e tumores do esófago com indicação para quimiorradioterapia definitiva. A utilização de um sistema push com gastropexia pode ser equacionada para evitar o risco de metastização no estoma. A gastrostomia radiológica e cirúrgica são alternativas válidas quando a abordagem endoscópica não é possível. A nutrição pós-pilórica está recomendada nos doentes intolerantes à alimentação intragástrica, podendo ser realizada através de sondas nasoentéricas, PEG com extensão jejunal, jejunostomia endoscópica percutânea ou jejunostomia cirúrgica. As PMAE esofágicas e entéricas estão indicadas para paliação, permitindo manter a via oral e melhorar a qualidade de vida. A gastrojejunostomia cirúrgica ou guiada por ecoendoscopia deve ser equacionada perante a falência técnica das próteses entéricas ou alternativamente quando uma sobrevida prolongada é expectável. As estratégias de intervenção nutricional dependem da técnica selecionada. Protocolos institucionais e algoritmos de decisão devem ser desenvolvidos em contexto multidisciplinar para otimizar os cuidados nutricionais prestados ao doente oncológico. Conclusões: Os gastrenterologistas desempenham um papel central no suporte nutricional do doente oncológico com a realização de procedimentos endoscópicos que permitem manter a via oral e/ou entérica. A seleção da técnica mais adequada deve ter em conta a neoplasia subjacente, a estratégia terapêutica, os objetivos nutricionais e a sobrevida esperada.
Palavras-Chave: Endoscopia, Nutrição, Cancro, Obstrução gastrointestinal
Introduction
Cancer is among the leading causes of morbidity and mortality worldwide [1]. Digestive oncology is a major area of gastroenterology practice, comprising a wide range of diseases responsible for a large number of admissions to gastroenterology wards and endoscopy units. The incidence of gastrointestinal (GI) tumours has been increasing during the last years, and nowadays it accounts for 10% of the overall mortality in Portugal [2, 3].
Most patients with digestive tumours have a high nutritional risk [4]. The European Society for Clinical Nutrition and Metabolism defines malnutrition as a state resulting from lack of uptake of nutrients that leads to an altered body composition with an impaired clinical outcome. The diagnostic criteria for malnutrition have recently been reviewed, requiring a BMI below 18.5 kg/m2 or, alternatively, unintentional weight loss associated with either a reduced BMI or a low fat free mass index [5].
Weight loss is extremely common amongst cancer patients, and malnutrition occurs in 40–80% of cases, leading to poor outcome in terms of mortality, tolerance to chemotherapy, infection risk and worse surgical results [6–8]. Cancer-related malnutrition is caused by (1) inability to ingest food due to lumen obstruction that causes dysphagia and recurrent vomiting; (2) harmful effects of surgery and medical treatment that prevent an adequate nutritional intake due to swallowing dysfunction, severe mucositis, nausea and anorexia; and (3) metabolic modifications induced by the cancer-associated systemic inflammatory response [6–9]. Nutritional counselling and oral nutritional supplements (ONS) are the first-line approach in the nutritional support of cancer patients. Enteral nutrition through tube feeding is recommended if inability to eat for more than 7 days is anticipated or the dietary intake is below 60% of the estimated requirements for more than 10 days [9–11].
Upper GI obstruction is a usual consequence of digestive cancer with a potential negative impact on patients nutritional status and quality of life [4, 6]. Head and neck (H&N) cancer, particularly pharyngeal and laryngeal lesions, oesophagogastric tumours and all neoplasia causing gastric outlet obstruction (GOO), such as tumours of pancreatic, hepatobiliary and duodenal origin, are important causes of intolerance to oral intake [12–15]. In fact, gastroenterologists play a central role in the nutritional management of cancer patients through their endoscopic techniques that allow maintenance of oral and/or enteral feeding. Radiologic and surgical interventions are alternatives if an endoscopic approach is not feasible.
The present article aims to: (1) review the endoscopic, radiologic and surgical techniques used for nutritional support of cancer patients; (2) address the strategies for nutritional intervention according to the selected technique; and (3) establish a decision-making algorithm to define the best technique to be applied in a specific tumour setting.
2 Materials and Methods
A narrative non-systematic review was performed based on an electronic search through the medical literature using PubMed and UpToDate. The keywords “Cancer,” “Malnutrition,” “Gastrointestinal Obstruction,” “Head and Neck Cancer,” “Esophageal Cancer,” “Gastric Outlet Obstruction,” “Enteral Nutrition,” “PEG,” “Gastrostomy,” “Jejunostomy,” “Esophageal Stent,” “Enteric Stent” and “Gastrojejunostomy” were used. English-language articles were considered. No publication time restriction was defined; nevertheless, review articles and guidelines from gastroenterology, endoscopy, nutrition and cancer societies that have been published during the last 10 years were prioritized. Data regarding malnutrition in cancer patients and techniques for nutritional support were included. Decision-making algorithms are proposed by the authors at the end of this review.
3 Results and Discussion
3.1 Techniques of Nutritional Support for Cancer Patients without Oral Feeding
Dysphagia, odynophagia and GOO are common consequences of cancer [12–15]. Several techniques are available to deal with these conditions and optimize nutritional support mostly in two different situations: when patients are under cancer treatment with curative intent (chemotherapy, radiotherapy or awaiting surgical resection) or when they are referred for symptom palliation in cases of advanced disease and a poor performance status. The techniques may be grouped into three main types, namely tube feeding, endoscopic stents and gastrojejunostomy (GJ). Table 1 summarizes the main points of each technique.
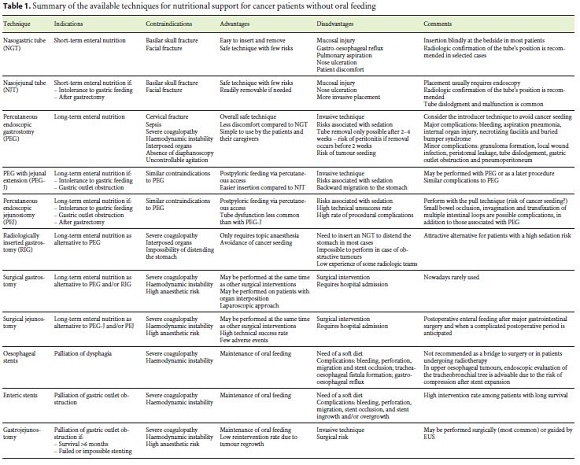
3.1.1 Tube Feeding
Tube feeding is the cornerstone of enteral nutrition and plays an important role in patients with H&N and oesophageal cancers. It may be performed using nasal or transabdominal tubes placed using endoscopic or nonendoscopic methods [11, 16].
3.1.1.1 Nasal Tubes
Nasogastric tubes (NGT) and nasojejunal tubes (NJT) are flexible single- or double-lumen tubes that pass proximally from the nose into the stomach or small bowel, allowing gastric or postpyloric feeding. NGT are made of polyvinyl chloride, polyurethane or silicone and display several different sizes. The more flexible and less traumatic silicone or polyurethane tubes are therefore preferred. Although no specific recommendations regarding the material and size of NGT were provided given the absence of clinical studies, it should be noted that thinner tubes may be more comfortable for patients but thicker tubes allow easy insertion of home-made formulas and medications. NJT are more flexible, have smaller diameters and vary in length. Most NGT are placed safely using a blind technique. NJT and NGT placement in patients with partial obstructive lesions or large hiatal hernias often requires endoscopic or fluoroscopic guidance. Routine radiologic confirmation of the NJT position is advisable and may also be considered with NGT if insertion was not easy and some doubt about the intragastric position of the tip persists [17, 18].
Nasal tubes are generally recommended for a short period, particularly if tube feeding is not anticipated to exceed 3–4 weeks, given the associated discomfort and the potential risks of a more prolonged use [19]. However, they may be considered the first-line technique for patients with obstructive tumours proximal to the cardia in order to initiate nutritional support without delay. NJT may also be inserted to bypass lesions located distally to the stomach, in gastrectomized patients and in those who do not tolerate gastric feeding due to recurrent aspiration, severe gastro-oesophageal reflux, gastroparesis and GOO [20].
3.1.1.2 Transabdominal Tubes
Transabdominal feeding tubes may be placed using an endoscopic, radiologic or surgical approach. Although endoscopic tube placement is usually considered the first option as it is simple, practical and readily available, alternative techniques may be necessary, especially if abdominal wall transillumination is not achieved (due to overweight, visceral interposition or the presence of large hiatal hernias) or access to the stomach/jejunum is prevented by complete pharyngo-oesophageal obstruction. The choice of the procedure will depend on local expertise, anatomic considerations and whether the patient is undergoing surgery for other reasons.
3.1.1.2.1 Endoscopic Tube Placement
3.1.1.2.1.1 Percutaneous Endoscopic Gastrostomy
Percutaneous endoscopic gastrostomy (PEG) is a safe and effective technique for long-term enteral nutrition. It is recommended if dysphagia is anticipated for more than 3–4 weeks [10, 18–21]. There are several commercially available PEG kits, mostly involving a pull-through technique. Tube calibres usually range from 12 to 24 Fr; however, a study comparing 12- and 20-Fr PEG tubes showed no difference regarding complications or long-term patency [22]. Other systems (Freka® Pexact and Cook® Medical) may also be used to perform a gastropexy, allowing percutaneous introduction of the gastrostomy tube under endoscopic guidance, which avoids passage of the PEG tube through the superior aerodigestive structures (introducer technique) [23].
Actually, patients with proximal GI cancers are at risk of seeding from the primary tumour site to the PEG tract by mechanical transfer. In 1989, Preyer and Thul [24] reported the first patient with PEG tract metastasis, and since then, new cases have been identified annually [24–27]. In a recent prospective controlled study involving 50 patients, positive cytology for malignant cells was described in 22.5% of the stomas immediately after PEG placement using the pull method, and after 3–6 months of follow-up, persistence of these cells was detected in 9%. Although no patient developed clinically manifest stoma metastasis, most had advanced disease at the time of PEG placement and died before the end of the study. There was a trend for short survival in the 9% group of patients with microscopic cancer seeding [28]. Although clinically evident metastasis seems to be uncommon, the median time for its appearance was described as 6–8 months and its incidence has been underdiagnosed due to the short patient follow-up after PEG that was reported in different series [29]. The incidence rate has been estimated at 0.1–3% amongst patients with H&N cancer. Although some cases of favourable evolution have been reported, this adverse event is often associated with a poor oncologic outcome [24, 26, 29]. Thus, the use of a direct gastric puncture technique for percutaneous feeding tube placement in patients with proximal GI cancer has been recommended by several authors, especially in patients receiving radical therapy with curative intent [20, 25]. Comparative studies have shown similar complication rates, except for a more common occurrence of delayed tube disfunction and minor bleeding episodes with the introducer approach [30, 31].
H&N tumours usually compromise swallowing due to involvement of the mouth, pharynx and upper oesophageal sphincter. These patients often undergo high-dose chemoradiotherapy that also induces severe mucositis. Chemoradiotherapy is the base for oesophageal cancer treatment, as only a few candidates are suitable for direct surgery [32]. Thus, nutritional intake may become markedly reduced [33]. In patients undergoing chemoradiotherapy for H&N tumours, PEG is associated with low procedure-related morbidity and may provide better nutritional outcomes than nasogastric tube feeding [20]. However, the timing of PEG tube placement in this clinical setting has not been clearly established [33]. PEG feeding was traditionally started after swallowing impairment or a compromised nutritional status. In contrast, some authors have reported the beginning of enteral nutrition prior to treatment, showing that prophylactic PEG placement and early tube feeding were associated with limited weight loss, allowing effective and safe nutrition during chemoradiotherapy [34–36]. Current evidence does not show significant differences in survival rates with the use of prophylactic PEGs; nevertheless, the potentially high risk of sedation and procedure complications in patients who have undergone PEG with established malnutrition and a poor performance status should also be considered [37]. The evidence to support the ideal timing of PEG in patients undergoing H&N cancer treatment is weak and should be decided case by case considering the baseline nutritional status. Close nutritional and clinical monitoring is essential to identify patients who will benefit from an early PEG to avoid nutritional deterioration. This must be balanced with some evidence suggesting that gastrostomy leads to more prolonged tube dependency and long-term dysphagia [38–40].
Concerns that PEG may render the stomach unusable for the formation of a gastric tube at the time of oesophagectomy in patients with oesophageal cancer have also been raised by some authors. Despite certain guidelines recommending against its routine use, recent studies have described no impact on postoperative outcomes. Local surgeon preferences and technical skills should be taken into account [20, 41].
3.1.1.2.1.2 PEG with Jejunal Extension and Percutaneous Endoscopic Jejunostomy
PEG with jejunal extension (PEG-J) and direct percutaneous endoscopic jejunostomy (PEJ) allow direct administration of feed into the small bowel. These procedures are recommended if tube feeding is indicated for more than 3–4 weeks, as an alternative to NJT [20]. Most experience was obtained with PEG-J, in which a jejunal tube is passed through the PEG tube at the time of PEG placement or as a separate procedure using a previously inserted PEG. The procedure is easier if PEG is performed on the gastric antrum, allowing a more direct route for the extension tube to cross the pylorus and reducing the risk of gastric looping, which may favour further displacement. Backward migration to the stomach is a common event, which may be reduced if the tip is initially placed beyond the ligament of Treitz [20]. In order to reduce repeated manipulation and the risk of gastric migration of the jejunal tube, the connection of a drainage PEG to the feeding PEG-J was proposed for GOO patients [42, 43].
PEJ is an alternative approach, involving the direct placement of the feeding tube into the jejunum. A paediatric colonoscope or an enteroscope is needed unless the patient had a prior gastrectomy. Standard PEG tube kits can be used for PEJ, but the selection of a small-sized internal bumper will minimize the risk of occluding the small bowel. The other principles of the technique and postprocedural care are the same as with PEG; however, significant failure of tube placement (15–20%) and more frequent complications were described [32].
Either PEG-J or PEJ should be offered as the primary procedure for patients who do not tolerate gastric feeding due to severe gastro-oesophageal reflux, delayed gastric emptying, regurgitation or recurrent aspiration. PEJ should be considered if recurrent failure of PEG-J develops. Tube dysfunction and the need for reintervention are significantly lower with PEJ, which should be preferred if long-term postpyloric feeding is indicated. Nevertheless, there are conflicting data regarding whether jejunal feeding truly reduces the rate of reflux and aspiration [20].
3.1.1.2.2 Non-Endoscopic Tube Placement
3.1.1.2.2.1 Radiologically Inserted Gastrostomy
Radiologically inserted gastrostomy (RIG) is performed using fluoroscopic guidance. The stomach is distended with air using an NGT, and the site for abdominal puncture is selected with fluoroscopy. A gastropexy is first accomplished. Air aspiration confirms the intragastric location and a guidewire is advanced. The gastrostomy tube is placed over the guidewire. At the end of the procedure, contrast is injected to confirm the correct tube position. Whether there is a difference in morbidity and mortality rates between endoscopic and radiologic gastrostomy tube placement is not clear. RIG may be considered for patients with H&N or oesophageal cancers in order to avoid tumour seeding to the gastrostomy tract if a PEG cannot be performed using the introducer technique. Since RIG just commonly requires abdominal topical anaesthesia with no need of sedation, it may also be a useful technique for patients with significant comorbidities and a high anaesthetic risk. Nevertheless, although no endoscopy is necessary, RIG usually requires the passage of an NGT, which may be impossible in some cancer patients. Furthermore, few radiology teams seem to have acquired extensive experience with this technique [44–48].
3.1.1.2.2.2 Surgical Gastrostomy
Surgical gastrostomy (SG) can be performed via a laparoscopic or an open approach. Laparoscopic gastrostomy is performed similarly to RIG, using T-fasteners to affix the stomach to the abdominal wall before the tube is placed. Open gastrostomy involves a gastric incision to place the tube. Studies comparing SG with PEG have shown no differences in morbidity and mortality rates; nevertheless, PEG is less expensive and is easier and faster to perform [49]. SG is commonly reserved for patients who will undergo another surgical procedure or in a setting in which PEG and RIG are contraindicated or not locally available [49, 50].
3.1.1.2.2.3 Surgical Jejunostomy
Fine needle catheter jejunostomy is a common alternative often achieved during upper digestive tract surgery such as oesophagectomy, gastrectomy and pancreatic interventions. It is mainly performed on patients for whom a complicated postoperative recovery is expected; those with a prolonged fasting period being at risk of malnutrition; or patients who will subsequently need chemotherapy or radiotherapy and have not previously had a percutaneous feeding tube placed. With this technique, a largebore needle is tunnelled through the subserosa before entering the jejunal lumen, and then a 8- to 9-Fr feeding catheter is inserted and further exteriorized through the abdominal wall. Surgical jejunostomy is an effective approach providing enteral access for early enteral nutrition and to deliver medications in the postoperative period. This technique is also useful to have a prolonged enteral access in patients who develop surgical complications [51, 52]. Radiologic jejunostomy was also addressed by some authors as a non-surgical alternative; however, the available data are still limited [53].
3.1.2 Endoscopic Stent Placement
Self-expandable metal stents (SEMS) are endoscopic devices commonly used for the non-surgical management of obstructive GI neoplasms, particularly oesophageal cancer, extra-oesophageal malignancies that compress or invade the oesophagus and tumours that cause GOO, namely gastric and pancreatic cancers. Since they allow maintenance of oral feeding, a positive impact on nutrition and quality of life may be expected.
3.1.2.1 Oesophageal Stents
In recent years, different designs of oesophageal stents have emerged to improve dysphagia in patients with malignant oesophageal tumours, tracheo-oesophageal fistulas or extrinsic compression. They comprise (1) uncovered SEMS; (2) fully covered SEMS, in which the entire length of the stent is covered; (3) partially covered SEMS, in which the proximal and distal ends of the stent are devoid of a covering; and (4) fully covered self-expandable plastic stents. Fully and partially covered SEMS are predominantly used for the treatment of malignant dysphagia, and self-expandable plastic stents do not appear to present advantages over SEMS in this clinical setting [54].
More than 95% of cancer patients undergoing oesophageal stent placement show improvement in dysphagia, and 75–100% of those who present with tracheo-oesophageal fistulas achieve successful fistula closure. However, subsequent tumour ingrowth or overgrowth is common and requires additional interventions for recurrent symptoms in up to 50% of cases [55, 56].
Oesophageal stenting for malignant obstruction is nowadays recommended as a palliation technique for patients with unresectable tumours or metastatic disease, or for individuals unfit for curative treatment due to a poor performance status. Current guidelines do not advise SEMS placement in dysphagic patients as a bridge to surgery or prior to chemoradiotherapy, since it is associated with adverse events such as stent migration (35%) [54]. More importantly, a negative impact on oncologic outcomes was also documented, including lower rates of R0 resection, a lower median time to recurrence, and increased morbidity and mortality. Furthermore, in palliative patients who will undergo palliative radiotherapy, SEMS placement should be postponed until the end of this therapy. In these circumstances, if the patient has to start enteral nutritional support, tube feeding should be considered [55–60].
3.1.2.2 Enteric Stents
The main goal of enteral stenting for malignant gastroduodenal obstruction is to restore lumen patency in symptomatic patients with unresectable malignancy. Enteric stents may be covered or uncovered, with the latter being preferred due to the lower risk of migration; nevertheless, tumour ingrowth is a potential disadvantage. Stent placement is not recommended if GOO is asymptomatic and the patient is tolerating a normal diet, nor as a bridge to surgery. Ideally, individuals should have a short life expectancy from less than 2 to 6 months [61, 62]. The presence of other sites of small bowel obstruction should be investigated, since stenting of a proximal stricture is unlikely to be clinically effective [63]. However, contrary to what would be expected, some data suggest that enteral stent placement may actually work in patients with peritoneal carcinomatosis, as was described by Mendelsohn et al. [64] in a retrospective analysis of 215 patients, who found similar outcomes regarding clinical success, need for reintervention and major complications. Actually, the decision on whether to palliate GOO patients with peritoneal carcinomatosis using SEMS remains empirical and need to be taken case by case until further clinical studies are available.
In patients with coexistent biliary obstruction, a selfexpandable metal biliary stent should be considered prior to – or in the same endoscopic period as – gastroduodenal stent placement, because it may be harder to achieve biliary cannulation once a duodenal stent has been deployed across the papilla [65, 66].
Enteric stents can be placed successfully in over 90% of patients, with clinical success rates above 80%. Although they are associated with less morbidity and lower procedure-related mortality rates and costs, when compared with surgical palliation, 15–40% of patients require reintervention for recurrent symptoms. Also, abnormal gastric motility caused by neural tumour involvement may prevent any significant improvement in some patients after GOO palliation [61, 67–70].
3.1.3 Gastrojejunostomy
3.1.3.1 Surgical Gastrojejunostomy
GJ is a surgical procedure in which an anastomosis is created between the stomach and a proximal loop of the jejunum. This technique is an alternative to gastric drainage if GOO occurs, allowing oral feeding, and it can be performed via a laparoscopic, minimally invasive approach. GJ is usually reserved for palliation in patients who have failed or are not candidates for gastroduodenal stents. When compared to stenting, palliative GJ is associated with lower reintervention rates and more long-term symptom relief without significant differences in mortality rates. It may be considered if patients life expectancy is longer than 6 months [70, 71].
3.1.3.2 EUS-Guided Gastrojejunostomy
EUS-guided GJ is an innovative procedure that has been described as an alternative to surgical GJ. In this procedure, an endosonographic placement of a lumen-apposing metal stent is performed to create a gastrojejunal anastomosis, which bypasses the gastroduodenal obstruction [72, 73]. A recent international multicentre study that included 26 patients showed a technical and clinical success rate of 92 and 85%, respectively [73]. When compared to laparoscopic GJ, EUS-guided GJ seems to be associated with fewer adverse events and lower costs, as was described by a Spanish group in a comparative study [74]. Further studies comparing this approach with luminal stent placement are warranted.
3.2 Nutritional Intervention for Cancer Patients without Oral Feeding
Nutrition intervention is mandatory in cancer therapy. For these patients, the energy and protein requirements are estimated to be 25–30 kcal/kg/day and 1.2–1.5 g of protein/kg/day, respectively. If severe protein depletion occurs, higher doses up to 2.0 g/kg/day may be necessary to maintain or restore the lean body mass [4, 8, 73]. In malnourished patients or those at risk of refeeding syndrome, feeding should be performed gradually, starting with 25% of the estimated requirements and slowly increasing the rate over 4–7 days [75–78].
Prepyloric tube feeding enables large volumes and high osmotic loads. For patients with NGT, PEG, RIG, SG or GJ, standard and fibre-enriched formulas can be safely and effectively employed. High-energy and highprotein formulas may also be considered, depending on the patients nutritional requirements, volume tolerance and GI symptoms. Infusion into the stomach allows either bolus or continuous feeding and higher feeding rates. If not tolerated, switching to continuous infusion can reduce discomfort and maximize nutritional support [79–82]. In postpyloric feeding, osmolality and volume may become relevant. For patients with NJT, PEG-J, PEJ or SJ, isotonic formulas with continuous feeding at a controlled rate are needed due to the lack of small bowel reservoir capacity. Jejunal feeding does not stimulate pancreatic secretions, although most studies support polymeric formulas. Whole protein feeds may be well tolerated and should be standard practice. If malabsorption occurs, a period of hydrolysed protein feed is recommended [78, 83, 84].
As an alternative to commercial enteral formulas, home-made blenderized tube feedings can be provided. Nevertheless, they are more likely to cause tube blockage, are often nutritionally unbalanced and present an increased risk of bacterial contamination. Also, they are not suitable for patients on postpyloric feeding [78, 85, 86].
After oesophageal/enteric stenting, oral feeding should be initiated with fluids and gradually progress to a soft diet. To prevent blockage, food should be very well chewed and have a moist and soft swallowed consistence. Recommendations state several small daily meals with adequate fluid intake avoiding high-fibre, dry and stringy foods. If nutrient intake remains inadequate with normal food, ONS should be prescribed [87].
Clinical studies have reported positive results with immunonutrient supplementation in cancer patients; n–3 polyunsaturated fatty acids and arginine supplements proved to be beneficial, improving inflammatory markers and body weight, maintaining the lean body mass and reducing chemoradiotherapy-induced mucositis [4, 8, 81, 87–89]. Immunoenhancing enteral formulas containing arginine, nucleic acids and n–3 polyunsaturated fatty acids are also recommended for patients undergoing major cancer surgery [79, 90].
Early referral for dietary counselling should be considered at the time of diagnosis in order to adopt the best individualized nutritional intervention during the course of cancer treatment.
3.3 Algorithms for Decision-Making in Clinical Practice
3.3.1 Nutritional Support in H&N Cancer
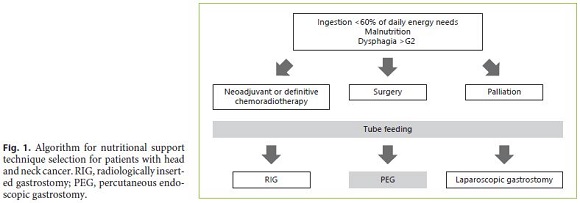
H&N cancer patients should promptly start tube feeding if malnutrition is established or when grade 3–4 dysphagia develops. If oral ingestion targeting at least 60% of daily energy needs is not possible even through ONS, tube feeding is recommended. This may occur prior to or during cancer treatment. NGT should be used as a transient approach, but PEG is indicated for long-term enteral nutrition. Given its equivalent safety to that of the pull method, the gastropexy push system (introducer technique) may be considered for cancer patients treated with a curative intent; nevertheless, the evidence for its routine application to all patients is scarce, as cancer seeding seems to be infrequent and the overall risk low. RIG is an alternative if there is a high anaesthetic risk, and SG is reserved for patients who fail a less invasive approach (Fig. 1).
3.3.2 Nutritional Support in Oesophageal Cancer
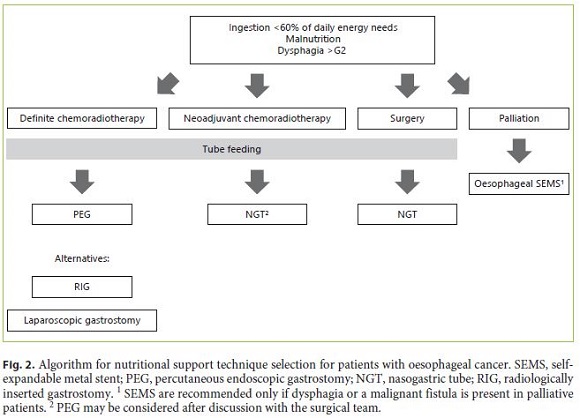
Patients with oesophageal tumours undergoing definitive chemoradiotherapy should receive tube feeding through PEG. RIG or laparoscopic gastrostomy are alternatives as in H&N cancer. For patients eligible for surgical resection who need preoperative tube feeding, NGT should be used, although the possibility of PEG may be considered after discussion with the surgical team. Oesophageal SEMS are indicated for palliation, but they should be postponed in patients undergoing palliative radiotherapy. They may be an effective choice to palliate dysphagia in obstructive tumours involving the proximal, middle and distal oesophagus (Fig. 2).
3.3.3 Nutritional Support in Gastric Outlet
Obstructive Cancers
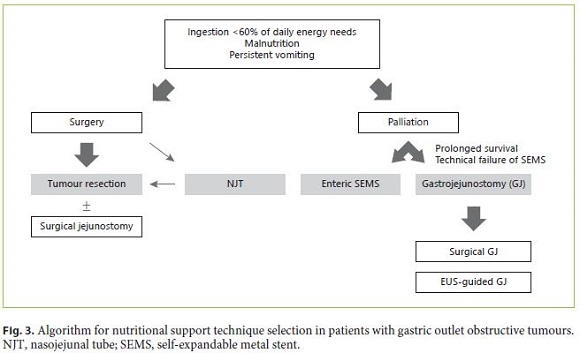
Surgical resection is the best option to manage patients with GOO undergoing curative therapy. NJT feeding may be a transient approach to optimize the nutritional status before surgery; nevertheless, neoadjuvant therapy is not indicated for patients with obstructive tumours. Surgical jejunostomy is an alternative method that may be performed at the time of surgery, allowing effective postoperative nutritional support. In palliative patients, enteric SEMS can effectively manage GOO, especially if the expected survival is below 6 months. If the life expectancy is longer or SEMS cannot be placed or fail, GJ is the best option. The choice between a surgical and an EUS-guided approach will depend on the available skills and expertise (Fig. 3).
4 Conclusions
Digestive tumours and H&N cancer are complex and heterogeneous disorders that usually impair oral feeding. Gastroenterologists play a central role in the nutritional support of these patients by performing endoscopic techniques that maintain oral and enteral nutrition. When selecting the most adequate technique one must consider the cancer type, the oncologic therapeutic program, the nutritional aims and the patients expected survival. Nutritional recommendations in cancer require an interdisciplinary approach. Patients should be routinely evaluated and have an individualized nutritional plan aiming to prevent malnutrition and improve cancer-related outcomes.
References
1 Nagai H, Kim YH. Cancer prevention from the perspective of global cancer burden patterns. J Thorac Dis. 2017 Mar;9(3):448–51.
2 Cremers MI. The European Section and Board of Gastroenterology and Hepatology: whats new? GE Port J Gastroenterol. 2017 May;24(3):105–7.
3 Pedroto I, Nunes S, Matos L, Maçoas F, Macedo G, Cotter J, et al. Rede Nacional de Especialidade Hospitalar e de Referenciação – Gastrenterologia e Hepatologia [accessed Jan 2019]. Available from: https://www.sns.gov.pt.
4 Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, et al. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017 Feb;36(1):11–48.
5 Cederholm T, Bosaeus I, Barazzoni R, Bauer J, Van Gossum A, Klek S, et al. Diagnostic criteria for malnutrition – an ESPEN Consensus Statement. Clin Nutr. 2015 Jun;34(3):335–40.
6 Nitenberg G, Raynard B. Nutritional support of the cancer patient: issues and dilemmas. Crit Rev Oncol Hematol. 2000 Jun;34(3):137–68.
7 Lees J. Incidence of weight loss in head and neck cancer patients on commencing radiotherapy treatment at a regional oncology centre. Eur J Cancer Care (Engl). 1999 Sep;8(3):133–6.
8 Arends J, Baracos V, Bertz H, Bozzetti F, Calder PC, Deutz NE, et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin Nutr. 2017 Oct;36(5):1187–96.
9 Evans WJ, Morley JE, Argilés J, Bales C, Baracos V, Guttridge D, et al. Cachexia: a new definition. Clin Nutr. 2008 Dec;27(6):793–9.
10 Lochs H, Valentini L, Schütz T, Allison SP, Howard P, Pichard C, et al. ESPEN Guidelines on adult enteral nutrition. Clin Nutr. 2006 Apr;25:177–360.
11 McClave SA, DiBaise JK, Mullin GE, Martindale RG. ACG Clinical Guideline: Nutrition Therapy in the Adult Hospitalized Patient. Am J Gastroenterol. 2016 Mar;111(3):315–34.
12 Nugent B, Lewis S, OSullivan JM. Enteral feeding methods for nutritional management in patients with head and neck cancers being treated with radiotherapy and/or chemotherapy. Cochrane Database Syst Rev. 2013 Jan;(1):CD007904. [ Links ]
13 Chen MJ, Wu IC, Chen YJ, Wang TE, Chang YF, Yang CL, et al. Nutrition therapy in esophageal cancer – consensus statement of the Gastroenterological Society of Taiwan. Dis Esophagus. 2018 Aug;31(8).
14 Tendler DA. Malignant gastric outlet obstruction: bridging another divide. Am J Gastroenterol. 2002 Jan;97(1):4–6.
15 Adler DG, Baron TH. Endoscopic palliation of malignant gastric outlet obstruction using self-expanding metal stents: experience in 36 patients. Am J Gastroenterol. 2002 Jan;97(1):72–8.
16 Cederholm T, Barazzoni R, Austin P, Ballmer P, Biolo G, Bischoff SC, et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin Nutr. 2017 Feb;36(1):49–64.
17 Best C. How to set up and administer an enteral feed via a nasogastric tube. Nurs Stand. 2017 Jul;31(45):42–7.
18 Kwon RS, Banerjee S, Desilets D, Diehl DL, Farraye FA, Kaul V, et al.; ASGE Technology Committee. Enteral nutrition access devices. Gastrointest Endosc. 2010 Aug;72(2):236–48.
19 Löser C, Aschl G, Hébuterne X, Mathus-Vliegen EM, Muscaritoli M, Niv Y, et al. ESPEN guidelines on artificial enteral nutrition – percutaneous endoscopic gastrostomy (PEG). Clin Nutr. 2005 Oct;24(5):848–61.
20 Westaby D, Young A, OToole P, Smith G, Sanders DS. The provision of a percutaneously placed enteral tube feeding service. Gut. 2010 Dec;59(12):1592–605.
21 Blumenstein I, Shastri YM, Stein J. Gastroenteric tube feeding: techniques, problems and solutions. World J Gastroenterol. 2014 Jul;20(26):8505–24.
22 Duncan HD, Bray MJ, Kapadia SA, Bowling TE, Cole SJ, Gabe SM, et al. Prospective randomized comparison of two different sized percutaneous endoscopically placed gastrostomy tubes. Clin Nutr. 1996 Dec;15(6):317–20.
23 Schaefer H, Vivaldi C, Herbold T, Brinkmann S, Maus M, Bludau M, et al. Percutaneous Endoscopic Gastrostomy in Direct Puncture Technique: When, Why and How? Dig Surg. 2017;34(1):52–9.
24 Preyer S, Thul P. Gastric metastasis of squamous cell carcinoma of the head and neck after percutaneous endoscopic gastrostomy – report of a case. Endoscopy. 1989 Nov;21(6):295.
25 Greaves JR. Head and Neck Cancer Tumor Seeding at the Percutaneous Endoscopic Gastrostomy Site. Nutr Clin Pract. 2018 Feb;33(1):73–80.
26 Fonseca J, Adriana C, Fróis-Borges M, Meira T, Oliveira G, Santos JC. Ostomy metastasis after pull endoscopic gastrostomy: a unique favorable outcome. Nutr Hosp. 2015 Apr;31(4):1879–81.
27 Cappell MS. Risk factors and risk reduction of malignant seeding of the percutaneous endoscopic gastrostomy track from pharyngoesophageal malignancy: a review of all 44 known reported cases. Am J Gastroenterol. 2007 Jun;102(6):1307–11.
28 Ellrichmann M, Sergeev P, Bethge J, Arlt A, Topalidis T, Ambrosch P, et al. Prospective evaluation of malignant cell seeding after percutaneous endoscopic gastrostomy in patients with oropharyngeal/esophageal cancers. Endoscopy. 2013 Jul;45(7):526–31.
29 Rowell NP. Tumor implantation following percutaneous endoscopic gastrostomy insertion for head and neck and oesophageal cancer: review of the literature. Head Neck. 2019 Jun;41(6):2007–15.
30 Köhler G, Kalcher V, Koch OO, Luketina RR, Emmanuel K, Spaun G. Comparison of 231 patients receiving either “pull-through” or “push” percutaneous endoscopic gastrostomy. Surg Endosc. 2015 Jan;29(1):170–5.
31 Retes FA, Kawaguti FS, de Lima MS, da Costa Martins B, Uemura RS, de Paulo GA, et al. Comparison of the pull and introducer percutaneous endoscopic gastrostomy techniques in patients with head and neck cancer. United European Gastroenterol J. 2017 Apr;5(3):365–73.
32 Allum WH, Blazeby JM, Griffin SM, Cunningham D, Jankowski JA, Wong R; Association of Upper Gastrointestinal Surgeons of Great Britain and Ireland, the British Society of Gastroenterology and the British Association of Surgical Oncology. Guidelines for the management of oesophageal and gastric cancer. Gut. 2011 Nov;60(11):1449–72.
33 Fonseca J, Santos CA, Brito J. Malnutrition and Clinical Outcome of 234 Head and Neck Cancer Patients who Underwent Percutaneous Endoscopic Gastrostomy. Nutr Cancer. 2016 May-Jun;68(4):589–97.
34 Hardy S, Haas K, Vanston VJ, Angelo M. Prophylactic Feeding Tubes in Head and Neck Cancers #318. J Palliat Med. 2016 Dec;19(12):1343–4.
35 Lee JH, Machtay M, Unger LD, Weinstein GS, Weber RS, Chalian AA, et al. Prophylactic gastrostomy tubes in patients undergoing intensive irradiation for cancer of the head and neck. Arch Otolaryngol Head Neck Surg. 1998 Aug;124(8):871–5.
36 Williams GF, Teo MT, Sen M, Dyker KE, Coyle C, Prestwich RJ. Enteral feeding outcomes after chemoradiotherapy for oropharynx cancer: a role for a prophylactic gastrostomy? Oral Oncol. 2012 May;48(5):434–40.
37 Langius JA, Zandbergen MC, Eerenstein SE, van Tulder MW, Leemans CR, Kramer MH, et al. Effect of nutritional interventions on nutritional status, quality of life and mortality in patients with head and neck cancer receiving (chemo)radiotherapy: a systematic review. Clin Nutr. 2013 Oct;32(5):671–8.
38 McClelland S 3rd, Andrews JZ, Chaudhry H, Teckie S, Goenka A. Prophylactic versus reactive gastrostomy tube placement in advanced head and neck cancer treated with definitive chemoradiotherapy: a systematic review. Oral Oncol. 2018 Dec;87:77–81.
39 National Collaborating Centre for Cancer (UK). Cancer of the Upper Aerodigestive Tract: Assessment and Management in People Aged 16 and Over. National Institute for Health and Care Excellence: Clinical Guidelines. London: National Institute for Health and Care Excellence (UK);2016. [ Links ]
40 Silander E, Nyman J, Bove M, Johansson L, Larsson S, Hammerlid E. Impact of prophylactic percutaneous endoscopic gastrostomy on malnutrition and quality of life in patients with head and neck cancer: a randomized study. Head Neck. 2012 Jan;34(1):1–9.
41 Margolis M, Alexander P, Trachiotis GD, Gharagozloo F, Lipman T. Percutaneous endoscopic gastrostomy before multimodality therapy in patients with esophageal cancer. Ann Thorac Surg. 2003 Nov;76(5):1694–7.
42 Dittrich A, Schubert B, Kramer M, Lenz F, Kast K, Schuler U, et al. Benefits and risks of a percutaneous endoscopic gastrostomy (PEG) for decompression in patients with malignant gastrointestinal obstruction. Support Care Cancer. 2017 Sep;25(9):2849–56.
43 Fonseca J, Santos CA. Percutaneous endoscopic gastrostomy with jejunal extension plus percutaneous endoscopic gastrostomy (PEG-J plus PEG) in patients with gastric/duodenal cancer outlet obstruction. Arq Gastroenterol. 2015 Jan-Mar;52(1):72–5.
44 Maple JT, Petersen BT, Baron TH, Gostout CJ, Wong Kee Song LM, Buttar NS. Direct percutaneous endoscopic jejunostomy: outcomes in 307 consecutive attempts. Am J Gastroenterol. 2005 Dec;100(12):2681–8.
45 Given MF, Hanson JJ, Lee MJ. Interventional radiology techniques for provision of enteral feeding. Cardiovasc Intervent Radiol. 2005 Nov-Dec;28(6):692–703.
46 Sofue K, Takeuchi Y, Tsurusaki M, Shibamoto K, Sakamoto N, Kitajima K, et al. Value of Percutaneous Radiologic Gastrostomy for Patients with Advanced Esophageal Cancer. Ann Surg Oncol. 2016 Oct;23(11):3623–31.
47 Lim JH, Choi SH, Lee C, Seo JY, Kang HY, Yang JI, et al. Thirty-day mortality after percutaneous gastrostomy by endoscopic versus radiologic placement: a systematic review and meta-analysis. Intest Res. 2016 Oct;14(4):333–42.
48 Galaski A, Peng WW, Ellis M, Darling P, Common A, Tucker E. Gastrostomy tube placement by radiological versus endoscopic methods in an acute care setting: a retrospective review of frequency, indications, complications and outcomes. Can J Gastroenterol. 2009 Feb;23(2):109–14.
49 Bravo JG, Ide E, Kondo A, de Moura DT, de Moura ET, Sakai P, et al. Percutaneous endoscopic versus surgical gastrostomy in patients with benign and malignant diseases: a systematic review and meta-analysis. Clinics (São Paulo). 2016 Mar;71(3):169–78.
50 Oliveira GP, Santos CA, Fonseca J. The role of surgical gastrostomy in the age of endoscopic gastrostomy: a 13 years and 543 patients retrospective study. Rev Esp Enferm Dig. 2016 Dec;108(12):776–9.
51 Eeftinck Schattenkerk M, Obertop H, Bruining HA, van Rooyen W, van Houten H. Needle catheter jejunostomy (NCJ) for early postoperative feeding: experience in 210 patients. Neth J Surg. 1983 Dec;35(5):163–6.
52 Minarich MJ, Schwarz RE. Experience with a simplified feeding jejunostomy technique for enteral nutrition following major visceral operations. Transl Gastroenterol Hepatol. 2018 Jul;3:44. [ Links ]
53 Shin JH, Park AW. Updates on percutaneous radiologic gastrostomy/gastrojejunostomy and jejunostomy. Gut Liver. 2010 Sep;4 Suppl 1:S25–31.
54 Spaander MC, Baron TH, Siersema PD, Fuccio L, Schumacher B, Escorsell À, et al. Esophageal stenting for benign and malignant disease: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2016 Oct;48(10):939–48.
55 Philips P, North DA, Scoggins C, Schlegel M, Martin RC. Gastric-Esophageal Stenting for Malignant Dysphagia: Results of Prospective Clinical Trial Evaluation of Long-Term Gastroesophageal Reflux and Quality of Life-Related Symptoms. J Am Coll Surg. 2015 Jul;221(1):165–73.
56 Verschuur EM, Kuipers EJ, Siersema PD. Esophageal stents for malignant strictures close to the upper esophageal sphincter. Gastrointest Endosc. 2007 Dec;66(6):1082–90.
57 Mariette C, Gronnier C, Duhamel A, Mabrut JY, Bail JP, Carrere N, et al.; FREGAT Working Group–FRENCH–AFC; FREGAT Working Group-FRENCH-AFC. Self-expanding covered metallic stent as a bridge to surgery in esophageal cancer: impact on oncologic outcomes. J Am Coll Surg. 2015 Mar;220(3):287–96.
58 Mão-de-Ferro S, Serrano M, Ferreira S, Rosa I, Lage P, Alexandre DP, et al. Stents in patients with esophageal cancer before chemoradiotherapy: high risk of complications and no impact on the nutritional status. Eur J Clin Nutr. 2016 Mar;70(3):409–10.
59 Francis SR, Orton A, Thorpe C, Stoddard G, Lloyd S, Anker CJ. Toxicity and Outcomes in Patients with and without Esophageal Stents in Locally Advanced Esophageal Cancer. Int J Radiat Oncol Biol Phys. 2017 Nov;99(4):884–94.
60 Jones CM, Griffiths EA. Should oesophageal stents be used before neo-adjuvant therapy to treat dysphagia in patients awaiting oesophagectomy? Best evidence topic (BET). Int J Surg. 2014 Nov;12(11):1172–80.
61 Jeurnink SM, Steyerberg EW, van Hooft JE, van Eijck CH, Schwartz MP, Vleggaar FP, et al.; Dutch SUSTENT Study Group. Surgical gastrojejunostomy or endoscopic stent placement for the palliation of malignant gastric outlet obstruction (SUSTENT study): a multicenter randomized trial. Gastrointest Endosc. 2010 Mar;71(3):490–9.
62 van Heek NT, van Geenen RC, Busch OR, Gouma DJ. Palliative treatment in “peri”-pancreatic carcinoma: stenting or surgical therapy? Acta Gastroenterol Belg. 2002 Jul- Sep;65(3):171–5.
63 Pais-Cunha I, Castro R, Libânio D, Pita I, Bastos RP, Silva R, et al. Endoscopic stenting for palliation of intra-abdominal gastrointestinal malignant obstruction: predictive factors for clinical success. Eur J Gastroenterol Hepatol. 2018 Sep;30(9):1033–40.
64 Mendelsohn RB, Gerdes H, Markowitz AJ, DiMaio CJ, Schattner MA. Carcinomatosis is not a contraindication to enteral stenting in selected patients with malignant gastric outlet obstruction. Gastrointest Endosc. 2011 Jun;73(6):1135–40.
65 Mutignani M, Tringali A, Shah SG, Perri V, Familiari P, Iacopini F, et al. Combined endoscopic stent insertion in malignant biliary and duodenal obstruction. Endoscopy. 2007 May;39(5):440–7.
66 Baron TH. Management of simultaneous biliary and duodenal obstruction: the endoscopic perspective. Gut Liver. 2010 Sep;4 Suppl 1:S50–6.
67 van Hooft JE, Uitdehaag MJ, Bruno MJ, Timmer R, Siersema PD, Dijkgraaf MG, et al. Efficacy and safety of the new WallFlex enteral stent in palliative treatment of malignant gastric outlet obstruction (DUOFLEX study): a prospective multicenter study. Gastrointest Endosc. 2009 May;69(6):1059–66.
68 Masci E, Viale E, Mangiavillano B, Contin G, Lomazzi A, Buffoli F, et al. Enteral self-expandable metal stent for malignant luminal obstruction of the upper and lower gastrointestinal tract: a prospective multicentric study. J Clin Gastroenterol. 2008 Apr;42(4):389–94.
69 Jeurnink SM, van Eijck CH, Steyerberg EW, Kuipers EJ, Siersema PD. Stent versus gastrojejunostomy for the palliation of gastric outlet obstruction: a systematic review. BMC Gastroenterol. 2007 Jun;7:18. [ Links ]
70 Tringali A, Didden P, Repici A, Spaander M, Bourke MJ, Williams SJ, et al. Endoscopic treatment of malignant gastric and duodenal strictures: a prospective, multicenter study. Gastrointest Endosc. 2014 Jan;79(1):66–75.
71 Fiori E, Lamazza A, Demasi E, Decesare A, Schillaci A, Sterpetti AV. Endoscopic stenting for gastric outlet obstruction in patients with unresectable antro pyloric cancer. Systematic review of the literature and final results of a prospective study. The point of view of a surgical group. Am J Surg. 2013 Aug;206(2):210–7.
72 Dawod E, Nieto JM. Endoscopic ultrasound guided gastrojejunostomy. Transl Gastroenterol Hepatol. 2018 Nov;3:93. [ Links ]
73 Tyberg A, Perez-Miranda M, Sanchez-Ocaña R, Peñas I, de la Serna C, Shah J, et al. Endoscopic ultrasound-guided gastrojejunostomy with a lumen-apposing metal stent: a multicenter, international experience. Endosc Int Open. 2016 Mar;4(3):E276–81.
74 Perez-Miranda M, Tyberg A, Poletto D, Toscano E, Gaidhane M, Desai AP, et al. EUSguided Gastrojejunostomy versus Laparoscopic Gastrojejunostomy: An International Collaborative Study. J Clin Gastroenterol. 2017 Nov/Dec;51(10):896–9.
75 Bozzetti F. Nutritional support of the oncology patient. Crit Rev Oncol Hematol. 2013 Aug;87(2):172–200.
76 Matthews KL, Palmer MA, Capra SM. Dietitians opinions regarding refeeding syndrome, clinical guidelines and extended scope of practice. Nutr Diet. 2018 Sep;75(4):397–405.
77 Nunes G, Brito M, Santos CA, Fonseca J. Refeeding syndrome in the gastroenterology practice: how concerned should we be? Eur J Gastroenterol Hepatol. 2018 Nov;30(11):1270–6.
78 Boullata JI, Carrera AL, Harvey L, Escuro AA, Hudson L, Mays A, et al.; ASPEN Safe Practices for Enteral Nutrition Therapy Task Force, American Society for Parenteral and Enteral Nutrition. ASPEN Safe Practices for Enteral Nutrition Therapy. JPEN J Parenter Enteral Nutr. 2017 Jan;41(1):15–103.
79 August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009 Sep-Oct;33(5):472–500.
80 Jabbar A, McClave SA. Pre-pyloric versus post-pyloric feeding. Clin Nutr. 2005 Oct;24(5):719–26.
81 Arends J, Bodoky G, Bozzetti F, Fearon K, Muscaritoli M, Selga G, et al.; DGEM (German Society for Nutritional Medicine); ESPEN (European Society for Parenteral and Enteral Nutrition). ESPEN Guidelines on Enteral Nutrition: non-surgical oncology. Clin Nutr. 2006 Apr;25(2):245–59.
82 Stroud M, Duncan H, Nightingale J; British Society of Gastroenterology. Guidelines for enteral feeding in adult hospital patients. Gut. 2003 Dec;52 Suppl 7:vii1–12.
83 Niv E, Fireman Z, Vaisman N. Post-pyloric feeding. World J Gastroenterol. 2009 Mar;15(11):1281–8.
84 Silk DB. Formulation of enteral diets for use in jejunal enteral feeding. Proc Nutr Soc. 2008 Aug;67(3):270–2.
85 Papakostas P, Tsaousi G, Stavrou G, Rachovitsas D, Tsiropoulos G, Rova C, et al. Percutaneous endoscopic gastrostomy feeding of locally advanced oro-pharygo-laryngeal cancer patients: blenderized or commercial food? Oral Oncol. 2017 Nov;74:135–41.
86 Vieira MM, Santos VF, Bottoni A, Morais TB. Nutritional and microbiological quality of commercial and homemade blenderized whole food enteral diets for home-based enteral nutritional therapy in adults. Clin Nutr. 2018 Feb;37(1):177–81.
87 Mochamat CH, Cuhls H, Marinova M, Kaasa S, Stieber C, Conrad R, et al. A systematic review on the role of vitamins, minerals, proteins, and other supplements for the treatment of cachexia in cancer: a European Palliative Care Research Centre cachexia project. J Cachexia Sarcopenia Muscle. 2017 Feb;8(1):25–39.
88 Murphy RA, Mourtzakis M, Mazurak VC. n-3 polyunsaturated fatty acids: the potential role for supplementation in cancer. Curr Opin Clin Nutr Metab Care. 2012 May;15(3):246–51.
89 de Aguiar Pastore Silva J, Emilia de Souza Fabre M, Waitzberg DL. Omega-3 supplements for patients in chemotherapy and/or radiotherapy: a systematic review. Clin Nutr. 2015 Jun;34(3):359–66.
90 Weimann A, Braga M, Carli F, Higashiguchi T, Hübner M, Klek S, et al. ESPEN guideline: clinical nutrition in surgery. Clin Nutr. 2017 Jun;36(3):623–50.
Statement of Ethics
The authors have no ethical conflicts to disclose.
Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Sources
The authors have received no funding for the present paper.
* Corresponding author.
Gonçalo Nunes
Gastroenterology Department
Hospital Garcia de Orta, Av. Torrado da Silva
PT–2805-267 Almada (Portugal)
E-Mail goncalo.n@hotmail.com
Received: March 18, 2019; Accepted after revision: July 27, 2019
Author Contributions
G. Nunes performed the literature search, selected the bibliography and wrote the paper; A.T. Barata performed the literature search and selected the bibliography; J. Fonseca, P. Pimentel-Nunes and M. Dinis-Ribeiro conceived the structure of the paper and critically reviewed the text. All authors approved the final version of the paper.














